Abstract
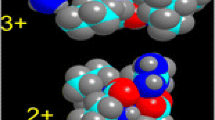
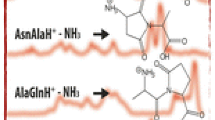
The sodium ion affinities (binding energies) of nineteen peptides containing 2–4 residues have been determined by experimental and computational approaches. Na+-bound heterodimers with amino acid and peptide ligands (Pep1, Pep2) were produced by electrospray ionization. The dissociations of these Pep1-Na+-Pep2 ions to Pep1-Na+ and Pep2-Na+ were examined by collisionally activated dissociation to construct a ladder of relative affinities via the kinetic method. The accuracy of this ladder was subsequently ascertained by experiments using several excitation energies for four peptide pairs. The relative scale was converted to absolute affinities by anchoring the relative values to the known Na+ affinity of GlyGly. The Na+ affinities of AlaAla, HisGly, GlyHis, GlyGlyGly, AlaAlaAla, GlyGlyGlyGly, and AlaAlaAlaAla were also calculated at the MP2(full)/6-311+G(2d,2p) level of ab initio theory using geometries that were optimized at the MP2(full)/6-31G(d) level for AlaAla or HF/6-31G(d) level for the other peptides; the resulting values agree well with experimental Na+ affinities. Increasing the peptide size is found to dramatically augment the Na+ binding energy. The calculations show that in nearly all cases, all available carbonyl oxygens are sodium binding sites in the most stable structures. Whenever side chains are available, as in HisGly and GlyHis, specific additional binding sites are provided to the cation. Oligoglycines and oligoalanines have similar binding modes for the di- and tripeptides, but differ significantly for the tetrapeptides: while the lowest energy structure of GlyGlyGlyGly-Na+ has the peptide folded around the ion with all four carbonyl oxygens in close contact with Na+, that of AlaAlaAlaAla_Na+ involves a pseudo-cyclic peptide in which the C and N termini interact via hydrogen bonding, while Na+ sits on top of the oxygens of three nearly parallel C=O bonds.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Grossman, G. P. Thirst and Sodium Appetite—Physiological Basis; Academic Press, Inc.: San Diego, 1990; pp 177–202.
Michell, A. R., Ed.; The Clinical Biology of Sodium: The Physiology and Pathophysiology of Sodium in Mammals; Pergamon: Oxford, UK, 1995; p 16.
Skou, J. C.; Norby, J. G.; Maunsbach, A. B.; Esmann, M. Progress in Clinical and Biological Research, 268A; Alan R. Liss, Inc.: New York, 1987; pp 401–632.
Aidley, D. J.; Stanfield, P. R. Ion Channels: Molecules in Action; Cambridge University Press: Cambridge, 1994; pp 1–58.
Bojesen, J. S.; Breindahl, T.; Andersen, U. N. On the Sodium and Lithium Ion Affinities of Some α-Amino Acids. Org. Mass Spectrom. 1993, 28, 1448–1452.
Klassen, J. S.; Anderson, S. G.; Blades, A. T.; Kebarle, P. Reaction Enthalpies for M+L=M++L, where M+=Na+ and K+ and L=Acetamide, N-Methylacetamide, N,N-Dimethylacetamide, Glycine, and Glycylglycine, from Determinations of the Collision-Induced Dissociation Thresholds. J. Phys. Chem. 1996, 100, 14218–14227.
Jockusch, R. A.; Price, W. D.; Williams, E. R. Structure of cationized arginine (Arg·M+, M=H, Li, Na, K, Rb, and Cs) in the Gas Phase: Further Evidence for Zwitterionic Arginine. J. Phys. Chem. A 1999, 103, 9266–9274.
Dunbar, R. C. Complexation of Na+ and K+ to Aromatic Amino Acids: A Density Functional Computational Study of Cation-π Interactions. J. Phys. Chem. A 2000, 104, 8067–8074.
Ryzhov, V.; Dunbar, R. C.; Cerda, B. A.; Wesdemiotis, C. Cation-π Effects in the Complexation of Na+ and K+ with Phe, Tyr, and Trp in the Gas Phase. J. Am. Soc. Mass Spectrom. 2000, 11, 1037–1046.
