Abstract
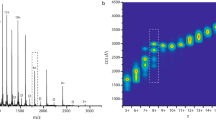
The protein α-synuclein, implicated in Parkinson’s disease, was studied by combining nano-electrospray ionization (N-ESI) mass spectrometry and ion mobility. It was found that both the charge-state distribution in the mass spectra and the average protein shape deduced from ion mobility data, depend on the pH of the spray solution. Negative-ion N-ESI of pH 7 solutions yielded a broad charge-state distribution from −6 to −16, centered at −11, and ion mobility data consistent with extended protein structures. Data obtained for pH 2.5 solutions, on the other hand, showed a narrow charge-state distribution from −6 to −11, centered at −8, and ion mobilities in agreement with compact α-synuclein structures. The data indicated that there are two distinct families of structures: one consisting of relatively compact proteins with eight or less negative charges and one consisting of relatively extended structures with nine or more charges. The average cross section of a-synuclein at pH 2.5 is 33% smaller than for the extended protein sprayed from pH 7 solution. Significant dimer formation was observed when sprayed from pH 7 solution but no dimers were observed from the low pH solution. A plausible mechanism for aggregate formation in solution is proposed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dunnett, S. B.; Bjorklund, A. Prospects for new restorative and neuroprotective treatments in Parkinson’s disease. Nature Suppl. 1999, 399(6738), A32-A39.
Baba, M.; Nakajo, S.; Tu, P. H.; Tomita, T.; Nakaya, K.; Lee, V. M. Y.; Trojanowski, J. Q.; Iwatsubo, T. Aggregation of Alpha-synuclein in Lewy bodies of sporadic Parkinson’s disease and dementia with Lewy Bodies. Am. J. Path. 1998, 152, 879–884.
Spillantini, M. G.; Schmidt, M. L.; Lee, V. M. Y.; Trojanowski, J. Q.; Jakes, R.; Goedert, M. Alpha-synuclein in Lewy bodies. Nature 1997, 388, 839–840.
Selkoe, D. J. Translating cell biology into therapeutic advances in Alzheimer’s disease. Nature Suppl. 1999, 399, A23-A31.
Bussell, R.; Eliezer, D. Residual structure and dynamics in Parkinson’s disease-associated mutants of alpha-synuclein. J. Biol. Chem. 2001, 276, 45996–46003.
Serpell, L. C.; Berriman, J.; Jakes, R.; Goedert, M.; Crowther, R. A. Fiber diffraction of synthetic alpha-synuclein filaments shows amyloid-like cross-beta conformation. Proc. Natl. Acad. Sci. USA 2000, 97, 4897–4902.
Uversky, V. N.; Li, J.; Fink, A. L. Metal-triggered structural transformations, aggregation, and fibrillation of human alpha-synuclein—A possible molecular link between Parkinson’s disease and heavy metal exposure. J. Biol. Chem. 2001, 276, 44284–44296.
Eliezer, D.; Kutluay, E.; Bussell, R.; Brown, G. Conformational properties of alpha-synuclein in its free and lipid-associated states. J. Mol. Biol. 2001, 307, 1061–1073.
Volles, M. J.; Lee, S. J.; Rochet, J. C.; Shtilerman, M. D.; Ding, T. T.; Kessler, J. C.; Lansbury, P. T. Vesicle permeabilization by protofibrillar alpha-synuclein: Implications for the pathogenesis and treatment of Parkinson’s disease. Biochemistry 2001, 40, 7812–7819.
Conway, K. A.; Harper, J. D.; Lansbury, P. T. Accelerated in vitro fibril formation by a mutant alpha-synuclein linked to early-onset Parkinson disease. Nature Med. 1998, 4, 1318–1320.
Benjamin, D. R.; Robinson, C. V.; Hendrick, J. P.; Hartl, U.; Dobson, C. M. Mass Sepctrometry of Ribosomes and Ribosomal subunits. Proc. Nat. Acad. 1998, 95, 7391–7395.
