Abstract
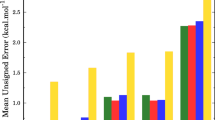
This work describes the first experimental studies of deuterium kinetic isotope effects (KIEs) for the gas-phase E2 reactions of microsolvated systems. The reactions of F−(H2O)n and OH−(H2O)n, where n = 0, 1, with (CH3)3CX (X = Cl, Br), as well as the deuterated analogs of the ionic and neutral reactants, were studied utilizing the flowing afterglow-selected ion flow tube technique. The E2 reactivity is found to decrease with solvation. Small, normal kinetic isotope effects are observed for the deuteration of the alkyl halide, while moderately inverse kinetic isotope effects are observed for the deuteration of the solvent. Minimal clustering of the product ions is observed, but there are intriguing differences in the nature and extent of the clustering process. Electronic structure calculations of the transition states provide qualitative insight into these microsolvated E2 reactions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wladkowski, B. D.; Brauman, J. I. Substitution Versus Elimination in Gas-Phase Ionic Reactions. J. Am. Chem. Soc. 1992, 114, 10643–10644.
Villano, S. M.; Kato, S.; Bierbaum, V. M. Deuterium Kinetic Isotope Effects in Gas-Phase Sn2 and E2 Reactions: Comparison of Experiment and Theory. J. Am. Chem. Soc. 2006, 128, 736–737.
Hu, W.-P.; Truhlar, D. G. Factors Affecting Competitive Ion-Molecule Reactions: ClO− + C2H5Cl and C2D5Cl via E2 and Sn2 Channels. J. Am. Chem. Soc. 1996, 118, 860–869.
de Koning, L. J.; Nibbering, N. M. M. On the Mechanism of Base-Induced Gas-Phase Elimination Reactions of Ethers. J. Am. Chem. Soc. 1987, 109, 1715–1722.
Gronert, S. Mass Spectrometric Studies of Organic Ion/Molecule Reactions. Chem. Rev. 2001, 101, 329–360.
Munsch, T. E.; Wenthold, P. G. Organic Gas-Phase Ion Chemistry. Annu. Rep. Prog. Chem B 2003, 99, 420–446.
Jones, M. E.; Ellison, G. B. Gas-Phase E2 Reactions: Methoxide Ion and Bromopropane. J. Am. Chem. Soc. 1989, 111, 1645–1654.
Gronert, S. Gas Phase Studies of the Competition Between Substitution and Elimination Reactions. Acc. Chem. Res. 2003, 36, 848–857.
Gronert, S.; DePuy, C. H.; Bierbaum, V. M. Deuterium Isotope Effects in Gas-Phase Reactions of Alkyl Halides: Distinguishing E2 and Sn2 Pathways. J. Am. Chem. Soc. 1991, 113, 4009–4010.
Hu, W.-P.; Truhlar, D. G. Modeling Transition State Solvation at the Single-Molecule Level: Test of Correlated ab Initio Predictions against Experiment for the Gas-Phase Sn2 Reaction of Microhydrated Fluoride with Methyl Chloride. J. Am. Chem. Soc. 1994, 116, 7797–7800.
Tachikawa, H. Collision Energy Dependence on the Microsolvated Sn2 Reaction of F−(H2O) with CH3Cl: A Full Dimensional Ab Initio Direct Dynamics Study. J. Phys. Chem. A 2001, 105, 1260–1266.
Ohta, K.; Morokuma, K. An MO study of Sn2 Reactions in Hydrated Gas Clusters: Hydrated Hydroxide [(H2O)nOH−] + Hydrated Methyl Chloride [MeCl(H2O)m] → methanol + chloride + (n + m)water. J. Phys. Chem. 1985, 89, 5845–5849.
Seeley, J. V.; Morris, R. A.; Viggiano, A. A. Temperature Dependencies of the Rate Constants and Branching Ratios for the Reactions of F−(H2O)0-5 with CH3Br. J. Phys. Chem. A. 1997, 101, 4598–4601.
