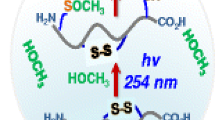
Abstract
Evidence for photo-induced radical disulfide bond scrambling in the gas phase during matrix-assisted laser desorption/ionization mass spectrometry (MALDI-MS) is described. The phenomenon was observed during the analysis of tryptic peptides from insulin and was confirmed in the determination of disulfide bonds in the rhamnose-binding lectin SEL24K from the Chinook salmon Oncorhynchus tshawytscha. A possible mechanism for this surprising scrambling is proposed. Despite this finding, the disulfide bond pattern in SEL24K was assigned unambiguously by a multi-enzyme digestion strategy in combination with MALDI mass spectrometry. The pattern was found to be symmetrical in the tandem repeat sequence of SEL24K. To the best of our knowledge, this is the first report of disulfide bond scrambling in the gas phase during MALDI-MS analysis. This observation has important ramifications for unambiguous assignment of disulfide bonds.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wu, J.; Watson, J. T. A novel methodology for assignment of disulfide bond pairings in proteins. Protein Sci. 1997, 6, 391–398.
Gallegos-Perez, J.-L.; Rangel-Ordonez, L.; Bowman, S. R.; Ngowe, C. O.; Watson, J. T. Study of primary amines for nucleophilic cleavage of cyanylated cystinyl proteins in disulfide mass mapping methodology. Anal. Biochem. 2005, 346, 311–319.
Wefing, S.; Schnaible, V.; Hoffmann, D. SearchXLinks: A Program for the Identification of Disulfide Bonds in Proteins from Mass Spectra. Anal. Chem. 2006, 78, 1235–1241.
Ryle, A. P.; Sanger, F. Disulfide interchange reactions. Biochem. J. 1955, 60, 535–540.
Bach, R. D.; Dmitrenko, O.; Thorpe, C. Mechanism of Thiolate-Disulfide Interchange Reactions in Biochemistry. J. Org. Chem. 2008, 73, 12–21.
Patterson, S. D.; Katta, V. Prompt fragmentation of disulfide-linked peptides during matrix-assisted laser desorption ionization mass spectrometry. Anal. Chem. 1994, 66(21), 3727–3732.
Fukuyama, Y.; Iwamoto, S.; Tanaka, K. Rapid sequencing and disulfide mapping of peptides containing disulfide bonds by using 1,5-diaminonaphthalene as a reductive matrix. J. Mass Spectrom. 2006, 41, 191–201.
Yi, G.-S.; Cheong, C. Disulfide bond formation between the N-terminal region of P56LCK and the cytoplasmic domain of CD8 studied by electrospray ionization and matrix-assisted desorption/ionization time-of-flight mass spectrometry. Biochem. Mol. Biol. Int. 1996, 40, 409–416.
Gorman, J. J.; Ferguson, B. L.; Speelman, D.; Mills, J. Determination of the disulfide bond arrangement of human respiratory syncytial virus attachment (G) protein by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Protein Sci. 1997, 6, 1308–1315.
Cosenza, L.; Sweeney, E.; Murphy, J. R. Disulfide bond assignment in human interleukin-7 by matrix-assisted laser desorption/ionization mass spectroscopy and site-directed cysteine to serine mutational analysis. J. Biol. Chem. 1997, 272, 32995–33000.
Navale, V.; Kaushal, P.; Hunt, S.; Burducea, I.; Gentz, R.; Khan, F.; Vertes, A. Peptide mapping and disulfide bond analysis of myeloid progenitor inhibitory chemokine and keratinocyte growth factor by matrix-assisted laser desorption ionization mass spectrometry. Anal. Biochem. 1999, 267, 125–134.
Watson, J. T.; Sparkman, O. D. Introduction to Mass Spectrometry, Instrumentation, Applications, and Strategies for Data Interpretation; 4th ed.; John Wiley and Sons: Hoboken, NJ, 2007, p 726–734.
Lioe, H.; O’Hair, R. A. J. A Novel Salt Bridge Mechanism Highlights the Need for Nonmobile Proton Conditions to Promote Disulfide Bond Cleavage in Protonated Peptides Under Low-Energy Collisional Activation. J. Am. Soc. Mass Spectrom. 2007, 18, 1109–1123.
Fagerquist, C. K. Collision-activated cleavage of a peptide/antibiotic disulfide linkage: Possible evidence for intramolecular disulfide bond rearrangement upon collisional activation. Rapid Commun. Mass Spectrom. 2004, 18, 685–700.
Zhang, J.; Frankevich, V.; Knochenmuss, R.; Friess, S. D.; Zenobi, R. Reduction of Cu(II) in matrix-assisted laser desorption/ionization mass spectrometry. J. Am. Soc. Mass Spectrom. 2003, 14, 42–50.
Qiao, L.; Roussel, C.; Wan, J.; Kong, J.; Yang, P.; Girault, H. H.; Liu, B. MALDI in-source photo-oxidation reactions for online peptide tagging. Angew. Chem. Int. Edit. 2008, 47, 2646–2648.
Murata, K.; Fisher, A. J.; Hedrick, J. L. Crystallization and X-ray analysis of the salmon-egg lectin SEL24K. Acta Crystallogr. F 2007, F63(5), 396–398.
Zenner, B. D. The isolation and characterization of an egg lectin from king salmon, Oncorhynchus tshawytscha. Ph.D. Thesis, University of California, Davis, 1991.
