Abstract
Oligoribonucleotides (RNA) and modified oligonucleotides were subjected to low-energy collision-induced dissociation in a hybrid quadrupole time-of-flight mass spectrometer to investigate their fragmentation pathways. Only very restricted data are available on gas-phase dissociation of oligoribonucleotides and their analogs and the fundamental mechanistic aspects still need to be defined to develop mass spectrometry-based protocols for sequence identification. Such methods are needed, because chemically modified oligonucleotides can not be submitted to standard sequencing protocols.
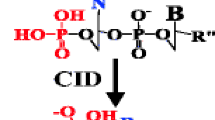
In contrast to the dissociation of DNA, dissociation of RNA was found to be independent of nucleobase loss and it is characterized by cleavage of the 5′-P-O bond, resulting in the formation of c- and their complementary y-type ions. To evaluate the influence of different 2′-substituents, several modified tetraribonucleotides were analyzed. Oligoribonucleotides incorporating a 2′-methoxy-ribose or a 2′-fluoro-ribose show fragmentation that does not exhibit any preferred dissociation pathway because all different types of fragment ions are generated with comparable abundance. To analyze the role of the nucleobases in the fragmentation of the phosphodiester backbone, an oligonucleotide lacking the nucleobase at one position has been studied. Experiments indicated that the dissociation mechanism of RNA is not influenced by the nucleobase, thus, supporting a mechanism where dissociation is initiated by formation of an intramolecular cyclic transition state with the 2′-hydroxyl proton bridged to the 5′-phosphate oxygen.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zamecnik, P. C.; Stephenson, M. L. Inhibition of Rous Sarcoma Virus Replication and Cell Transformation by a Specific Oligodeoxynucleotide. Proc. Natl. Acad. Sci. U.S.A. 1978, 75, 280–284.
Stephenson, M. L.; Zamecnik, P. C. Inhibition of Rous sarcoma Viral RNA Translation by a Specific Oligodeoxyribonucleotide. Proc. Natl. Acad. Sci. U.S.A. 1978, 75, 285–288.
Leumann, C. J. DNA Analogues: From Supramolecular Principles to Biological Properties. Bioorg. Med. Chem. 2002, 10, 841–854.
Baker, B. F.; Monia, B. P. Novel Mechanisms for Antisense-Mediated Regulation of Gene Expression. Biochim. Biophys. Acta 1999, 1489, 3–18.
Goodchild, J. Oligonucleotide Therapeutics: 25 Years Agrowing. Curr. Opin. Mol. Ther. 2004, 6, 120–128.
Altmann, K. H.; Dean, N. M.; Fabbro, D.; Freier, S. M.; Geiger, T.; Häner, R.; Hüsken, D.; Martin, P.; Monia, B. P.; Müller, M.; Natt, F.; Nicklin, P.; Phillips, J.; Piels, U.; Sasmor, H.; Moser, H. E. Second Generation of Antisense Oligonucleotides: From Nuclease Resistance to Biological Efficacy in Animals. Chimia. 1996, 50, 168–176.
Myers, K. J.; Dean, N. M. Sensible Use of Antisense: How to Use Oligonucleotides as Research Tools. TIPS 2000, 21, 19–23.
Campbell, J. M.; Bacon, T. A.; Wickstrom, E. Oligodeoxynucleoside Phosphorothioate Stability in Subcellular Extracts, Culture Media, Sera and Cerebrospinal Fluid. J. Biochem. Biophys. Methods 1990, 20, 259–267.
Stein, C. A.; Tonkinson, J. L.; Yakubov, L. Phosphorothioate Oligodeoxynucleotides-Anti-Sense Inhibitors of Gene Expression? Pharmacol. Ther. 1991, 52, 365–384.
Tang, W.; Zhu, L.; Smith, L. M. Controlling DNA Fragmentation in MALDI-MS by Chemical Modification. Anal. Chem. 1997, 69, 302–312.
Sanger, F.; Nicklen, S.; Coulson, A. R. D. N. A. Sequencing with Chain-Terminating Inhibitors. Proc. Natl. Acad. Sci. U.S.A. 1977, 74, 5463–5467.
Dovichi, N. J.; Zhang, J. How capillary electrophoresis sequenced the human genome. Angew. Chem. Int. Ed. 2000, 39, 4463–4468.
Wan, K. X.; Gross, J.; Hillenkamp, F.; Gross, M. L. Fragmentation Mechanisms of Oligodeoxynucleotides Studied by H/D Exchange and Electrospray Ionization Tandem Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2001, 12, 193–205.
Wang, Z.; Wan, K. X.; Ramanathan, R; Taylor, John S.; Gross, M. L. Structure and Fragmentation Mechanisms of Isomeric T-Rich Oligodeoxynucleotides: A Comparison of Four Tandem Mass Spectrometric Methods. J. Am. Soc. Mass. Spectrom. 1998, 9, 683–691.
