Abstract
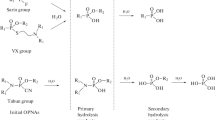
Bis(2-hydroxyethylthio)alkanes and bis(2-hydroxyethylthioalkyl)ethers are important biological and environmental degradation products of sulfur mustard analogs known as sesqui- and oxy-mustards. We used atmospheric pressure chemical ionization mass spectrometry (APCI MS) to acquire characteristic spectra of these compounds in positive and negative ionization modes. Positive APCI mass spectra exhibited [M + H]+; negative APCI MS generated [M + O2]−, [M − H]−, and [M − 3H]−; and both positive and negative APCI mass spectra contained fragment ions due to in-source collision-induced dissociation. Product ion scans confirmed the origin of fragment ions observed in single-stage MS. Although the spectra of these compounds were very similar, positive and negative APCI mass spectra of the oxy-mustard hydrolysis product, bis(2-hydroxyethylthiomethyl)ether, differed from the spectra of the other compounds in a manner that suggested a rearrangement to the sesqui-mustard hydrolysis product, bis(2-hydroxyethylthio)methane. We evaluated the [M + O2]− adduct ion for quantification via liquid chromatography-MS/MS in the multiple-reaction monitoring (MRM) mode by constructing calibration curves from three precursor/product ion transitions for all the analytes. Analytical figures of merit generated from the calibration curves indicated the stability and suitability of these transitions for quantification at concentrations in the low ng/mL range. Thus, we are the first to propose a quantitative method predicated on the measurement of product ions generated from the superoxide adduct anion of the sesqui-and oxy-mustard hydrolysis products.
Article PDF
Similar content being viewed by others
References
Report of the Mission Dispatched by the Security-General to Investigate Allegations of the Use of Chemical Weapons in the Conflict Between the Islamic Republic of Iran and Iraq; United Nations Report S/17911: New York, 1986.
Balali-Mood, M.; Hefazi, M. The Clinical Toxicology of Sulfur Mustard. Arch. Iranian Med. 2005, 8(3), 162–179.
Convention on the Prohibition of the Development, Production, Stockpiling, and Use of Chemical Weapons and on their Destruction; The Technical Secretariat of the Organization for the Prohibition of Chemical Weapons: The Hague, The Netherlands, 1992.
Bennett, G. M.; Whincop, E. M. Some Derivations of Monothioethlyene Glycol. J. Chem. Soc. Trans. 1921, 1860–1864.
Munro, N. B.; Talmage, S. S.; Griffin, G. D.; Waters, L. C.; Watson, A. P.; King, J. F.; Hauschild, V. The Sources, Fate, and Toxicity of Chemical Warfare Agent Degradation Products. Environ. Health Perspect. 1999, 107(12), 933–974.
D’Agostino, P. A.; Provost, L. R.; Hancock, J. R. Analysis of Mustard Hydrolysis Products by Packed Capillary Liquid Chromatography-Electrospray Mass Spectrometry. J. Chromatogr. A 1998, 808, 177–184.
D’Agostino, P. A.; Provost, L. R. Capillary Column Isobutane Chemical Ionization Mass Spectrometry of Mustard and Related Compounds. Biomed. Environ. Mass Spectrom. 1988, 15(10), 553–564.
Gasson, E. J.; McCombie, H.; Williams, A. H.; Woodward, F. N. New Organic Sulfur Vesicants: Part IV. 1:2-Di-(2-Chloroethylthio)Ethane and Its Analogues. J. Chem. Soc. 1948, 44–46.
Robinson, J. P. Chemical Warfare. Sci. J. 1967, 4, 33–40.
Watson, A. P.; Griffin, G. D. Toxicity of Vesicant Agents Scheduled for Destruction by the Chemical Stockpile Disposal Program. Environ. Health Perspect. 1992, 98, 259–280.
Roberts, J. J.; Warwick, G. P. Studies of the Mode of Action of Alkylating Agents: VI. The Metabolism of Bis-2-chloroethylsulphide (Mustard Gas) and Related Compounds. Biochem. Pharmacol. 1963, 12, 1329–1334.
Davison, C.; Rozman, R. S.; Smith, P. K. Metabolism of Bis-β-chloroethyl Sulfide (Sulfur Mustard Gas). Biochem. Pharmacol. 1961, 7(1), 65–74.
