Abstract
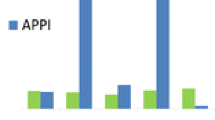
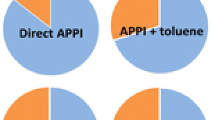
The role of propionitrile in the production of [M+H]+ under atmospheric pressure photoionization (APPI) was investigated. In dopant-assisted APPI using acetone and anisole, protonated acetone and anisole radical cations were the most prominent ions observed. In dopant-free or direct APPI in acetonitrile, however, a major ion in acetonitrile was detected and identified as propionitrile, using high accuracy mass measurement and collision induced dissociation studies. Vaporizing ca. 10−5 M althiazide and bendroflumethazide under direct APPI in acetonitrile produced their corresponding protonated species [M+H]+. In addition to protonated acetonitrile, its dimers, and acetonitrile/water clusters, protonated propionitrile, propionitrile dimer, and propionitrile/water clusters were also observed. The role of propionitrile, an impurity in acetonitrile and/or a possible product of ion-molecule reaction, in the production of [M+H]+ of althiazide and bendroflumethazide was further investigated in the absence of dopant using propionitrile-d5. The formation of [M+D]+ species was observed, suggesting a possible role of propionitrile in the protonation process. Additionally, an increase in the [M+H]+ signal of althiazide and bendroflumethazide was observed as a function of propionitrile concentration in acetonitrile. Theoretical data from the literature supported the assumption that one possible mechanism, among others, for the formation of [M+H]+ could be attributed to photo-initiated isomerization of propionitrile. The most stable isomers of propionitrile, based on their calculated ionization energy (IE) and relative energy (ΔE), were assumed to undergo proton transfer to the analytes, and mechanisms were proposed.
Article PDF
Similar content being viewed by others
References
Robb, D. B.; Covey, T. R.; Bruins, A. P. Atmospheric Pressure Photoionization: An Ionization Method for Liquid Chromatography-Mass Spectrometry. Anal. Chem. 2000, 72(15), 3653–3659.
Syage, J. A.; Evans, M. D. Photoionization Mass Spectrometry as a Powerful New Tool for Drug Discovery. Spectroscopy 2001, 16(11), 14–21.
Hanold, K. A.; Fischer, S. M.; Cormia, P. H.; Miller, C. E.; Syage, J. A. Atmospheric Pressure Photoionization. 1. General Properties for LC/MS. Anal. Chem. 2004, 76(10), 2842–2851.
Hsieh, Y.; Merkle, K.; Wang, G.; Brisson, Jean-Marc; Korfmacher, W. A. High-Performance Liquid Chromatography-Atmospheric Pressure Photoionization/Tandem Mass Spectrometric Analysis for Small Molecules in Plasma. Anal. Chem. 2003, 75(13), 3122–3127.
Hsieh, Y.; Merkle, K.; Wang, G. Zirconia-Based Column High-Performance Liquid Chromatography/Atmospheric Pressure Photoionization Tandem Mass Spectrometric Analyses of Drug Molecules in Rat Plasma. Rapid Commun. Mass Spectrom. 2003, 17(15), 1775–1780.
Takino, M.; Daishima, S.; Nakahara, T. Determination of Perfluoro-Octane Sulfonate in River Water by Liquid Chromatography/Atmospheric Pressure Photoionization Mass Spectrometry by Automated On-Line Extraction Using Turbulent Flow Chromatography. Rapid Commun. Mass Spectrom. 2003, 17(5), 383–390.
Takino, M.; Daishima, S.; Nakahara, T. Determination of Chloramphenicol Residues in Fish Meats by Liquid Chromatography-Atmospheric Pressure Photoionization Mass Spectrometry. J. Chromatogr. A 2003, 1011(1/2), 67–75.
Kauppila, T. J.; Kuuranne, T.; Meurer, E. C.; Eberlin, M. N.; Kotiaho, T.; Kostiainen, R. Atmospheric Pressure Photoionization Mass Spectrometry: Ionization Mechanism and the Effect of Solvent on the Ionization of Naphthalenes. Anal. Chem. 2002, 74(21), 5470–5479.
Constapel, M.; Schellentrager, M.; Schmitz, O. J.; Gab, S.; Brockmann, K. J.; Giese, R.; Benter, Th Atmospheric Pressure Laser Ionization: A Novel Ionization Method for Liquid Chromatography/Mass Spectrometry. Rapid Commun. Mass Spectrom. 2005, 19(3), 326–336.
Syage, J. A. Mechanism of [M+H]+ Formation in Photoionization Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2004, 15(11), 1521–1533.
Kauppila, T. J.; Kostiainen, R.; Bruins, A. P. Anisole, a New Dopant for Atmospheric Pressure Photoionization Mass Spectrometry of Low Proton Affinity, Low Ionization Energy Compounds. Rapid Commun. Mass Spectrom. 2004, 18(7), 808–815.
Bos, S. J.; Leeuwen, S. M.; Karst, U. From Fundamentals to Applications: Recent Developments in Atmospheric Pressure Photoionization Mass Spectrometry. Anal. Bioanal. Chem. 2006, 384(1), 85–99.
Kauppila, T. J.; Bruins, A. P.; Kostiainen, R. Effect of the Solvent Flow Rate on the Ionization Efficiency in Atmospheric Pressure Photoionization-Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2005, 16(8), 1399–1407.
Kauppila, T. J.; Kotiaho, T.; Kostiainen, R.; Bruins, A. P. Negative Ion-Atmospheric Pressure Photoionization-Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2004, 15(2), 203–211.