McLuckey, S. A.; Habibi-Goudarzi, S. Decompositions of Multiply Charged Oligonucleotide Anions. J. Am. Chem. Soc. 1993, 115, 12085–12095.
McLuckey, S. A.; Van Berkel, G. J.; Glish, G. L. Tandem Mass Spectrometry of Small, Multiply Charged Oligonucleotides. J. Am. Soc. Mass Spectrom. 1992, 3, 60–70.
Bartlett, M. G.; McCloskey, J. A.; Manalili, S.; Griffey, R. H. The Effect of Backbone Charge on the Collision-Induced Dissociation of Oligonucleotides. J. Mass Spectrom. 1996, 31, 1277–1283.
Wan, K. X.; Gross, M. L. Fragmentation Mechanisms of Oligodeoxynucleotides: Effects of Replacing Phosphates with Methylphosphonates and Thymines with Other Bases in T-Rich Sequences. J. Am. Soc. Mass Spectrom. 2001, 12, 580–589.
Yacyshyn, B. R.; Chey, W. Y.; Goff, J.; Salzberg, B.; Baerg, R.; Buchman, A. L.; Tami, J.; Yu, R.; Gibiansky, E.; Shanahan, W. R. Double Blind, Placebo Controlled Trial of the Remission Inducing and Steroid Sparing Properties of an ICAM-1 Antisense Oligodeoxynucleotide, Alicaforsen (ISIS 2302), in Active Steroid Dependent Crohn’s Disease. Gut 2002, 51, 30–36.
Sewell, K. L.; Geary, R. S.; Baker, B. F.; Glover, J. M.; Mant, T. G.; Yu, R. Z.; Tami, J. A.; Dorr, A. Phase I Trial of ISIS 104838, a 2′-Methoxyethyl Modified Antisense Oligonucleotide Targeting Tumor Necrosis Factor-a. JPET 2002, 303, 1334–1343.
Herbst, R. S.; Frankel, S. R. Oblimersen Sodium (Genasense bcl-2 Antisense Oligonucleotide): A Rational Therapeutic to Enhance Apoptosis in Therapy of Lung Cancer. Clin. Cancer Res. 2004, 10, 4245–4248.
Wang, B. H.; Hopkins, C. E.; Belenky, A. B.; Cohen, A. S. Sequencing of Modified Oligonucleotides Using In-Source Fragmentation and Delayed Pulsed Ion Extraction Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry. Int. J. Mass Spectrom. Ion Proccess 1997, 169/170, 331–350.
Sannes-Lowery, K. A.; Hofstadler, S. A. Sequence Confirmation of Modified Oligonucleotides Using IRMPD in the External Ion Reservoir of an Electrospray Ionization Fourier Transform Ion Cyclotron Mass Spectrometer. J. Am. Soc. Mass Spectrom. 2003, 14, 825–833.
Cerny, R. L.; Tomer, K. B.; Gross, M. L.; Grotjahn, L. Fast Atom Bombardment Combined with Tandem Mass Spectrometry for Determining Structures of Small Oligonucleotides. Anal. Biochem. 1987, 165, 175–182.
Kirpekar, F.; Krogh, T. N. RNA Fragmentation Studied in a Matrix-Assisted Laser Desorption/Ionisation Tandem Quadrupole/Orthogonal Time-of-Flight Mass Spectrometer. Rapid Commun. Mass Spectrom. 2001, 15, 8–14.
Schürch, S.; Bernal-Mendez, E.; Leumann, C. J. Electrospray Tandem Mass Spectrometry of Mixed-Sequence RNA/DNA Oligonucleotides. J. Am. Soc. Mass Spectrom. 2002, 13, 936–945.
Nordhoff, E.; Cramer, R.; Karas, M.; Hillenkamp, F.; Kirpekar, F.; Kristiansen, K.; Roepstorff, P. Ion Stability of Nucleic Acids in Infrared Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry. Nucleic Acids Res. 1993, 21, 3347–3357.
Ni, J.; Pomerantz, S. C.; Rozenski, J.; Zhang, Y.; McCloskey, J. A. Interpretation of Oligonucleotide Mass Spectra for Determination of Sequence Using Electrospray Ionization and Tandem Mass Spectrometry. Anal. Chem. 1996, 68, 1989–1999.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online June 23, 2005
Rights and permissions
About this article
Cite this article
Tromp, J.M., Schürch, S. Gas-phase dissociation of oligoribonucleotides and their analogs studied by electrospray ionization tandem mass spectrometry. J Am Soc Mass Spectrom 16, 1262–1268 (2005). https://doi.org/10.1016/j.jasms.2005.03.024
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2005.03.024