Black, R. M.; Brewster, K.; Clarke, R. J.; Hambrook, J. L.; Harrison, J. M.; Howells, D. J. Biological Fate of Sulphur Mustard, 1,1′-Thiobis(2-chloroethane): Isolation and Identification of Urinary Metabolites Following Intraperitoneal Administration to Rat. Xenobiotica 1992, 22(4), 405–418.
Fidder, A.; Noort, D.; de Jong, L. P. A.; Benschop, H. P.; Hulst, A. G. N7-(2-Hydroxyethylthioethyl)-Guanine: A Novel Urinary Metabolite Following Exposure to Sulphur Mustard. Arch. Toxicol. 1996, 70(12), 854–855.
Sandelowsky, I.; Simon, G. A.; Bel, P.; Barak, R.; Vincze, A. N1-(2-Hydroxyethylthioethyl)-4-Methyl imidazole (4-met-1-imid-Thiodiglycol) in Plasma and Urine: A Novel Metabolite Following Dermal Exposure to Sulphur Mustard. Arch. Toxicol. 1992, 66(4), 296–297.
Bartelt-Hunt, S. L.; Barlaz, M. A.; Knappe, D. R. U.; Kjeldsen, P. Fate of Chemical Warfare Agents and Toxic Industrial Chemicals in Landfill. Environ. Sci. Technol. 2006, 40(13), 4219–4225.
Timperley, C. M.; Black, R. M.; Bird, M.; Holden, I.; Mundy, J. L.; Read, R. W. Hydrolysis and Oxidation Products of the Chemical Warfare Agents 1,2-Bis[(2-Chloroethyl)thio]Ethane Q and 2,2′-Bis(2-Chloroethylthio)Diethyl Ether T. Phosphorus, Sulfur, Silicon 2003, 178(9), 2027–2046.
D’Agostino, P. A.; Provost, L. R. Capillary Column Electron Impact and Ammonia Chemical Ionization Gas Chromatographic-Mass Spectrometric and Gas Chromatographic-Tandem Mass Spectrometric Analysis of Mustard Hydrolysis Products. J. Chromatogr. A 1993, 645(2), 283–292.
St. Quintin, T. D.; Leslie, D. R.; Collins, J. G. Hydrolysis of Sesqui-mustards. Aust. J. Chem. 2003, 56(4), 309–313.
D’Agostino, P. A.; Provost, L. R. Gas Chromatographic Retention Indices of Sulfur Vesicants and Related Compounds. J. Chromatogr. 1988, 436(3), 399–411.
D’Agostino, P. A.; Provost, L. R.; Hansen, A. S.; Luoma, G. A. Identification of Mustard Related Compounds in Aqueous Samples by Gas Chromatography/Mass Spectrometry. Biomed. Environ. Mass Spectrom. 1989, 18, 484–491.
Black, R. M.; Read, R. W. Improved Methodology for the Detection and Quantitation of Urinary Metabolites of Sulphur Mustard Using Gas Chromatography-Tandem Mass Spectrometry. J. Chromatogr. B 1995, 665(1), 97–105.
Black, R. M.; Read, R. W. Detection of Trace Levels of Thiodiglycol in Blood, Plasma, and Urine Using Gas Chromatography-Electron-Capture Negative-Ion Chemical Ionization Mass Spectrometry. J. Chromatgr. 1988, 449(1), 261–270.
Munavalli, S.; Jakubowski, E. M.; Durst, H. D. Liquid Chromatography Thermospray Mass Spectrometry of Mustard and Its Metabolites. J. Mass Spectrom. 1995, 30(12), 1716–1722.
Borrett, V. T.; Mathews, R. J.; Colton, R.; Traeger, J. C. Verification of the United Nations Chemical Weapons Convention: the Application of Electrospray Mass Spectrometry. Rapid Commun. Mass Spectrom. 1996, 10, 114–118.
D’Agostino, P. A.; Hancock, J. R.; Chenier, C. L. Packed Capillary Liquid Chromatography-Electrospray Ionization (Tandem) Mass Spectrometry of Mustard Hydrolysis Products in Soil. J. Chromatogr. A 2004, 1058(1/2), 97–105.
Smith, J. R.; Shih, M. L. Analysis of the Degradation Compounds of Chemical Warfare Agents using Liquid Chromatography/Mass Spectrometry. J. Appl. Toxicol. 2001, 21, S27-S34.
D’Agostino, P. A.; Hancock, J. R.; Chenier, C. L. Mass Spectrometric Analysis of Chemical Warfare Agents and their Degradation Products in Soil and Synthetic Samples. Eur. J Mass Spectrom. 2003, 9(6), 609–618.