Becker, H. D. Photochemical reactions with phenols: VI. Photo+ sensitized reduction and addition reactions of quinoid compounds. J. Org. Chem. 1969, 34, 2472–2474.
Steenken, S.; Neta, P. One-electron redox potentials of phenols: Hydroxy-and aminophenols and related compounds of biological interest. J. Phys. Chem. 1982, 86(18), 3661–3667.
De Kok, T. M. C. M.; Van Maanen, J. M. S.; Lankelma, J.; Ten Hoor, F.; Kleinjans, J. C. S. Electron spin resonance spectroscopy of oxygen radicals generated by synthetic fecapentaene-12 and reduction of FEC pentaene mutagenicity to Salmonella typhimurium by hydroxyl radical scavenging. Carcinogenesis 1992, 13(7), 1249–1255.
Mormann, M.; Eble, J.; Schwoeppe, C.; Mesters, R. M.; Berdel, W. E.; Peter-Katalinic, J.; Pohlentz, G. Fragmentation of intra-peptide and inter-peptide disulfide bonds of proteolytic peptides by nanoESI collision-induced dissociation. Anal. Bioanal. Chem. 2008, 392(5), 831–838.
Yu, H.; Murata, K.; Hedrick, J. L.; Almaraz, R.; Xiang, F.; Franz, A. H. The Disulfide Bond Pattern of Salmon Egg Lectin 24K from the Chinook salmon Oncorhynchus tshawytscha. Arch. Biochem. Biophys. 2007, 463, 1–11.
Terada, T.; Watanabe, Y.; Tateno, H.; Naganuma, T.; Ogawa, T.; Muramoto, K.; Kamiya, H. Structural Characterization of a Rhamnose-Binding Glycoprotein (lectin) from Spanish Mackerel Scomberomorous niphonius eggs. Biochim. Biophys. Acta 2007, 1770(4), 617–629.
Tateno, H.; Ogawa, T.; Muramoto, K.; Kamiya, H.; Hirai, T.; Saneyoshi, M. A novel rhamnose-binding lectin family from eggs of steelhead trout Oncorhynchus mykiss with different structures and tissue distribution. Biosci. Biotechnol. Biochem. 2001, 65, 1328–1338.
Gorman, J. J.; Wallis, T. P.; Pitt, J. J. Protein disulfide bond determination by mass spectrometry. Mass Spectrom. Rev. 2002, 21, 183–216.
Gilbert, B. C. Structure and reaction mechanisms in sulfur-radical chemistry revealed by ESR spectroscopy: NATO ASI Series, Series A: Life Sciences. Sulfur-Cent. React. Intermed. Chem. Biol. 1990, 197, 135–154.
Russell, G. A.; Ngoviwatchai, P. Free radical processes in the reactions of organometallics with organic sulfur compounds: NATO ASI Series, Series A: Life Sciences. Sulfur-Cent. React. Intermed. Chem. Biol. 1990, 197, 291–302.
Asmus, K. D. Stabilization of oxidized sulfur centers in organic sulfides: Radical cations and odd-electron sulfur-sulfur bonds. Acc. Chem. Res. 1979, 12, 436–442.
Benson, S. W. Thermochemistry and kinetics of sulfur-containing molecules and radicals. Chem. Rev. 1978, 78, 23–35.
Hadley J. H., Jr.; Gordy, W. Nuclear coupling of sulfur-33 and the nature of free radicals in irradiated crystals of N-acetyl-L-cysteine. Proc. Natl. Acad. Sci. U.S.A. 1977, 74, 216–220.
Hadley, J. H., Jr.; Gordy, W. Nuclear coupling of sulfur-33 and the nature of free radicals in irradiated crystals of cysteine hydrochloride and N-acetyl methionine. Proc. Natl. Acad. Sci. U.S.A. 1975, 72, 3486–3490.
Vanhooren, A.; Devreese, B.; Vanhee, K.; Van Beeumen, J.; Hanssens, I. Photoexcitation of tryptophan groups induces reduction of two disulfide bonds in goat β-lactalbumin. Biochemistry 2002, 41, 11035–11043.
Kubwabo, C.; Vais, N.; Benoit, F. M. Characterization of microcystins using in-source collision-induced dissociation. Rapid Commun. Mass Spectrom. 2005, 19, 597–604.
Bure, C.; Le Falher, G.; Lange, C.; Delmas, A. Fragmentation study of peptide acetals and aldehydes using in-source collision-induced dissociation. J. Mass Spectrom. 2004, 39, 817–823.
Williams, J. D.; Flanagan, M.; Lopez, L.; Fischer, S.; Miller, L. A. D. Using accurate mass electrospray ionization-time-of-flight mass spectrometry with in-source collision-induced dissociation to sequence peptide mixtures. J. Chromatogr. A 2003, 1020, 11–26.
Musker, W. K.; Gorewit, B. V.; Roush, P. B.; Wolford, T. L. Formation of cyclic disulfide cation radicals in the electron impact induced fragmentation of mesocyclic dithioethers. J. Org. Chem. 1978, 43, 3235–3236.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online May 13, 2009
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Zhao, L., Almaraz, R.T., Xiang, F. et al. Gas-phase scrambling of disulfide bonds during matrix-assisted laser desorption/ionization mass spectrometry analysis. J Am Soc Mass Spectrom 20, 1603–1616 (2009). https://doi.org/10.1016/j.jasms.2009.04.021
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2009.04.021