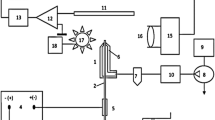
Abstract
Electrospray ionization mass spectrometry was used to investigate complex formation of different metal complexes in a continuous-flow ligand-exchange reactor. A computer program was developed based on normal equilibrium calculations to predict the formation of various metal-ligand complexes. Corresponding to these calculations, infusion electrospray mass spectrometric experiments were performed to investigate the actual complex formation in solution. The data clearly show good correlation between the theoretically calculated formation of metal-ligand complexes and the experimental mass spectrometric data. Moreover, the approach demonstrates that the influence of the pH can be investigated using a similar approach. Indirectly, these infusion experiments provide information on relative binding constants of different ligands towards a metal-ion. To demonstrate this, a continuous-flow ligand-exchange detection system with mass spectrometric detection was developed. Injection of ligands, with different affinity for the metal-ion, into the reactor shows good correlation between binding constants and the response in the ligand-exchange detection system. Additional information on the introduced ligand, and the complexes formed after introduction of the ligand, can be obtained from interpretation of the mass spectra.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Farrell, N. Biomedical Uses and Applications of Inorganic Chemistry: An Overview. Coord. Chem. Rev. 2002, 232, 1–4.
Molenveld, P.; Engbersen, J. F. J.; Kooijman, H.; Spek, A. L.; Reinhoudt, D. N. Efficient Catalytic Phosphate Diester Cleavage by the Synergetic Action of Two Cu(II) Centers in a Dinuclear Cis-Diaqua Cu(II) Calix[4]Arene Enzyme Model. J. Am. Chem. Soc. 1998, 120, 6726–6737.
Chen, P. Electrospray Ionization Tandem Mass Spectrometry in High-Throughput Screening of Homogeneous Catalysts. Angew. Chem. Int. Ed. 2003, 42, 2832–2847.
Chaparro, A. L.; Vachet, R. W. Tandem Mass Spectrometry of Cu(II) Complexes: The Effects of Ligand Donor Group on Dissociation. J. Mass Spectrom. 2003, 38, 333–342.
Traeger, J. C. Electrospray Mass Spectrometry of Organometallic Compounds. Int. J. Mass Spectrom. 2000, 200, 387–401.
Krabbe, J. G.; Lingeman, H.; Niessen, W. M. A.; Irth, H. Ligand-Exchange Detection of Phosphorylated Peptides Using Liquid Chromatography Electrospray Mass Spectrometry. Anal. Chem. 2003, 75, 6853–6860.
Krabbe, J. G.; Lingeman, H.; Niessen, W. M. A.; Irth, H. Screening for Metal Ligands by Liquid Chromatography-Ligand-Exchange-Electrospray Mass Spectrometry. J. Chromatogr. A. 2005, 1093, 36–46.
Satterfield, M.; Brodbelt, J. S. Relative Binding Energies of Gas-Phase Pyridyl Ligand/Metal-Complexes by Energy-Variable Collisionally Activated Dissociation in a Quadrupole Ion Trap. Inorg. Chem. 2001, 40, 5393–5400.
Tsierkezos, N. G.; Schroder, D.; Schwarz, H. Complexation of Nickel(II) by Ethylenediamine Investigated by Means of Electrospray Ionization Mass Spectrometry. Int. J. Mass Spectrom. 2004, 235, 33–42.
Vincenti, M. Host-Guest Chemistry in the Mass Spectrometer. J. Mass Spectrom. 1995, 30, 925–939.
Jellen, E. E.; Chappell, A. M.; Ryzhov, V. Effects of Size of Noncovalent Complexes on Their Stability During Collision-Induced Dissociation. Rapid Commun. Mass Spectrom. 2002, 16, 1799–1804.
Kumar, M. R.; Prabhakar, S.; Kumar, M. K.; Reddy, T. J.; Vairamani, M. Dissociation of Gas-Phase Dimeric Complexes of Lactic Acid and Transition Metal-Ions Formed Under Electrospray Ionization Conditions; The Role of Reduction of the Metal-Ion. Rapid Commun. Mass Spectrom. 2005, 19, 113–120.
