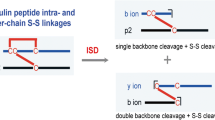
Abstract
Thio-ether bonds in the cysteinyl side chain of peptides, formed with the most commonly used cysteine blocking reagent iodoacetamide, after conversion to sulfoxide, releases a neutral fragment mass in a low-energy MS/MS experiment in the gas phase of the mass spectrometer [6]. In this study, we show that the neutral loss fragments produced from the mono-oxidized thio-ether bonds (sulfoxide) in peptides, formed by alkyl halide or double-bond containing cysteine blocking reagents are different under low-energy MS/MS conditions. We have evaluated the low-energy fragmentation patterns of mono-oxidized modified peptides with different cysteine blocking reagents, such as iodoacetamide, 3-maleimidopropionic acid, and 4-vinylpyridine using FTICR-MS. We propose that the mechanisms of gas-phase fragmentation of mono-oxidized thio-ether bonds in the side chain of peptides, formed by iodoacetamide and double-bond containing cysteine blocking reagents, maleimide and vinylpyridine, are different because of the availability of acidic β-hydrogens in these compounds. Moreover, we investigated the fragmentation characteristics of mono-oxidized thio-ether bonds within the peptide sequence to develop novel mass-spectrometry identifiable chemical cross-linkers. This methionine type of oxidized thio-ether bond within the peptide sequence did not show anticipated low-energy fragmentation. Electron capture dissociation (ECD) of the side chain thio-ether bond containing oxidized peptides was also studied. ECD spectra of the oxidized peptides showed a greater extent of peptide backbone cleavage, compared with CID spectra. This fragmentation information is critical to researchers for accurate data analysis of this undesired modification in proteomics research, as well as other methods that may utilize sulfoxide derivatives.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bogdanov, B.; Smith, R. D. Proteomics by FTICR Mass Spectrometry: Top Down and Bottom Up. Mass Spectrom. Rev. 2005, 24, 168–200.
Sechi, S.; Chait, B. T. Modification of Cysteine Residues by Alkylation: A Tool in Peptide Mapping and Protein Identification. Anal. Chem. 1998, 70, 5150–5158.
Schilling, B.; Yoo, C. B.; Collins, C. J.; Gibson, B. W. Determining Cysteine Oxidation Status Using Differential Alkylation. Int. J. Mass Spectrom. 2004, 236, 117–127.
Fullmer, C. S. Identification of Cysteine-Containing Peptides in Protein Digests by High-Performance Liquid Chromatography. Anal. Biochem. 1984, 142, 336–339.
March, J. Advance Organic Chemistry Reactions, Mechanisms and Structure; 4th ed. John Wiley & Sons, Inc.: New York, 1999, pp 795–797.
Steen, H.; Mann, M. Similarity Between Condensed Phase and Gas Phase Chemistry: Fragmentation of Peptides Containing Oxidized Cysteine Residues and Its Implications for Proteomics. J. Am. Soc. Mass Spectrom. 2001, 12, 228–232.
Steen, H.; Mann, M. A New Derivatization Strategy for the Analysis of Phosphopeptides by Precursor Ion Scanning in Positive Ion Mode. J. Am. Soc. Mass Spectrom. 2002, 13, 996–1003.
Anderson, G. A.; Bruce, J. E., Eds; ICR-2LS; Pacific Northwest National Laboratory: Richland, WA, 1995.
Tang, X.; Munske, G. R.; Siems, W. F.; Bruce, J. E. Mass Spectrometry Identifiable Cross-Linking Strategy for Studying Protein-Protein Interactions. Anal. Chem. 2005, 77, 311–318.
Chowdhury, S. M.; Munske, G. R.; Tang, X.; Bruce, J. E. CAD, ECD, of Several Mass Spectrometry-Identifiable Chemical Cross-Linkers. Anal. Chem. in press.
Caravatti, P.; Allemann, M. The “Infinity Cell”: A New Trapped-Ion Cell with Radiofrequency Covered Trapping Electrodes for Fourier Transform Ion Cyclotron Resonance Mass Spectrometry. Org. Mass Spectrom. 1991, 26, 514–518.
Mormann, M.; Maček, B.; De Peredo, A. G.; Hofsteenge, J.; Peter-Katalinič, J. Structural Studies on Protein O-Fucosylation by Electron Capture Dissociation. Int. J. Mass Spectrom 2004, 234, 11–21.
Adamson, J. T.; Håkansson, K. Infrared Multiphoton Dissociation and Electron Capture Dissociation of High-Mannose Type Glycopeptides. J. Proteome Res 2006, 5, 493–501.
Galeva, N. A.; Esch, S. W.; Williams, T. D.; Markille, L. M.; Squier, T. C. Rapid Method for Quantifying the Extent of Methionine Oxidation in Intact Calmodulin. J. Am. Soc. Mass Spectrom 2005, 16, 1470–1480.
Reid, G. E.; Roberts, K. D.; Kapp, E. A.; Simpson, R. I. Statistical and Mechanistic Approaches to Understanding the Gas-Phase Fragmentation Behavior of Methionine Sulfoxide Containing Peptides. J. Proteome Res 2004, 3, 751–759.
Guan, Z.; Yates, N. A.; Bakhtiar, R. Detection and Characterization of Methionine Oxidation in Peptides by Collision-Induced Dissociation and Electron Capture Dissociation. J. Am. Soc. Mass Spectrom 2003, 14, 605–613.
Smakman, R.; Boer, D. J. T. The Mass Spectra of Some Aliphatic and Alicyclic Sulphoxides and Sulphones. Org. Mass Spectrom 1970, 3, 1561–1588.
Lacombe, S.; Banchereau, M. L. E.; Simon, M.; Pfister-Guillouzo, G. Sulfenic Acids in the Gas Phase: A Photoelectron Study. J. Am. Chem. Soc 1996, 118, 1131–1138.
Turecek, F.; Drinkwater, D. E.; McLafferty, F. W. Gas-Phase Chemistry of CH3SOH, −CH2+SHOH, CH3SO·, and CH2SOH by Neutralization-Reionization Mass Spectrometry. J. Am. Chem. Soc. 1989, 111, 7696–7701.
Gross, E.; Morell, J. L. Structure of Niacin. J. Am. Chem. Soc. 1971, 93, 4634–4635.
Xie, L.; Van Der. Donk, W. A. Post-Translational Modifications During Lantibiotic Biosynthesis. Curr. Opin. Chem. Biol 2004, 8, 498–507.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online November 27, 2006
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chowdhury, S.M., Munske, G.R., Ronald, R.C. et al. Evaluation of low energy CID and ECD fragmentation behavior of mono-oxidized thio-ether bonds in peptides. J Am Soc Mass Spectrom 18, 493–501 (2007). https://doi.org/10.1016/j.jasms.2006.10.019
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2006.10.019