Cerda, B. A.; Wesdemiotis, C. Zwitterionic Versus Charge-Solvated Structures in the Binding of Arginine to Alkali Metal Ions in the Gas Phase. Analyst 2000, 125, 657–660.
Talley, J. M.; Cerda, B. A.; Ohanessian, G.; Wesdemiotis, C. Alkali Metal Ion Binding to Amino Acids Versus Their Methyl Esters: Affinity Trends and Structural Changes in the Gas Phase. Chem. Eur. J. 2002, 8, 1377–1388.
Kish, M. M.; Ohanessian, G.; Wesdemiotis, C. The Na+ Affinities of α-Amino Acids: Side-Chain Substituent Effects. Int. J. Mass Spec. 2003, 227, 509–524.
Gapeev, A.; Dunbar, R. C. Na+ Affinities of Gas-Phase Amino Acids by Ligand Exchange Equilibrium. Int. J. Mass Spectrom. 2003, 228, 825–839.
Moision, R. M.; Armentrout, P. B. Experimental and Theoretical Dissection of Sodium Cation/Glycine Interactions. J. Phys. Chem. A 2002, 106, 10350–10362.
Hoyau, S.; Ohanessian, G. Interaction of Alkali Metal Cations (Li+-Cs+) with Glycine in the Gas Phase: A Theoretical Study. Chem. Eur. J. 1998, 4, 1561–1569.
Hoyau, S.; Norrman, K.; McMahon, T. B.; Ohanessian, G. A Quantitative Basis for a Scale of Na+ Affinities of Organic and Small Biological Molecules in the Gas Phase. J. Am. Chem. Soc. 1999, 121, 8864–8875.
Cerda, B. A.; Hoyau, S.; Ohanessian, G.; Wesdemiotis, C. Na+ Binding to Cyclic and Linear Dipeptides: Bond Energies, Entropies of Na+ Complexation, and Attachment Sites from the Dissociation of Na+-Bound Heterodimers and Ab Initio Calculations. J. Am. Chem. Soc. 1998, 120, 2437–2448.
Cooks, R. G.; Wong, P. S. H. Kinetic Method of Making Thermochemical Determinations: Advances and Applications. Acc. Chem. Res. 1998, 31, 379–386.
Cooks, R. G.; Koskinen, J. T.; Thomas, P. D. The Kinetic Method of Making Thermochemical Determinations. J. Mass Spectrom. 1999, 34, 85–92.
Feng, W. Y.; Gronert, S.; Lebrilla, C. B. Lithium and Sodium Ion Binding Energies of N-Acetyl and N-Glycyl Amino Acids. J. Am. Chem. Soc. 1999, 121, 1365–1371.
Cooks, R. G.; Patrick, J. S.; Kotiaho, T.; McLuckey, S. A. Thermochemical Determinations by the Kinetic Method. Mass Spectrom. Rev. 1994, 13, 287–339.
Kish, M. M.; Wesdemiotis, C.; Ohanessian, G. The Sodium Ion Affinity of Glycylglycine. J. Phys. Chem. B 2004, 108, 3086–3091.
Feller, D. A Complete Basis Set Estimate of Cation-π Bond Strengths: Na+(Ethylene) and Na+(Benzene). Chem. Phys. Lett. 2000, 322, 543–548.
Feller, D.; Dixon, D. A.; Nicholas, J. B. Binding Enthalpies for Alkali Cation-Benzene Complexes Revisited. J. Phys. Chem. A 2000, 104, 11414–11419.
Cao, J.; Aubry, C.; Holmes, J. L. Proton Affinities of Simple Amines; Entropies and Enthalpies of Activation and Their Effect on the Kinetic Method for Evaluating Proton Affinities. J. Phys. Chem. A 2000, 104, 10045–10052.
Hahn, I.-S.; Wesdemiotis, C. Protonation Thermochemistry of β-Alanine: An Evaluation of Proton Affinities and Entropies Determined by the Extended Kinetic Method. Int. J. Mass Spectrom. 2003, 222, 465–479.
