Abstract
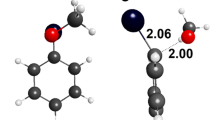
In the gas-phase reactions of halonitro- and dinitrophenide anions with X (X = F, Cl, Br, NO2) and NO2 groups in ortho or para position to each other with selected C-H acids: CH3CN, CH3COCH3, and CH3NO2, products of the SNAr-type reaction are formed. Nitrophenide anions are generated by decarboxylation of the respective nitrobenzenecarboxylate anions in ESI ion source and the SNAr reaction takes place either in the medium-pressure zone of the ion source or in the collision chamber of the triple quadrupole mass spectrometer. In the case of F, Cl, and NO2 derivatives, the main ionic product is the respective [NO2-Ph-CHR]− anion (R=CN, COCH3, NO2). In the case of Br derivatives, the main ionic product is Br− ion because it has lower proton affinity than the [NO2-Ph-CHR]− anion (for R=CN, COCH3). For some halonitrophenide anion C-H acid pairs of reactants, the SNAr reaction is competed by the formation of halophenolate anions. This reaction can be rationalized by the single electron-transfer mechanism or by homolytic C-H bond cleavage in the proton-bound complex, both resulting in the formation of the halonitrobenzene radical anion, which in turn undergoes -NO2 to -ONO rearrangement followed by the NO· elimination.
Article PDF
Similar content being viewed by others
References
Terrier, F. Nucleophilic Aromatic Displacement: The influence of the nitro group; VCH Publishers: New York, 1991.
Buncel, E.; Dust, J. M.; Terrier, F. Rationalizing the Regioselectivity in Polynitroarene Anionic σ-Adduct Formation: Relevance to Nucleophilic Aromatic Substitution. Chem. Rev. 1995, 95, 2262–2280.
Makosza, M. Electrophilic and Nucleophilic Aromatic Substitution: Analogous and Complementary Processes. Russ. Chem. Bull. 1996, 45, 491–504
Makosza, M. Nucleophilic Aromatic Substitution Revisited. Pol. J. Chem. 1992, 66, 3–23.
Bowie, J. H. Bimolecular Reactions of Nucleophiles in the Gas Phase. Acc. Chem. Res. 1980, 13, 76–82.
Squires, R. R. Gas-Phase Carbanion Chemistry. Acc. Chem. Res. 1992, 25, 461–467.
Gronert, S. Mass Spectrometric Studies of Organic Ion/Molecule Reactions. Chem. Rev. 2001, 101, 329–360.
DePuy, C. H. An Introduction to the Gas Phase Chemistry of Anions. Int. J. Mass Spectrom. 2000, 200, 79–96.
DePuy, C. H. Understanding Organic Gas-Phase Anion Molecule Reactions. J. Org. Chem. 2002, 67, 2393–2401.
Briscese, S. M. J.; Riveros, J. M. Gas Phase Nucleophilic Reactions of Aromatic Systems. J. Am. Chem. Soc. 1975, 97, 230–231.
Bowie, J. H.; Stapleton, B. J. Electron Impact Studies: CXIII Aromatic Nucleophilic Substitution in the Gas Phase. The Dinitrobenzenes. An Ion Cyclotron Resonance Study. Aust. J. Chem. 1977, 30, 795–800.
Giroldo, T.; Xavier, L. A.; Riveros, J. M. An Unusually Fast Nucleophilic Aromatic Displacement Reaction: The Gas-Phase Reaction of Fluoride Ions with Nitrobenzene. Angew. Chem. Int. Ed. 2004, 43, 3588–3590.
Meot-Ner, N.; Kafafi, S. A. Carbon Acidities of Aromatic Compounds. J. Am. Chem. Soc. 1988, 110, 6298–6303.
Dzidic, I.; Caroll, D. I.; Stillwell, R. N.; Horning, E. C. Atmospheric Pressure Ionization (API) Mass Spectrometry: Formation of Phenoxide Ions from Chlorinated Aromatic Compounds. Anal. Chem. 1975, 47, 1308–1312.
Sullivan, S. A.; Beauchamp, J. L. Reactions of Fluoroethylenes with Strong Bases in the Gas Phase. J. Am. Chem. Soc. 1977, 99, 5017–5022.
Ingemann, S.; Nibbering, N. M. M.; Sullivan, S. A.; DePuy, C. H. Nucleophilic Aromatic Substitution in a Gas Phase: The Importance of F− Ion-Molecule Complexes Formed in Gas-Phase Reactions between Nucleophiles and Some Alkyl Pentafluorophenyl Ethers. J. Am. Chem. Soc. 1982, 104, 6520–6527.
Ingemann, S.; Nibbering, N. M. M. Gas-Phase Reactions of Anions with 1-, 3-, and 4-Fluoroanisole. J. Org. Chem. 1982, 48, 183–191.
Ingemann, S.; Nibbering, N. M. M. Gas-Phase Reactions between Anions and Alkyl Pentafluorophenyl Ethers. Nouv. J. Chim. 1984, 8, 299–304.
Freriks, I. L.; de Koning, L. J.; Nibbering, N. M. M. Gas-Phase Ambident Reactivity of Acyclic Enolate Anions. J. Am. Chem. Soc. 1991, 113, 9119–9124.