Rostom, A. A.; Fucini, P.; Benjamin, D. R.; Juenemann, R.; Nierhaus, K. H.; Hartl, F. U.; Dobson, C. M.; Robinson, C. V. Detection and selective dissociation of intact Ribosomes in a mass spectrometer. Proc. Natl. Acad. Sci. 2000, 97, 5185–5190.
Nettleton, E. J.; Tito, P.; Sunde, M.; Bouchard, M.; Dobson, C. M.; Robinson, C. V. Characterization of the oligomeric states of insulin in self-assembly and amyloid fibril formation by mass spectrometry. J. Biophys. 2000, 79, 1053–1065.
von Helden, G.; Hsu, M. T.; Kemper, P. R.; Bowers, M. T. Structures of carbon cluster ions from 3 to 60 atoms: Linears to rings to fullerenes. J. Chem. Phys. 1991, 95, 3835–3837.
Clemmer, D. E.; Jarrold, M. F. Ion Mobility measurements and their applications to clusters and biomolecules. J. Mass Spectrom. 1997, 92, 577.
Wyttenbach, T.; Bowers, M. T. Gas-Phase conformations: The Ion Mobility/Ion Chromatography method. Top. Curr. Chem. 2003, 225, 207–232.
Wyttenbach, T.; von Helden, G.; Bowers, M. T. Gas-Phase conformation of biological molecules: Bradykinin. J. Am. Chem. Soc. 1996, 118, 8355–8364.
Gidden, J.; Wyttenbach, T.; Jackson, A. T.; Scrivens, J. H.; Bowers, M. T. Gas-phase conformations of synthetic polymers: Poly(ethylene glycol), Poly(propylene glycol), and Poly(tetramethylene glycol). J. Am. Chem. Soc. 2000, 122, 4692–4699.
Gidden, J.; Bowers, M. T. Gas-phase conformations of deprotonated trinucleotides (dGTT−, dTGT−, and dTTG−): The question of zwitterion formation. J. Am. Soc. Mass Spectrom. 2003, 14, 161–170.
Clemmer, D. E.; Hudgins, R. R.; Jarrold, M. F. Naked protein conformation: Cytochrome c in the gas phase. J. Am. Chem. Soc. 1995, 117, 1014.
Shelimov, K. B.; Clemmer, D. E.; Hudgins, R. R.; Jarrold, M. F. Protein structure in vacuo: Gas-phase conformations of BPTI and Cytochrome c. J. Am. Chem. Soc. 1997, 119, 2240.
Hudgins, R. R.; Ratner, M. A.; Jarrold, M. F. Design of helices that are stable in vacuo. J. Am. Chem. Soc. 1998, 120, 12974.
Kohtani, M.; Kinnear, B. S.; Jarrold, M. F. Metal-ion enhanced helicity in the gas phase. J. Am. Chem. Soc. 2000, 122, 12377.
Li, J.; Taraszka, J. A.; Counterman, A. E.; Clemmer, D. E. Influence of solvent composition and capillary temperature on the conformations of electrosprayed ions: unfolding of compact ubiquitin conformers from pseudonative and denatured solutions. Int. J. Mass Spectrom. 1999, 185, 37.
Counterman, A. E.; Clemmer, D. E. Large Anhydrous Polyalanine ions: Evidence for extended helices and onset of a more compact state. J. Am. Chem. Soc. 2001, 123, 1490.
Jakes, R.; Spillantini, M. G.; Goedert, M. Identification of 2 distinct synucleins from human brain. FEBS Lett. 1994, 345, 27–32.
Der-Sarkissian, A.; Jao, C. C.; Chen, J.; Langen, R. Structural organization of alpha-synuclein fibrils studied by site-directed spin labeling. J. Biol. Chem. 2003, 37530–37535.
Wyttenbach, T.; Kemper, P. R.; Bowers, M. T. Design of a New Electrospray Ion Mobility Mass Spectrometer. Int. J. Mass Spectrom. 2001, 212, 13–23.
Mason, E. A.; McDaniel, E. W. Transport Properties of Ions in Gases. Wiley, New York 1978.