Tak, V.; Kanaujia, P. K.; Pardasani, D.; Gupta, A. K.; Palit, M.; Srivastava, R. K.; Dubey, D. K. Electrospray Ionization Tandem Mass Spectral Analysis of Oxidation Products of Precursors of Sulfur Mustards. Rapid Commun. Mass Spectrom. 2006, 20(16), 2387–2394.
Black, R.; Read, R. W. Application of Liquid Chromatography-Atmospheric Pressure Chemical Ionization Mass Spectrometry and Tandem Mass Spectrometry, to the Analysis and Identification of Degradation Products of Chemical Warfare Agents. J. Chromatogr. A 1997, 759, 79–92.
Read, R. W.; Black, R. M. Rapid Screening Procedures for the Hydrolysis Products of Chemical Warfare Agents using Positive and Negative Ion Liquid Chromatography-Mass Spectrometry with Atmospheric Pressure Chemical Ionization. J. Chromatogr. A 1999, 862, 169–177.
Willoughby, R.; Sheehan, E.; Mitrovich, S. A Global View of LC/MS: How to Solve your Most Challenging Analytical Problems. Global View Publishing: Pittsburgh, 2002, p 65.
Horning, E. C.; Horning, M. G.; Carroll, D. I.; Dzidic, I.; Stillwell, R. N. New Picogram Detection System Based on a Mass Spectrometer with an External Ionization Source at Atmospheric Pressure. Anal. Chem. 1973, 45(6), 936–943.
Grimalt, S.; Pozo, O. J.; Marin, J. M.; Sancho, J. V.; Hernandez, F. Evaluation of Different Quantitative Approaches for the Determination of Non-easily Ionizable Molecules by Different Atmospheric Pressure Interfaces Used in Liquid Chromatography Tandem Mass Spectrometry: Abamectin as Case of Study. J. Am. Soc. Mass Spectrom. 2005, 16(10), 1619–1630.
Diehl, G.; Karst, U. On-line Electrochemistry—MS and Related Techniques. Anal. Bioanal. Chem. 2002, 373(6), 390–398.
Hunt, D. F.; McEwen, C. N.; Harvey, T. M. Positive and Negative Chemical Ionization Mass spectrometry Using a Townsend Discharge Ion Source. Anal. Chem. 1975, 47(11), 1730–1734.
Chapman, J. R. Practical Organic Mass Spectrometry; John Wiley and Sons, Inc.: New York, 1993, pp 115.
Budzikiewicz, H. Negative Chemical Ionization (NCI) of Organic-Compounds. Mass Spectrom. Rev. 1986, 5(4), 345–380.
Moruzzi, J. L.; Phelps, A. V. Survey of Negative-Ion-Molecule Reactions in O2, CO2, H2O, CO, and Mixtures of These Gases at High Pressures. J. Chem. Phys. 1966, 45(12), 4617–4627.
Horning, E. C.; Carroll, D. I.; Dzidic, I.; Haegele, K. D.; Horning, M. G.; Stillwell, R. N. Liquid Chromatograph-Mass Spectrometer-Computer Analytical Systems: A Continuous-Flow System Based on Atmospheric Pressure Ionization Mass Spectrometry. J. Chromatogr. 1974, 99, 13–21.
Dzidic, I.; Carroll, D. I.; Stillwell, R. N.; Horning, E. C. Atmospheric Pressure Ionization (API) Mass Spectrometry: Formation of Phenoxide Ions from Chlorinated Aromatic Compounds. Anal. Chem. 1975, 47(8), 1308–1312.
Dzidic, I.; Carroll, D. I.; Stillwell, R. N.; Horning, E. C. Gas Phase Reactions: Ionization by Proton Transfer to Superoxide Anions. J. Am. Chem. Soc. 1974, 96(16), 5258–5259.
Taylor, J. K. Quality Assurance of Chemical Measurements; CRC Press, Inc.: Boca Raton, FL, 1987; pp 79–83.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online April 29, 2007
Rights and permissions
About this article
Cite this article
Lemire, S.W., Ash, D.H., Johnson, R.C. et al. Mass spectral behavior of the hydrolysis products of sesqui- and oxy-mustard type chemical warfare agents in atmospheric pressure chemical ionization. J Am Soc Mass Spectrom 18, 1364–1374 (2007). https://doi.org/10.1016/j.jasms.2007.04.016
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2007.04.016