Abstract
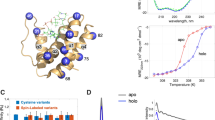
Electrospray ionization mass spectrometry (ESI-MS) can be used to monitor conformational changes of proteins in solution based on the charge state distribution (CSD) of the corresponding gas-phase ions, although relatively few studies of acidic proteins have been reported. Here, we have compared the CSD and solution structure of recombinant Vibrio harveyi acyl carrier protein (rACP), a small acidic protein whose secondary and tertiary structure can be manipulated by pH, fatty acylation, and site-directed mutagenesis. Circular dichroism and intrinsic fluorescence demonstrated that apo-rACP adopts a folded helical conformation in aqueous solution below pH 6 or in 50% acetonitrile/0.1% formic acid, but is unfolded at neutral and basic pH values. A rACP mutant, in which seven conserved acidic residues were replaced with their corresponding neutral amides, was folded over the entire pH range of 5 to 9. However, under the same solvent conditions, both wild type and mutant ACPs exhibited similar CSDs (6+–9+ species) at all pH values. Covalent attachment of myristic acid to the phosphopantetheine prosthetic group of rACP, which is known to stabilize a folded conformation in solution, also had little influence on its CSD in either positive or negative ion modes. Overall, our results are consistent with ACP as a “natively unfolded” protein in a dynamic conformational equilibrium, which allows access to (de)protonation events during the electrospray process.
Article PDF
Similar content being viewed by others
References
Eyles, S. J.; Kaltashov, I. A. Methods to Study Protein Dynamics and Folding by Mass Spectrometry. Methods 2004, 34, 88–99.
Heck, A. J. R.; van den Heuvel, R. H. H. Investigation of Intact Protein Complexes by Mass Spectrometry. Mass Spectrom. Rev. 2004, 23, 368–389.
Konermann, L.; Simmons, D. A. Protein-Folding Kinetics and Mechanisms Studied by Pulse-Labeling and Mass Spectrometry. Mass Spectrom. Rev. 2003, 22, 1–26.
Maier, C. S.; Deinzer, M. L. Protein Conformations, Interactions, and H/D Exchange. Methods Enzymol. 2005, 402, 312–360.
Yan, X.; Watson, J.; Ho, P. S.; Deinzer, M. L. Mass Spectrometric Approaches Using Electrospray Ionization Charge States and Hydrogen-Deuterium Exchange for Determining Protein Structures and Their Conformational Changes. Mol. Cell. Proteomics 2004, 3, 10–23.
Chowdhury, S. K.; Katta, V.; Chait, B. T. Probing Conformational Changes in Proteins by Mass Spectrometry. J. Am. Chem. Soc. 1990, 112, 9012–9013.
Katta, V.; Chait, B. T. Conformational Changes in Proteins Probed by Hydrogen-Exchange Electrospray-Ionization Mass Spectrometry. Rapid Commun. Mass Spectrom. 1991, 5, 214–217.
Loo, J. A.; Edmonds, C. G.; Udseth, H. R.; Smith, R. D. Effect of Reducing Disulfide-Containing Proteins on Electrospray Ionization Mass Spectra. Anal. Chem. 1990, 62, 693–698.
Samalikova, M.; Grandori, R. Role of Opposite Charges in Protein Electrospray Ionization Mass Spectrometry. J. Mass Spectrom. 2003, 38, 941–947.
Kaltashov, I. A.; Mohimen, A. Estimates of Protein Surface Areas in Solution by Electrospray Ionization Mass Spectrometry. Anal. Chem. 2005, 77, 5370–5379.
Samalikova, M.; Matecko, I.; Muller, N.; Grandori, R. Interpreting Conformational Effects in Protein Nano-ESI-MS Spectra. Anal. Bioanal. Chem. 2004, 378, 1112–1123.
Vaidyanathan, S.; Kell, D. B.; Goodacre, R. Selective Detection of Proteins in Mixtures Using Electrospray Ionization Mass Spectrometry: Influence of Instrumental Settings and Implications for Proteomics. Anal. Chem. 2004, 76, 5024–5032.
Grandori, R.; Matecko, I.; Muller, N. Uncoupled Analysis of Secondary and Tertiary Protein Structure by Circular Dichroism and Electrospray Ionization Mass Spectrometry. J. Mass Spectrom. 2002, 37, 191–196.
Konermann, L.; Douglas, D. J. Acid-Induced Unfolding of Cytochrome c at Different Methanol Concentrations: Electrospray Ionization Mass Spectrometry Specifically Monitors Changes in the Tertiary Structure. Biochemistry 1997, 36, 12296–12302.
Konermann, L.; Douglas, D. J. Unfolding of Proteins Monitored by Electrospray Ionization Mass Spectrometry: A Comparison of Positive and Negative Ion Modes. J. Am. Soc. Mass Spectrom. 1998, 9, 1248–1254.
Watt, S. J.; Oakley, A.; Sheil, M. M.; Beck, J. L. Comparison of Negative and Positive Ion Electrospray Ionization Mass Spectra of Calmodulin and Its Complex with Trifluoperazine. Rapid Commun. Mass Spectrom. 2005, 19, 2123–2130.
