Abstract
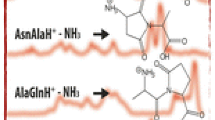
Aspartic acid (Asp)-containing peptides with the fixed charge derivative tris(2,4,6trimethoxyphenyl) phosphonium (tTMP-P+) were explored computationally and experimentally by hydrogen/deuterium (H/D) exchange and by fragmentation studies to probe the phenomenon of selective cleavage C-terminal to Asp in the absence of a “mobile” proton. Ab initio modeling of the tTMP-P+ electrostatic potential shows that the positive charge is distributed on the phosphonium group and therefore is not initiating or directing fragmentation as would a “mobile” proton. Geometry optimizations and vibrational analyses of different Asp conformations show that the Asp structure with a hydrogen bond between the side-chain hydroxy and backbone carbonyl lies 2.8 kcal/mol above the lowest energy conformer. In reactions with D2O, the phosphonium-derived doubly charged peptide (H+)P+LDIFSDF rapidly exchanges all 12 of its exchangeable hydrogens for deuterium and also displays a nonexchanging population. With no added proton, P+LDIFSDF exchanges a maximum of 4 of 11 exchangeable hydrogens for deuterium. No exchange is observed when all acidic groups are converted to the corresponding methyl esters. Together, these H/D exchange results indicate that the acidic hydrogens are “mobile locally” because they are able to participate in exchange even in the absence of an added proton. Fragmentation of two distinct (H+)P+LDIFSDF ion populations shows that the nonexchanging population displays selective cleavage, whereas the exchanging population fragments more evenly across the peptide backbone. This result indicates that H/D exchange can sometimes distinguish between and provide a means of separation of different protonation motifs and that these protonation motifs can have an effect on the fragmentation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Gavin, A. C.; Bosche, M.; Krause, R.; Grandi, P.; et al. Functional Organization of the Yeast Proteome by Systematic Analysis of Protein Complexes, Nature 2002, 415, 141–147.
Yates, J. R. Database Searching Using Mass Spectrometry Data. Electrophoresis 1998, 19, 893–900.
Link, A. J.; Eng, J.; Schieltz D. M.; et al. Direct Analysis of Protein Complexes Using Mass Spectrometry, Nat. Biotechnol. 1999, 17, 676–682.
Washburn, M. P.; Wolters, D.; Yates, J. R. Large-Scale Analysis of the Yeast Proteome by Multidimensional Protein Identification Technology. Nat. Biotechnol. 2001, 19, 242–247.
Fenn, J. B.; Mann, M.; Meng, C. K.; Wong, S. F.; Whitehouse, C. M. Electrospray Ionization for Mass-Spectrometry of Large Biomolecules. Science 1989, 246, 64–71.
Eng, J. K.; McCormack, A. L.; Yates, J. R. An Approach to Correlate Tandem Mass-Spectral Data of Peptides with Amino-Acid-Sequences in a Protein Database. J. Am. Soc. Mass Spectrom. 1994, 5, 976–989.
Perkins, D. N.; Pappin, D. J. C.; Creasy, D. M.; Cottrell, J. S. Probability-Based Protein Identification by Searching Seqeuence Databases Using Mass Spectrometry Data. Electrophoresis 1999, 20, 3551–3567.
Fenyo, D. Identifying the Proteome: Software Tools. Curr. Opin. Biotechnol. 2000, 11, 391–395.
Biemann, K. Contributions of Mass Spectometry to Peptide and Protein Structure. Biomed. Environ. Mass Spectrom 1988, 16, 99–111.
Yates, J. R.; Eng, J. K.; McCormack, A. L.; Schieltz, D. Method to Correlate Tandem Mass-Spectra of Modified Peptides to Amino-Acid-Sequences in the Protein Database. Anal. Chem. 1995, 67, 1426–1436.
Liebler, D. C.; Hansen, B. T.; Davey, S. W.; Tiscareno, L.; Mason, D. E. Peptide Sequence Motif Analysis of Tandem MS Data with the SALSA Algorithm. Anal. Chem. 2002, 74, 203–210.
Goodlett, D. R.; Keller, A.; Watts, J. D.; Newitt, R.; Yi, E. C.; et al. Differential Stable Isotope Labeling of Peptides for Quantitation and De Novo Sequence Derivation. Rapid Commun. Mass Spectrom. 2001, 15, 1214–1221.
Taylor, J. A.; Johnson, R. S. Sequence Database Searches via De Novo Peptide Sequencing by Tandem Mass Spectrometry. Rapid Commun. Mass Spectrom. 1997, 11, 1067–1075.
Mann, M.; Wilm, M. Error Tolerant Identification of Peptides in Sequence Databases by Peptide Sequence Tags. Anal. Chem. 1994, 66, 4390–4399.