Bouchoux, G.; Djazi, F.; Gaillard, F.; Vierezet, D. Application of the Kinetic Method to Bifunctional Bases; MIKE and CID-MIKE Test Cases. Int. J. Mass Spectrom. 2003, 227, 479–496.
Bouchoux, G.; Buisson, D.-A.; Bourcier, S.; Sablier, M. Application of the Kinetic Method to Bifunctional Bases; ESI Tandem Quadrupole Experiments. Int. J. Mass Spectrom. 2003, 228, 1035–1054.
Laidler, K. J. Chemical Kinetics, 3rd ed.; Harper and Row Publishers: New York, 1987, p 112.
Armentrout, P. B.; Rodgers, M. T. An Absolute Sodium Cation Affinity Scale: Threshold Collision-Induced Dissociation Experiments and ab Initio Theory. J. Phys. Chem. A 2000, 104, 2238–2247.
Cerda, B. A.; Wesdemiotis, C. Li+, Na+, and K+ Binding to the DNA and RNA Nucleobases: Bond Energies and Attachment Sites from the Dissociation of Metal Ion-Bound Heterodimers. J. Am. Chem. Soc. 1996, 118, 11884–11892.
Cerda, B. A.; Wesdemiotis, C. Thermochemistry and Structures of Na+ Coordinated mono- and Disaccharide Stereoisomers. Int. J. Mass Spectrom. 1999, 189, 189–204.
Ervin, K. M. Microcanonical Analysis of the Kinetic Method: The Meaning of the “Apparent Entropy”. J. Am. Soc. Mass Spectrom. 2002, 13, 435–452.
Wesdemiotis, C. Entropy Considerations in Kinetic Method Experiments. J. Mass Spectrom. 2004, 39, 998–1003.
Drahos, L.; Vékey, K. Entropy Evaluation Using the Kinetic Method: Is It Feasible?. J. Mass Spectrom. 2003, 38, 1025–1042.
Armentrout, P. B. Entropy Measurements and the Kinetic Method: A Statistically Meaningful Approach. J. Am. Soc. Mass Spectrom. 2000, 11, 371–379.
McMahon, T. B.; Ohanessian, G. An Experimental and Ab Initio Study of the Nature of the Binding in Gas-Phase Complexes of Sodium Ions. Chem. Eur. J. 2000, 6, 2931–2941.
Cornell, W. D.; Cieplak, P.; Bayly, C. I.; Gould, I. R.; Merz, K. M.; Ferguson, D. M.; Spellmeyer, D. C.; Fox, T.; Caldwell, J. W.; Kollman, P. A. A Second Generation Force Field for the Simulation of Proteins, Nucleic Acids, and Organic Molecules. J. Am. Chem. Soc. 1995, 117, 5179–5197.
HyperChem 6.0.; Hypercube: Gainesville, FL, 2000.
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Montgomery, J. A., Jr.; Vreven, T.; Kudin, K. N.; Burant, J. C.; Millam, J. M.; Iyengar, S. S.; Tomasi, J.; Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G. A.; Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Klene, M.; Li, X.; Knox, J. E.; Hratchian, H. P.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W.; Ayala, P. Y.; Morokuma, K.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Zakrzewski, V. G.; Dapprich, S.; Daniels, A. D.; Strain, M. C.; Farkas, O.; Malick, D. K.; Rabuck, A. D.; Raghavachari, K.; Foresman, J. B.; Ortiz, J. V.; Cui, Q.; Baboul, A. G.; Clifford, S.; Cioslowski, J.; Stefanov, B. B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham, M. A.; Peng, C. Y.; Nanayakkara, A.; Challacombe, M.; Gill, P. M. W.; Johnson, B.; Chen, W.; Wong, M. W.; Gonzalez, C.; Pople, J. A. Gaussian 03, Revision C. 02.; Gaussian, Inc.: Wallingford, CT, 2004.
Benzakour, M.; Mcharfi, M.; Cartier, A.; Daoudi, A. Interactions of Peptides with Metallic Cations. I. Complexes Glycylglycine-M+ (M=Li, Na). J. Mol. Struct. Theochem. 2004, 710, 169–174.