Takashima, K.; Riveros, J. M. Gas-Phase Solvated Negative Ions. Mass Spectrom. Rev. 1999, 17, 409–430.
Tucker, S. C.; Truhlar, D. G. Effect of Nonequilibrium Solvation on Chemical Reaction Rates: Variational Transition-State-Theory Studies of the Microsolvated Reaction Cl−(H2O)n + CH3Cl. J. Am. Chem. Soc. 1990, 112, 3347–3361.
Viggiano, A. A.; Arnold, S. T.; Morris, R. A. Reactions of Mass-Selected Cluster Ions in a Thermal Bath Gas. Int. Rev. Phys. Chem. 1998, 17, 147–184.
ÓHair, R. A. J.; Davico, G. E.; Hacaloglu, J.; Dang, T. T.; DePuy, C. H.; Bierbaum, V. M. Measurements of Solvent and Secondary Kinetic Isotope Effects for the Gas-Phase Sn2 Reactions of Fluoride with Methyl Halides. J. Am. Chem. Soc. 1994, 116, 3609–3610.
Kato, S.; Hacaloglu, J.; Davico, G. E.; DePuy, C. H.; Bierbaum, V. M. Deuterium Kinetic Isotope Effects in the Gas-Phase Sn2 Reactions of Solvated Fluoride Ions with Methyl Halides. J. Phys. Chem. A 2004, 108, 9887–9891.
Bickelhaupt, F. M.; Baerends, E. J.; Nibbering, N. M. M. The Effect of Microsolvation on E2 and Sn2 Reactions: Theoretical Study of the Model System F− + C2H5F + nHF. Chem. Eur. J. 1996, 2, 196–207.
Wu, Y.-R.; Hu, W.-P. Reaction Dynamics Study on the Tunneling Effects of a Microsolvated E2 Reaction: FO−(H2O) + C2H5Cl → HOF(H2O) + C2H4 + Cl−. J. Am. Chem. Soc. 1999, 121, 10168–10177.
DePuy, C. H.; Gronert, S.; Mullin, A.; Bierbaum, V. M. Gas-Phase Sn2 and E2 Reactions of Alkyl Halides. J. Am. Chem. Soc. 1990, 112, 8650–8655.
Van Doren, J. M.; Barlow, S. E.; DePuy, C. H.; Bierbaum, V. M. The Tandem Flowing Afterglow-SIFT-Drift. Int. J. Mass Spectrom. Ion Processes 1987, 81, 85–100.
Bickelhaupt, F. M.; de Koning, L. J.; Nibbering, N. M. M. Anionic Ether Cleavage of Tetrahydrofuran in the Gas Phase. Tetrahedron 1993, 49, 2077–2092.
Gaussian 03, Rev B.05, Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Montgomery, J. J. A.; Vreven, T.; Kudin, K. N.; Burant, J. C.; Millam, J. M.; Iyengar, S. S.; Tomasi, J.; Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G. A.; Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Klene, M.; Li, X.; Knox, J. E.; Hratchian, H. P.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W. A. P. Y.; Morokuma, K.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Zakrzewski, V. G.; Dapprich, S.; Daniels, A. D.; Strain, M. C.; Farkas, O.; Malick, D. K.; Rabuck, A. D.; Raghavachari, K.; Foresman, J. B.; Ortiz, J. V.; Cui, Q.; Baboul, A. G.; Clifford, S.; Cioslowski, J.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham, M. A.; Peng, C. Y.; Nanayakkara, A.; Challacombe, M.; Gill, P. M. W.; Johnson, B.; Chen, W.; Wong, M. W.; Gonzalez, C.; Pople, J. A. Gaussian, Inc: Pittsburgh, PA, 2004.