Shevchenko, A.; Sunyaev, S.; Loboda, A.; Shevehenko, A.; et al. Charting the Proteomes of Organisms with Unsequenced Genomes by MALDI-Quadrupole Time of Flight Mass Spectrometry and BLAST Homology Searching. Anal. Chem. 2001, 73, 1917–1926.
Mann, M.; Hendrickson, R. C.; Pandey, A. Analysis of Proteins and Proteomes by Mass Spectrometry. Ann. Rev. Biochem. 2001, 70, 437–473.
Johnson, R. S.; Taylor, J. A. Searching Sequence Databases via De Novo Peptide Sequencing by Tandem Mass Spectrometry. Mol. Biotechnol. 2002, 22, 301–315.
Simpson, R. J.; Connolly, L. M.; Eddes, J. S.; et al. Proteomic Analysis of the Human Colon Carcinoma Cell Line (LIM 1215): Development of a Membrane Protein Database. Electrophoresis 2000, 21, 1707–1732.
Breci, L. A.; Tabb, D. L.; Yates, J. R.; Wysocki, V. H. Cleavage N-Terminal to Proline: Analysis of a Database of Peptide Tandem Mass Spectra. Anal. Chem. 2003, 75, 1963–1971.
Huang, Y. Y.; Wysocki, V. H.; Tabb, D. L.; Yates, J. R. The Influence of Histidine on Cleavage C-Terminal to Acidic Residues in Doubly Protonated Tryptic Peptides. Int. J. Mass Spectrom. 2002, 219, 233–244.
Kapp, E. A.; Schutz, F.; Reid, G. E.; Eddes, J. S.; et al. Mining a Tandem Mass Spectrometry Database to Determine the Trends and Global Factors Influencing Peptide Fragmentation. Anal. Chem. 2003, 75, 6251–6264.
Tabb, D. L.; Smith, L. L.; Breci, L. A.; et al. Statistical Characterization of Ion Trap Tandem Mass Spectra from Doubly Charged Tryptic Peptides. Anal Chem 2003, 75, 1155–1163.
Tabb, D. L.; Huang, Y. Y.; Wysocki, V. H.; Yates, J. R. Influence of Basic Residue Content on Fragment Ion Peak Intensities in Low-Energy—Collision-Induced Dissociation Spectra of Peptides. Anal. Chem. 2004, 76, 1243–1248.
Huang, Y. Y.; Triscari, J. M.; Pasa-Tolic, L.; et al. Dissociation Behavior of Doubly-Charged Tryptic Peptides: Correlation of Gas-Phase Cleavage Abundance with Ramachandran Plots. J. Am. Chem. Soc. 2004, 126, 3034–3035.
Ambihapathy, K.; Yalcin, T.; Leung, H. W.; Harrison, A. G. Pathways to Immonium Ions in the Fragmentation of Protonated Peptides. J. Mass Spectrom. 1997, 32, 209–215.
Nair, H.; Wysocki, V. H. Are Peptides without Basic Residues Protonated Primarily at the Amino Terminus? Int. J. Mass Spectrom. 1998, 174, 95–100.
Polce, M. J.; Ren, D.; Wesdemiotis, C. Special Feature: Commentary—Dissociation of the Peptide Bond in Protonated Peptides. J. Mass Spectrom. 2000, 35, 1391–1398.
Schlosser, A.; Lehmann, W. D. Special Feature: Commentary—Five-Membered Ring Formation in Unimolecular Reactions of Peptides: a Key Structural Element Controlling Low-Energy Collision-Induced Dissociation of Peptides. J. Mass Spectrom. 2000, 35, 1382–1390.
Salek, M.; Lehmann, W. D. Neutral Loss of Amino Acid Residues from Protonated Peptides in Collision-Induced Dissociation Generates N- or C-Terminal Sequence Ladders. J. Mass Spectrom. 2003, 38, 1143–1149.
Tsaprailis, G.; Nair, H.; Somogyi, A.; Wysocki, V. H.; et al. Influence of Secondary Structure on the Fragmentation of Protonated Peptides. J. Am. Chem. Soc. 1999, 121, 5142–5154.
Wysocki, V. H.; Tsaprailis, G.; Smith, L. L.; Breci, L. A. Special Feature: Commentary—Mobile and Localized Protons: A Framework for Understanding Peptide Dissociation. J. Mass Spectrom. 2000, 35, 1399–1406.
Yague, J.; Paradela, A.; Ramos, M.; et al. Peptide Rearrangement during Quadrupole Ion Trap Fragmentation: Added Complexity to MS/MS Spectra. Anal. Chem. 2003, 75, 1524–1535.
Grewal, R. N.; El Aribi, H.; Harrison, A. G.; et al. Fragmentation of Protonated Tripeptides: The Proline Effect Revisited. J. Phys. Chem. B 2004, 108, 4899–4908.