Simkin, Y. B.; Gluz, E. B.; Glukhovtsev, M. N.; Minkin, V. I. Theoretical Study of Mechanisms of Aromatic Nucleophilic Substitution in the Gas Phase. J. Mol. Struct. (Theochem.) 1993, 284, 123–137.
Chen, H.; Chen, H.; Cooks, R. G. Meisenheimer Complexes Bonded at Carbon and at Oxygen. J. Am. Soc. Mass Spectrom. 2004, 15, 998–1004.
Song, Y.; Chen, H.; Cooks, R. G. Reactivity of Acetonyl Anion with Nitroaromatics: An Atmospheric Pressure Chemical Ionization Study. Rapid Commun. Mass Spectrom. 2005, 19, 3493–3499.
Song, Y.; Cooks, R. G. Atmospheric Pressure Ion/Molecule Reactions for the Selective Detection of Nitroaromatic explosives using acetonitrile and air as reagents. Rapid Commun. Mass Spectrom. 2006, 20, 3130–3138.
Bieńkowski, T.; Danikiewicz, W. Generation and Reactions of Substituted Phenide Anions in an Electrospray Triple Quadrupole Mass Spectrometer. Rapid Commun. Mass Spectrom. 2003, 17, 697–705.
Danikiewicz, W.; Bieńkowski, T.; Poddebniak, D. Generation and Reactions of Anionic σ-Adducts of 1,3-Dinitrobenzene and 1,3,5-Trinitrobenzene with Carbanions in a Gas Phase, Using an Electrospray Ion Source as the Chemical Reactor. J. Am. Soc. Mass Spectrom. 2004, 15, 927–933.
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Zakrzewski, V. G.; Montgomery, J. A., Jr.; Stratmann, R. E.; Burant, J. C.; Dapprich, S.; Millam, J. M.; Daniels, A. D.; Kudin, K. N.; Strain, M. C.; Farkas, O.; Tomasi, J.; Barone, V.; Cossi, M.; Cammi, R.; Mennucci, B.; Pomelli, C.; Adamo, C.; Clifford, S.; Ochterski, J. W.; Petersson, G. A.; Ayala, P. Y.; Cui, Q.; Morokuma, K.; Salvador, P.; Dannenberg, J. J.; Malick, D. K.; Rabuck, A. D.; Raghavachari, K.; Foresman, J. B.; Cioslowski, J.; Ortiz, J. V.; Baboul, A. G.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Gomperts, R.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham, M. A.; Peng, C. Y.; Nanayakkara, A.; Challacombe, M.; Gill, P. M. W.; Johnson, B.; Chen, W.; Wong, M. W.; Andres, J. L.; Gonzalez, C.; Head-Gordon, M.; Replogle, E. S.; Pople, J. A. Gaussian 98W, Revision A.11; Gaussian, Inc: Pittsburgh, PA, 2001.
NIST Chemistry WebBook, NIST Standard Reference Database Number 69, June 2005, Eds. Linstrom, P. J.; Mallard, W. G., Eds.; available at http://webbook.nist.gov/chemistry/.
Buncel, E.; Dust, J. M.; Manderville, R. A. Ambident Reactivity of Enolate Ions toward 1,3,5-Trinitrobenzene: The First Observation of an Oxygen-Bonded Enolate Meisenheimer Complex. J. Am. Chem. Soc. 1996, 118, 6072–6073.
Bowie, J. H. Electron Impact Studies—LX: Negative-Ion Mass Spectrometry of Functional Groups Skeletal Rearrangements in Aryl Nitro Compounds. Org. Mass Spectrom. 1971, 5, 945–951.
Bowie, J. H. Electron Impact: Studies—CXXI. Release of Kinetic Energy During Negative-Ion Fragmentations. The loss of NO· from Substituted Nitrobenzene Molecular Anions. Aust. J. Chem. 1978, 31, 573–577.
Ashby, E. C. Single-Electron Transfer, a Major Reaction Pathway in Organic Chemistry: An Answer to Recent Criticisms. Acc. Chem. Res. 1988, 21, 414–421.
Bowman, W. R. Reactivity of Substituted Aliphatic Nitro-Compounds with Nucleophiles. Chem. Soc. Rev. 1988, 17, 283–316.
Makosza, M.; Kwast, A. Direct Nucleophilic Addition versus a Single-Electron Transfer Pathway of σ H Adduct Formation in Vicarious Nucleophilic Substitution of Hydrogen. Eur. J. Org. Chem. 2004, 2125–2130.
Chiavarino, B.; Crestoni, M. A.; Fornarini, S.; Lanucara, F.; Lemaire, J.; Maître, P. Meisenheimer Complexes Positively Characterized as Stable Intermediates in the Gas Phase. Angew. Chem. Int. Ed. 2007, 45, 1995–1998.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online April 25, 2007
Rights and permissions
About this article
Cite this article
Danikiewicz, W., Bieńkowski, T., Kozłowska, D. et al. Aromatic nucleophilic substitution (SNAr) Reactions of 1,2- and 1,4-halonitrobenzenes and 1,4-dinitrobenzene with carbanions in the gas phase. J Am Soc Mass Spectrom 18, 1351–1363 (2007). https://doi.org/10.1016/j.jasms.2007.04.005
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2007.04.005