Blair, S. M.; Brodbelt, J. S.; Marchand, A. P.; Kumar, K. A.; Chong, H. S. Evaluation of Binding Selectivities of Caged Crown Ligands Toward Heavy Metals by Electrospray Ionization/Quadrupole Ion Trap Mass Spectrometry. Anal. Chem. 2000, 72, 2433–2445.
Young, D. S.; Hung, H. Y.; Liu, L. K. An Easy and Rapid Method for Determination of Stability Constants by Electrospray Ionization Mass Spectrometry. Rapid Commun. Mass Spectrom. 1997, 11, 769–773.
Di Marco, V. B.; Bombi, G. G. Electrospray Mass Spectrometry (ESI-MS) in the Study of Metal-Ligand Solution Equilibria. Mass Spectrom. Rev. 2006, 25, 347–379.
Young, D. S.; Hung, H. Y.; Liu, L. K. Estimation of Selectivities and Relative Cationization Efficiencies of Different [Crown plus M](+) by Electrospray Mass Spectrometry. J. Mass Spectrom. 1997, 32, 432–437.
Goolsby, B. J.; Brodbelt, J. S.; Adou, E.; Blanda, M. Determination of Alkali Metal-Ion Binding Selectivities of Calixarenes by Matrix-Assisted Laser Desorption Ionization and Electrospray Ionization in a Quadrupole Ion Trap. Int. J. Mass Spectrom. 1999, 193, 197–204.
Wang, K. S.; Gokel, G. W. Correlation of Solution and Gas Phase Complexation Assessed by Electrospray Ionization Mass Spectrometry: Application to One-, Two-, and Three-ring Macrocycles. J. Org. Chem. 1996, 61, 4693–4697.
Gomez-Ariza, J. L.; Garcia-Barrera, T.; Lorenzo, F.; Bernal, V.; Villegas, M. J.; Oliveira, V. Use of Mass Spectrometry Techniques for the Characterization of Metal Bound to Proteins (Metallomics) in Biological Systems. Anal. Chim. Acta. 2004, 524, 15–22.
Press, W. H.; Flannery, B. P.; Teulkolsky, S. A.; Vetterling, W. T. Numerical Recipes in Pascal; 2nd ed.; Cambridge University Press: Cambridge, 1992; p 285.
Bure, C.; Lange, C. Comparison of Dissociation of Ions in an Electrospray Source, or a Collision Cell in Tandem Mass Spectrometry. Curr. Org. Chem. 2003, 7, 1613–1624.
Kubwabo, C.; Vais, N.; Benoit, F. M. Characterization of Microcystins Using In-Source Collision-Induced Dissociation. Rapid Commun. Mass Spectrom. 2005, 19, 597–604.
Vachet, R. W.; Callahan, J. H. Quadrupole Ion Trap Studies of the Structure and Reactivity of Transition Metal-Ion Pair Complexes. J. Mass Spectrom. 2000, 35, 311–320.
Shen, J.; Brodbelt, J. Formation of Doubly Charged Transition Metal-Polyether-Pyridyl Mixed-Ligand Complexes by Electrospray Ionization. J. Mass Spectrom. 1999, 34, 137–146.
Shvartsburg, A. A.; Wilkes, J. G.; Lay, J. O.; Siu, K. W. M. Fragmentation and Charge Transfer in Gas-Phase Complexes of Divalent Metal-Ions with Acetonitrile. Chem. Phys. Lett. 2001, 350, 216–224.
Seto, C.; Stone, J. A. The Reactions of Cu2 + (CH3CN)(n) (n = 2–4) and Cu2 + (CH3CN)(3)(H2O) at low collision energy with neutral molecules in a triple sector quadrupole instrument. Int. J. Mass Spectrom. 1998, 175, 263–276.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online January 18, 2007
Rights and permissions
About this article
Cite this article
Krabbe, J.G., de Boer, A.R., van der Zwan, G. et al. Metal-complex formation in continuous-flow ligand-exchange reactors studied by electrospray mass spectrometry. J Am Soc Mass Spectrom 18, 707–713 (2007). https://doi.org/10.1016/j.jasms.2006.12.006
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2006.12.006