Wong, C. H. S.; Ma, N. L.; Tsang, C. W. A Theoretical Study of Potassium Cation Binding to Glycylglycine (GG) and Alanylalanine (AA) Dipeptides. Chem. Eur. J. 2002, 8, 4909–4918.
Wyttenbach, T.; Bushnell, J. E.; Bowers, M. T. Salt Bridge Structures in the Absence of Solvent?: The Case for the Oligoglycines. J. Am. Chem. Soc. 1998, 120, 5098–5103.
Wyttenbach, T.; Witt, M.; Bowers, M. T. On the Stability of Amino Acid Zwitterions in the Gas Phase: The Influence of Derivatization, Proton Affinity, and Alkali Ion Addition. J. Am. Chem. Soc. 2000, 122, 3458–3464.
Strittmatter, E. F.; Williams, E. R. Computational Approach to the Proton Affinities of Glyn (n=1–10). Int. J. Mass Spectrom. 1999, 185/187, 935–948.
Shoeib, T.; Rodriquez, C. F.; Siu, K. W. M.; Hopkinson, A. C. A Comparison of Copper(I) and Silver(I) Complexes of Glycine, Diglycine, and Triglycine. Phys. Chem., Chem. Phys. 2001, 3, 853–861.
Kapota, C.; Ohanessian, G. The Low Energy Tautomers and Conformers of the Dipeptides HisGly and GlyHis and of Their Sodium Ion Complexes in the Gas Phase. Phys. Chem. Chem. Phys. 2005, 7, 3744–3755.
Cerda, B. A.; Wesdemiotis, C. The Relative Copper(I) Ion Affinities of Amino Acids in the Gas Phase. J. Am. Chem. Soc. 1995, 117, 9734–9739.
Lee, V. W.-M.; Li, H.; Lau, T.-C.; Guevremont, R.; Siu, K. W. M. Relative Silver(I) Ion Binding Energies of α-Amino Acids: A Determination by Means of the Kinetic Method. J. Am. Soc. Mass Spectrom. 1998, 9, 760–766.
Cheng, X.; Wu, Z.; Fenselau, C. Collision Energy Dependence of Proton-Bound Dimer Dissociation: Entropy Effects, Proton Affinities, and Intramolecular Hydrogen-Bonding in Protonated Peptides. J. Am. Chem. Soc. 1993, 115, 4844–4848.
Wang, Z.; Chu, I. K.; Rodriquez, C. F.; Hopkinson, A. C.; Siu, M. K. W. α,ω-Diaminoalkanes as Models for Bases that Dicoordinate the Proton: An Evaluation of the Kinetic Method for Estimating Proton Affinities. J. Phys. Chem. A 1999, 103, 8700–8705.
Schroeder, O. E.; Andriole, E. J.; Carver, K. L.; Colyer, K. E.; Poutsma, J. C. Proton Affinity of Lysine Homologues from the Extended Kinetic Method. J. Phys. Chem. A 2004, 108, 326–332.
Wind, J. J.; Papp, L.; Happel, M.; Hahn, K.; Andriole, E. J.; Poutsma, J. C. Proton Affinity of β-Oxalylaminoalanine (BOAA): Incorporation of Direct Entropy Correction into the Single-Reference Kinetic Method. J. Am. Soc. Mass Spectrom. 2005, 16, 1151–1161.
Drahos, L.; Vékey, K. How Closely Related are the Effective and the Real Temperature. J. Mass Spectrom.‘ 1999, 34, 79–84.
Ervin, K. M. Microcanonical Analysis of the Kinetic Method: The Meaning of the “Effective Temperature.”. Int. J. Mass Spectrom. 2000, 195/196, 271–284.
Kapota, C. Ph. D. Dissertation, Ecole Polytechnique, Palaiseau, 2005.
Author information
Authors and Affiliations
Corresponding author
Additional information
published online December 8, 2006
Rights and permissions
About this article
Cite this article
Wang, P., Wesdemiotis, C., Kapota, C. et al. The sodium ion affinities of simple Di-, Tri-, and tetrapeptides. J Am Soc Mass Spectrom 18, 541–552 (2007). https://doi.org/10.1016/j.jasms.2006.10.024
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2006.10.024