Gu, C. G.; Tsaprailis, G.; Breci, L.; Wysocki, V. H. Selective Gas-Phase Cleavage at the Peptide Bond Terminal to Asparatic Acid in Fixed-Charge Derivatives of Asp-Containing Peptides. Anal. Chem. 2000, 72, 5804–5813.
Tsaprailis, G.; Somogyi, A.; Nikolaev, E. N.; Wysocki, V. H. Refining the Model for Selective Cleavage at Acidic Residues in Arginine-Containing Protonated Peptides. Int. J. Mass Spectrom. 2000, 196, 467–479.
Wattenberg, A.; Organ, A. J.; Schneider, K.; et al. Sequence Dependent Fragmentation of Peptides Generated by MALDI Quadrupole Time-of-Flight (MALDI Q-TOF) Mass Spectrometry and Its Implications for Protein Identification. J. Am. Soc. Mass Spectrom. 2002, 13, 772–783.
Leymarie, N.; Berg, E. A.; McComb, M. E.; et al. Tandem Mass Spectrometry for Structural Characterization of Proline-Rich Proteins: Application to Salivary PRP-3. Anal. Chem. 2002, 74, 4124–4132.
Yu, W.; Vath, J. E.; Huberty, M. C.; Martin, S. A. Identification of the Facile Gas-Phase Cleavage of the Asp Pro and Asp Xxx Peptide-Bonds in Matrix-Assisted Laser-Desorption Time-of-Flight Mass-Spectrometry. Anal Chem 1993, 65, 3015–3023.
Burlet, O.; Yang, C. Y.; Gaskell, S. J. Influence of Cysteine to Cysteic Acid Oxidation on the Collision-Activated Decomposition of Protonated Peptides—Evidence for Intraionic Interactions. J. Am. Soc. Mass Spectrom. 1992, 3, 337–344.
Burlet, O.; Yang, C. Y.; Guyton, J. R.; Gaskell, S. J. Tandem Mass-Spectrometric Characterization of a Specific Cysteic Acid Residue in Oxidized Human Apoprotein B-100. J. Am. Soc. Mass Spectrom. 1995, 6, 242–247.
Qin, J.; Chait, B. T. Preferential Fragmentation of Protonated Gas-Phase Peptide Ions Adjacent to Acidic Amino-Acid-Residues. J. Am. Chem. Soc. 1995, 117, 5411–5412.
Summerfield, S. G.; Cox, K. A.; Gaskell, S. J. The Promotion of D-Type Ions during the Low Energy Collision-Induced Dissociation of Some Cysteic Acid-Containing Peptides. J. Am. Soc. Mass Spectrom. 1997, 8, 25–31.
Wang, Y. S.; Vivekananda, S.; Men, L. J.; Zhang, Q. B. Fragmentation of Protonated Ions of Peptides Containing Cysteine, Cysteine Sulfinic Acid, and Cysteine Sulfonic Acid. J. Am. Soc. Mass Spectrom. 2004, 15, 697–702.
Zhang, Z. Prediction of Low-Energy Collision-Induced Dissociation Spectra of Peptides. Anal Chem. 2004, 76, 3908–3922.
Huang, Y. Y.; Triscari, J. M.; Tseng, G. C.; et al. Statistical Characterization of the Charge State and Residue Dependence of Low Energy CID Peptide Dissociation Patterns. Anal. Chem. 2005, in press.
Komaromi, I.; Somogyi, A.; Wysocki, V. H. Proton Migration and Its Effect on the MS Fragmentation of N-Acetyl OMe Proline: MS/MS Experiments and Ab Initio and Density Functional Calculations. Int. J. Mass Spectrom. 2005, 241, 315–323.
Reid, G. E.; Simpson, R. J.; O’Hair, R. A. J. A Mass Spectrometric and Ab Initio Study of the Pathways for Dehydration of Simple Glycine and Cysteine-Containing Peptide M+H (+) Ions. J. Am. Soc. Mass Spectrom. 1998, 9, 945–956.
Paizs, B.; Lendvay, G.; Vekey, K.; Suhai, S. Formation of b(2)(+) Ions from Protonated Peptides: An Ab Initio Study. Rapid Commun. Mass Spectrom. 1999, 13, 525–533.
Nold, M. J.; Wesdemiotis, C.; Yalcin, T.; Harrison, A. G. Amide Bone Dissociation in Protonated Peptides. Structures of the N-Terminal Ionic and Neutral Fragments. Int. J. Mass Spectrom. Ion Process. 1997, 164, 137–153.
Vaisar, T.; Urban, J. Low-Energy Collision Induced Dissociation of Protonated Peptides. Importance of an Oxazolone Formation for a Peptide Bond Cleavage. Eur. Mass Spectrom. 1998, 4, 359–364.