Gaussian 98, Rev A.7, Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Montgomery, J. J. A.; Vreven, T.; Kudin, K. N.; Burant, J. C.; Millam, J. M.; Iyengar, S. S.; Tomasi, J.; Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G. A.; Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Klene, M.; Li, X.; Knox, J. E.; Hratchian, H. P.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W. A. P. Y.; Morokuma, K.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Zakrzewski, V. G.; Dapprich, S.; Daniels, A. D.; Strain, M. C.; Farkas, O.; Malick, D. K.; Rabuck, A. D.; Raghavachari, K.; Foresman, J. B.; Ortiz, J. V.; Cui, Q.; Baboul, A. G.; Clifford, S.; Cioslowski, J.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham, M. A.; Peng, C. Y.; Nanayakkara, A.; Challacombe, M.; Gill, P. M. W.; Johnson, B.; Chen, W.; Wong, M. W.; Gonzalez, C.; Pople, J. A. Gaussian, Inc: Pittsburgh, PA, 1998.
Glad, S. S.; Jensen, F. Basis Set and Correlation Effects on Transition State Geometries and Kinetic Isotope Effects. J. Phys. Chem. 1996, 100, 16892–16898.
Gronert, S.; Keeffe, J. R. Primary Semiclassical Kinetic Hydrogen Isotope Effects in Identity Carbon-to-Carbon Proton- and Hydride-Transfer Reactions, an ab Initio and DFT Computational Study. J. Org. Chem. 2006, 71, 5959–5968.
Hiraoka, K.; Fujita, K.; Ishida, M.; Ichikawa, T.; Okada, H.; Hiizumi, K.; Wada, A.; Takao, K. Gas-Phase Ion/Molecule Reactions in C5F8. J. Phys. Chem. A 2005, 109, 1049–1056.
McAllister, M. A. Characterization of Low-Barrier Hydrogen Bonds. 2: HF −2 : A density functional and ab Initio study. THEOCHEM 1998, 427, 39–53.
Su, T.; Chesnavich, W. J. Parametrization of the Ion-Polar Molecule Collision Rate Constant by Trajectory Calculations. J. Chem. Phys. 1982, 76, 5183–5185.
Miller, K. J.; Savchik, J. A New Empirical Method to Calculate Average Molecular Polarizabilities. J. Am. Chem. Soc. 1979, 101, 7206–7213.
CRC Handbook of Chemistry and Physics, 75th ed.; Lide, D. R., Ed.; CRC Press: Boca Raton, FL, 1994, pp. 9–49 10–200.
Chase, W. M., Jr.; Davies, C. A.; Downey, J. J. R.; Frurip, D. J.; McDonald, R. A.; Syverud, A. N. JANAF Thermochemical Tables, 3rd ed. Parts I and II. J. Phys. Chem. Ref. Data 1985, Suppl 1.
Linstrom, P. J., Mallard, W. G. NIST Chemistry WebBook, NIST Standard Reference Database Number 69, June 2005, National Institute of Standards and Technology: Gaithersburg, MD, 20899 (http://webbook.nist.gov).
Bohme, D. K.; Raksit, A. B. Gas-Phase Measurements of the Influence of Stepwise Solvation on the Kinetics of Sn2 Reactions of Solvated Fluoride Ion with Methyl Chloride and Methyl Bromide and of Solvated Chloride Ion with Methyl Bromide. Can. J. Chem. 1985, 63, 3007.
Westheimer, F. H. The Magnitude of the Primary Kinetic Isotope Effect for Compounds of Hydrogen and Deuterium. Chem. Rev. 1961, 61, 265–273.
Sun, L.; Song, K.; Hase, W. L. An Sn2 Reaction That Avoids Its Deep Potential Energy Minimum. Science. 2002, 296, 875–878.
Baer, T.; Hase, W. L. Unimolecular Reaction Dynamics: Theory and Experiment Oxford University Press: New York, 1996, pp. 1–448.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online April 19, 2007
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Eyet, N., Villano, S.M., Kato, S. et al. Deuterium kinetic isotope effects in microsolvated gas-phase E2 reactions. J Am Soc Mass Spectrom 18, 1046–1051 (2007). https://doi.org/10.1016/j.jasms.2007.02.016
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2007.02.016