Abstract
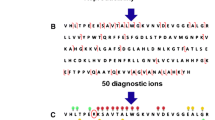
Many human hemoglobin variants occur in heterozygotes; that is, the variant and normal hemoglobins are present in the same sample. In a procedure for rapidly identifying such variants by mass spectrometry, mutations that increase the mass by 1 Da require a special approach. One of the steps in this procedure involves digesting the denatured hemoglobin with trypsin and analyzing the resulting peptide mixture by mass spectrometry to identify the mutant peptide. Generally the mutant peptide ion can then be selected as the precursor and sequenced by tandem mass spectrometry to identify or confirm the mutation. However, with heterozygotes in which the mass of the variant is 1 Da higher than normal, the first isotope of the mutant peptide occurs at essentially the same mass as the second isotope of the normal peptide, precluding analysis of the mutant peptide on its own. Product ions from the second isotope of a peptide are doublets, 1 Da apart. The way in which the relative abundance of the components in these doublets varies with the elemental composition of the product ions was predicted from the isotopic abundance of the elements and agreed well with experimental data. These results were applied to the identification of a variant that increases the mass by 1 Da in a heterozygote—that is, β131 Gln→Glu, hemoglobin Camden.
Article PDF
Similar content being viewed by others
References
Wild, B. J.; Green, B. N.; Cooper, E. K.; Lalloz, M. R. A.; Erten, S.; Stephens, A. D.; Layton, D. M. Rapid Identification of Hemoglobin Variants by Electrospray Ionization Mass Spectrometry. Blood Cells Mol. Dis. 2001, 27, 691–704.
Rai, D. K.; Griffiths, W. J.; Landin, B.; Wild, B. J.; Alvelius, G.; Green, B. N. Accurate Mass Measurement by Electrospray Ionization Quadrupole Mass Spectrometry: Detection of Variants Differing by <6 Da from Normal in Human Hemoglobin Heterozygotes. Anal. Chem. 2003, 75, 1978–1982.
Bozorgzadeh, M. H.; Morgan, R. P.; Beynon, J. H. Application of Mass-analysed Ion Kinetic Energy Spectrometry (MIKES) to the Determination of the Structures of Unknown Compounds. Analyst 1978, 103, 613–622.
Bateman, R. H.; Green, M. R.; Scott, G.; Clayton, E. A Combined Magnetic Sector-Time-of-Flight Mass Spectrometer for Structural Determination Studies by Tandem Mass Spectrometry. Rapid Commun. Mass Spectrom. 1995, 9, 1227–1233.
Cohen, P. T.; Yates, A.; Bellingham, A. J.; Huehns, E. R. Amino-Acid Substitution in the α1β1 Intersubunit Contact of Haemoglobin-Camden β131 (H9) Gln→Glu. Nature 1973, 243, 467.
Beynon, J. H. Mass Spectrometry and Its Applications to Organic Chemistry; Elsevier Publishing: Amsterdam, 1960; pp 294–297.
de Laeter, J. R.; Böhlke, J. K.; de Bièvre, P.; Hidaka, H.; Peiser, H. S.; Rosman, K. J. R.; Taylor, P. D. P. IUPAC Commission on Atomic Weights and Isotopic Abundances, Atomic Weights of the Elements: Review 2000. Pure Appl. Chem. 2003, 75, 683–800.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online May 17, 2007
Rights and permissions
About this article
Cite this article
Green, B.N., Williams, J.P. Prediction of product ion isotope ratios in the tandem electrospray ionization mass spectra from the second isotope of tryptic peptides: Identification of the variant β131 Gln→Glu, hemoglobin Camden. J Am Soc Mass Spectrom 18, 1493–1498 (2007). https://doi.org/10.1016/j.jasms.2007.05.005
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2007.05.005