Vaisar, T.; Urban, J. Gas-Phase Fragmentation of Protonated Mono-N-Methylated Peptides. Analogy with Solution-Phase Acid-Catalyzed Hydrolysis. J. Mass Spectrom. 1998, 33, 505–524.
Harrison, A. G.; Csizmadia, I. G.; Tang, T. H. Structure and Fragmentation of b(2) Ions in Peptide Mass Spectra. J. Am. Soc. Mass Spectrom. 2000, 11, 427–436.
Wyttenbach, T.; Paizs, B.; Barran, P.; The Effect of the Initial Water of Hydration on the Energetics, Structures, and H/D Exchange Mechanism of a Family of Pentapeptides: An Experimental and Theoretical Study. J. Am. Chem. Soc. 2003, 125, 13768–13775.
Campbell, S.; Rodgers, M. T.; Marzluff, E. M.; Beauchamp, J. L. Deuterium Exchange Reactions as a Probe of Biomolecule Structure. Fundamental Studies of Gas Phase H/D Exchange Reactions of Protonated Glycine Oligomers with D2O, CD3OD, CD3CO2D, and ND3. J. Am. Chem. Soc. 1995, 117, 12840–12854.
Gard, E.; Green, M. K.; Bregar, J.; Lebrilla, C. B. Gas-Phase Hydrogen/Deuterium Exchange as a Molecular Probe for the Interaction of Methanol and Protonated Peptides. J. Am. Soc. Mass Spectrom. 1994, 5, 623–631.
High Performance Computational Chemistry Group. NWChem, A Computational Chemistry Package for Parallel Computers; Pacific Northwest National Laboratory: Richland, WA, 2001.
Black, G. C., J.; Didier, B.; Feller, D.; Gracio, D.; Jones, D.; Keller, T.; Matsumoto, S.; Mendoza, E.; Olander, M.; Palmer, B.; Peden, N.; Schchardt, K.; Taylor, H.; Thomas, G.; Vorpagel, E.; Windus, T. Ecce, A Problem Solving Environment for Computational Software; 1.5 ed.; Pacific Northwest National Laboratory: Richland, WA 99352, 1999.
Dunning, T. H.; Hay, P. J. In Methods of Electronic Structure Theory; Schaefer, H. F., III, Ed.; Plenum Press: New York, 1977.
Magnusson, E.; Schaefer, H. F. Multiple D-Type Basis Functions for Molecules Containing 2nd Row Atoms. J. Chem. Phys. 1985, 83, 5721–5726.
Andzelm J.; et al. In Density Functional Methods in Chemistry; Labanowski, J., Andzelm, J., Eds.; Springer-Verlag: New York, 1991; pp 155.
Ziegler, T. Approximate Density Functional Theory as a Practical Tool in Molecular Energetics and Dynamics. Chemical Reviews 1991, 91, 651–667.
Becke, A. D. Density-Functional Thermochemistry. 3. The Role of Exact Exchange. J. Chem. Phys. 1993, 98, 5648–5652.
Lee, C.; Yang, W.; Parr, R. G. Development of the Colle-Salvetti Correlation-Energy Formula into a Functional of the Electron-Density. Physical Review B 1988, 37, 785–789.
Cieplak, P.; Cornell, W. D.; Bayly, C.; Kollman, P. A. Application of the Multimolecule and Multiconformational Resp Methodology to Biopolymers—Charge Derivation for DNA, RNA, and Proteins. J. Computat. Chem. 1995, 16, 1357–1377.
Bayly, C.; Cieplak, P.; Cornell, W. D.; Kollman, P. A. A Well-Behaved Electrostatic Potential Based Method Using Charge Restraints for Deriving Atomic Charges: The RESP Model. J. Phys. Chem. 1993, 97, 10269–10280.
Jiao, C. Q.; Ranatunga, D. R. A.; Vaughn, W. E.; Freiser, B. S. A Pulsed-Leak Valve for Use with Ion Trapping Mass Spectrometers. J. Am. Soc. Mass Spectrom. 1996, 7, 118–122.
Bartmess, J. E.; Georgiadis, R. M. Empirical-Methods for Determination of Ionization Gauge Relative Sensitivities for Different Gases. Vacuum 1983, 33, 149–153.
Gronert, S. Estimation of Effective Ion Temperatures in a Quadrupole Ion Trap. J. Am. Soc. Mass Spectrom. 1998, 9, 845–848.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online May 25, 2005
Rights and permissions
About this article
Cite this article
Herrmann, K.A., Wysocki, V.H. & Vorpagel, E.R. Computational investigation and hydrogen/deuterium exchange of the fixed charge derivative tris(2,4,6-Trimethoxyphenyl) phosphonium: Implications for the aspartic acid cleavage mechanism. J Am Soc Mass Spectrom 16, 1067–1080 (2005). https://doi.org/10.1016/j.jasms.2005.03.028
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2005.03.028