Case, D. A.; Pearlman, D. A.; Caldwell, J. W.; Cheatham, T. E., III.; Wang, J.; Ross, W. S.; Simmerling, C. L.; Darden, T. A.; Merz, K. M.; Stanton, R. V.; Cheng, A. L.; Vincent, J. J.; Crowley, M.; Tsui, V.; Radmer, R. J.; Duan, Y.; Pitera, J.; Massova, I.; Seibel, G. L.; Singh, U. C.; Weiner, P. K.; Kollman, P. A. AMBER 7 Unversity of California: San Francisco, 2002.
Mathews, C. K.; van Holde, K. E. Biochemistry. Benjamin/Cummings, Redwood City, California; 1990.
Shvartsburg, A. A.; Jarrold, M. F. An Exact hard-spheres scattering model for the mobilities of polyatomic ions. Chem Phys Let. 1996, 261, 86–91.
Chowdhury, S. K.; Katta, V.; Chait, B. T. Probing conformational changes in proteins by mass spectromery. J. Am. Chem. Soc. 1990, 112, 9012–9013.
Jarrold, M. F. Peptides and proteins in the vapor phase. Annu. Rev. Phys. Chem. 2000, 51, 179–207.
Uversky, V. N.; Gillespie, J. R.; Fink, A. L. Why are “Natively unfolded” proteins unstructured under physiologic conditions?. Proteins: Struct, Funct, and Genetic. 2000, 41, 415–427.
Uversky, V. N.; Li, J.; Fink, A. L. Evidence of partially folded intermediate in a-synuclein fibril formation. J. Biol. Chem. 2001, 276, 10737–10744.
von Helden, G.; Gotts, N. G.; Bowers, M. T. Experimental evidence for the formation of Fullerenes by collisional heating of carbon rings in the gas phase. Nature 1993, 363, 60–63.
Last, A. M.; Robinson, C. V. Protein folding and interactions revealed by Mass Spectrometry. Curr. Opin. Chem. Biol. 1999, 3, 564–570.
Wyttenbach, T.; von Helden, G.; Bowers, M. T. Conformations of alkali ion cationized Polyethers in the gas phase: Polyethylene Glycol and Bis[(benzo-15-crown-5)-15-ylmethyl Pimelate. Int. J. Mass Spectrom. Ion Proc. 1997, 165, 377.
Gidden, J.; Bushnell, J. E.; Bowers, M. T. Gas-Phase Conformations and Folding Energetics of Oligonucleotides: dTG− and dGT−. J. Am. Chem. Soc. 2001, 123, 5610–5611.
Bernstein, S. L.; Wyttenbach, T.; Baumketner, A.; Shea J.-E.; Bitan, G.; Teplow, D. T.; Bowers, M. T. Amyloid β-protein: Monomer structure and early aggregation states of Aβ42 and its Pro19 alloform. J. Am. Chem. Soc. (submitted).
Gidden, J.; Ferzoco, A.; Baker, E. S.; Bowers, M. T. Duplex formation and the onset of helicity in dCG oligonucleotides in a solvent-free environment. J. Am. Chem. Soc. (in press).
Gidden, J.; Baker, E. S.; Ferzoco, A.; Bowers, M. T. Structural motifs of DNA complexes in the gas phase. Int. J. Mass Spectrom. (in press).
Baker, E. S.; Bernstein, S. L.; Bowers, M. T. (mss in preparation).
Rostam, A. A.; Robinson, C. V. Detection of Intact GroEL chaperonin assembly by Mass Spectrometry. J. Am. Soc. 1999, 121, 4718–4719.
Bitan, G.; Kirkitadze, M. D.; Lomakin, A.; Vollers, S. S.; Benedek, G. B.; Teplow, D. B. Amyloid β-protein (Aβ) assembly: Aβ40 and Aβ oligomerization through distinct pathways. Proc. Natl. Acad. Sci. USA 2003, 100(1), 330–335.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online September 11, 2004
Rights and permissions
About this article
Cite this article
Bernstein, S.L., Liu, D., Wyttenbach, T. et al. α-Synuclein: Stable compact and extended monomeric structures and pH dependence of dimer formation. J Am Soc Mass Spectrom 15, 1435–1443 (2004). https://doi.org/10.1016/j.jasms.2004.08.003
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2004.08.003