Raffaelli, A.; Saba, A. Atmospheric Pressure Photoionization Mass Spectrometry. Mass Spectrom. Rev. 2003, 22(5), 318–331.
Song, L.; Wellman, A. D.; Yao, H.; Bartmess, J. E. Negative Ion-Atmospheric Pressure Photoionization: Electron Capture, Dissociative Electron Capture Proton Transfer and Anion Attachment. J. Am. Soc. Mass Spectrom. 2007, 18(10), 1789–1798.
Short, L. C.; Hanold, K. A.; Cai Sheng-Suan Syage, J. A. APPI-MS: Effects of Mobile Phases and VUV Lamps on the Detection of PAH Compounds. J. Am. Soc. Mass Spectrom. 2007, 18(4), 589–599.
Purcell, J. M.; Hendrickson, C. L.; Rodgers, R. P.; Marshall, A. G. Atmospheric Pressure Photoionization Proton Transfer for Complex Organic Mixtures Investigated by Fourier Transform Ion Cyclotron Resonance Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2007, 18(9), 1682–1689.
Short, L. C.; Hanold, K. A.; Cai, S.-S.; Syage, J. A. Electrospray Ionization/Atmospheric Pressure Photoionization Multimode Source for Low-Flow Liquid Chromatography/Mass Spectrometric Analysis. Rapid Commun. Mass Spectrom. 2007, 21(10), 1561–1566.
Marotta, E.; Traldi, P. On the Photo-Initiated Isomerization of Acetonitrile. Rapid Commun. Mass Spectrom. 2003, 17(24), 2846–2848.
NIST Chemistry Webbook, NIST Standard Reference Database Number 69, Mallard WG, Linstrom PJ (eds). National Institute of Standards and Technology, Gaithersburg, MD, June 2005. Available: http:// webbook.nist.gov/chemistry.
Kenny, G.; Miles, H. Purification of Acetonitrile. UK Pat. Appl. 1992, Patent GB2249308.
Kamel, A.; Jeanville, P.; J-Rivera, L. E.; Colizza, K. Collisionally-Induced Dissociation of Propionitrile Under APPI Mass Spectrometry: Evidence of an Intramolecular 1,3-Hydrogen Shift and Hydrogen Migration. Proceedings of the 55th ASMS Conference on Mass Spectrometry and Allied Topics; Indianapolis, IN, 2007.
Meyerson, S.; Karabatsos, G. J. 1,1-Elimination of Hydrogen Cyanide from Propionitrile Under Electron Impact: Hydrogen Migration in the Molecular Ion. Org. Mass Spectrom. 1974, 8, 289–295.
Franklin, J. L.; Wada, Y.; Natalis, P.; Hierl, P. M. Ion-Molecule Reactions in Acetonitrile and Propionitrile. J. Phys. Chem. 1966, 70(7), 2353–2361.
Salpin, J. Y.; Nguyen, M. T.; Bouchoux, G.; Gerbaux, P.; Flammang, R. Isomerization of Acetonitrile N-Methylide [CH3CNCH2]+ and N-Methylketenimine [CH3NCCH2]+ Radical Cations in the Gas Phase: Theoretical Study of the [C3,H5,N]+ Potential Energy Surface. J. Phys. Chem. A 1999, 103(7), 938–948.
Sugie, M.; Takeo, H.; Matsumura, C. Ab Initio Molecular Orbital Calculations of C3H5N Molecules. Kagaku Gijutsu Kenkyusho Hokoku 1989, 84(8), 503–510.
Meot-Ner, M.; Karpas Zeev, D.; Carol, A. Ion Chemistry of Cyanides and Isocyanides. 1. The Carbon Lone Pair as Proton Acceptor Proton Affinities of Isocyanides Alkyl Cation Nitrogen, Oxygen, and Carbon Lone Pair Donors. J. Am. Chem. Soc. 1986, 108(14), 3913–3919.
Wang, D.; Qian, X. Determination of the Kinetic Parameters of the Isomerization Reaction of Ethyl Isocyanide Using HeI Photoelectron Spectroscopy. J. Electron. Spectrosc. Relat. Phenom. 1997, 83(1), 41–44.
Maloney, K. M.; Rabinovitch, B. S. Thermal Isomerization of Isocyanides. Variation of Molecular Parameters. Ethyl Isocyanide. J. Phys. Chem. 1969, 73(6), 1652–1666.
Schneider, F. W.; Rabinovitch, B. S. The Thermal Unimolecular Isomerization of Methyl Isocyanide: Fall-Off Behavior. J. Am. Chem. Soc. 1962, 84, 4215–4230.
Wang, D.; Qian, X.; Zhang, Q. The Isonitrile-Nitrile Isomerization: Kinetic Parameters, Reaction Mechanism, and Relative Photoionization Cross-Section of the HOMO: A HeI Photoelectron Spectroscopy Study. Chem. Phys. Lett. 1997, 266(5/6), 560–567.
Casanova, J. Jr.; Werner, N. D.; Schuster, R. E. Isonitrile-Nitrile Isomerization. J. Org. Chem. 1966, 31(11), 3473–3482.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online July 3, 2008
Rights and permissions
About this article
Cite this article
Kamel, A., Jeanville, P., Colizza, K. et al. Mechanism of [M+H]+ formation in atmospheric pressure photoionization mass spectrometry: Identification of propionitrile in acetonitrile with high mass accuracy measurement and tandem mass spectrometry and evidence for its involvement in the protonation phenomenon. J Am Soc Mass Spectrom 19, 1579–1589 (2008). https://doi.org/10.1016/j.jasms.2008.06.027
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2008.06.027