Veenstra, T. D.; Johnson, K. L.; Tomlinson, A. J.; Naylor, S.; Kumar, R. Determination of Calcium-Binding Sites in Rat Brain Calbindin D28K by Electrospray Ionization Mass Spectrometry. Biochemistry 1997, 36, 3535–3542.
Johnson, K. A.; Verhagen, M. F. J. M.; Adams, M. W. W.; Amster, I. J. Differences Between Positive and Negative Ion Stabilities of Metal-Sulfur Cluster Proteins: An Electrospray Ionization Fourier Transform Ion Cyclotron Resonance Study. Int. J. Mass Spectrom. 2001, 204, 77–85.
White, S. W.; Zheng, J.; Zhang, Y. M.; Rock, C. O. The Structural Biology of Type II Fatty Acid Biosynthesis. Annu. Rev. Biochem. 2005, 74, 791–831.
Roujeinikova, A.; Baldock, C.; Simon, W. J.; Gilroy, J.; Baker, P. J.; Stuitje, A. R.; Rice, D. W.; Slabas, A. R.; Rafferty, J. B. X-Ray Crystallographic Studies on Butyryl-ACP Reveal Flexibility of the Structure Around a Putative Acyl Chain Binding Site. Structure (Camb.) 2002, 10, 825–835.
Zornetzer, G. A.; Fox, B. G.; Markely, J. L. Solution Structures of Spinach Acyl Carrier Protein with Decanoate and Stearate. Biochemistry 2006, 45, 5217–5227.
de la Roche, M. A.; Shen, Z.; Byers, D. M. Hydrodynamic Properties of Vibrio harveyi Acyl Carrier Protein and Its Fatty-Acylated Derivatives. Arch. Biochem. Biophys. 1997, 344, 159–164.
Flaman, A. S.; Chen, J. M.; Van Iderstine, S. C.; Byers, D. M. Site-Directed Mutagenesis of Acyl Carrier Protein (ACP) Reveals Amino Acid Residues Involved in ACP Structure and Acyl-ACP Synthetase activity. J. Biol. Chem. 2001, 276, 35934–35939.
Keating, M. M.; Gong, H.; Byers, D. M. Identification of a Key Residue in the Conformational Stability of Acyl Carrier Protein. Biochim. Biophys. Acta 2002, 1601, 208–214.
Shen, Z.; Fice, D.; Byers, D. M. Preparation of Fatty-Acylated Derivative of Acyl Carrier Protein Using Vibrio harveyi Acyl-ACP Synthetase. Anal. Biochem. 1992, 204, 34–39.
Gong, H.; Murphy, A.; McMaster, C. R.; Byers, D. M. Neutralization of Acidic Residues in Helix II Stabilizes the Folded Conformation of Acyl Carrier Protein and Variably Alters Its Function with Different Enzymes. J. Biol. Chem. 2007, 282, 4494–4503.
Pettersen, E. F.; Goddard, T. D.; Huang, C. C.; Couch, G. S.; Greenblatt, D. M.; Meng, E. C.; Ferrin, T. E. UCSF. Chimera—A Visualization System for Exploratory Research and Analysis. J. Comput. Chem. 2004, 25, 1605–1612.
Mayo, K. H.; Prestegard, J. H. Acyl Carrier Protein from Escherichia coli: Structural Characterization of Short-Chain Acylated Acyl Carrier Proteins by NMR. Biochemistry 1985, 24, 7834–7838.
Rock, C. O.; Cronan, J. E., Jr.; Armitage, I. M. Molecular Properties of Acyl Carrier Protein Derivatives. J. Biol. Chem. 1981, 256, 2669–2674.
Schulz, H. On the Structure-Function Relationship of Acyl Carrier Protein of Escherichia coli. J. Biol. Chem. 1975, 250, 2299–2304.
Abzalimov, R. R.; Frimpong, A. K.; Kaltashov, I. A. Gas-Phase Processes and Measurements of Macromolecular Properties in Solution: On the Possibility of False Positive and False Negative Signals of Protein Unfolding. Int. J. Mass Spectrom. 2006, 253, 207–216.
Horvath, L. A.; Sturtevant, J. M.; Prestegard, J. H. Kinetics and Thermodynamics of Thermal Denaturation in Acyl Carrier Protein. Protein Sci. 1994, 3, 103–108.
Fink, A. L. Natively Unfolded Proteins. Curr. Opin. Struct. Biol. 2005, 15, 35–41.
Wright, P. E.; Dyson, H. J. Intrinsically Unstructured Proteins: Reassessing the Protein Structure-Function Paradigm. J. Mol. Biol. 1999, 293, 321–331.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online May 24, 2007
Rights and permissions
About this article
Cite this article
Murphy, P.W., Rowland, E.E. & Byers, D.M. Electrospray ionization mass spectra of acyl carrier protein are insensitive to its solution phase conformation. J Am Soc Mass Spectrom 18, 1525–1532 (2007). https://doi.org/10.1016/j.jasms.2007.05.012
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2007.05.012