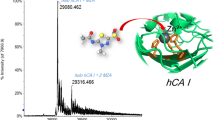
Abstract
In this study, a GlcNAc-6-O-Sulfotransferase, NodST and its complexation with the substrate 3′-phosphoadenosine 5′-phosphosulfate (PAPS) and the inhibitor 3′-phosphoadenosine 5′-phosphate (PAP) were studied using Fourier transform ion cyclotron resonance (FTICR) mass spectrometry. In addition, using isotopically labeled substrate, we have successfully confirmed a sulfated enzyme intermediate, which was predicted by the MS kinetic measurement. It is also shown that information regarding solution binding affinities can be obtained using electrospray ionization (ESI)-FTICR mass spectrometry. The relative binding constants, Kd(PAPS)/Kd(PAP), derived from the solution and gas phase were very similar, which suggests that the binding domain of this particular enzyme system, given known structures of other sulfotransferases, may be preserved during the transmission of the complex from solution to the gas phase.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Deng Y.; Smith D. L. Hydrogen Exchange Demonstrates Three Domains in Aldolase Unfold Sequentially. J. Mol. Biol. 1999, 294, 247–258.
Oda Y.; Nagasu T.; Chait B. T. Enrichment Analysis of Phosphorylated Proteins as a Tool for Probing the Phosphoproteome. Nat. Biotechnol. 2001, 19, 379–382.
Gygi S. P.; Rist B.; Aebersold R. Measuring Gene Expression by Quantitative Proteome Analysis. Curr. Opin. Biotechnol. 2000, 11, 396–401.
Aquilina J. A.; Benesch J. L. P.; Bateman O. A.; Slingsby C.; Robinson C. V. Polydispersity of a Mammalian Chaperone: Mass Spectrometry Reveals the Population of Oligomers in aB-Crystallin. Proc. Natl. Acad. Sci. U.S.A. 2003, 100, 10611–10616.
Loo J. A. Electrospray Ionization Mass Spectrometry: A Technology for Studying Noncovalent Macromolecular Complexes. Int. J. Mass Spectrom. Ion Processes. 2000, 200, 175–186.
Loo J. A. Studying Noncovalent Protein Complexes by Electrospray Ionization Mass Spectrometry. Mass Spectrom. Rev. 1997, 16, 1–23.
Sobott F.; Robinson C. V. Protein Complexes Gain Momentum. Curr. Opin. Struct. Biol. 2002, 12, 729–734.
Smith R. D.; Bruce J. E.; Wu Q.; Lei Q. P. New Mass Spectrometric Methods for the Study of Noncovalent Associations of Biopolymers. Chem. Soc. Rev. 1997, 26, 191–202.
Prinz H.; Lavie A.; Scheidig A. J.; Spangenberg O.; Konrad M. Binding of Nucleotides to Guanylate Kinase, p21ras, and Nucleoside-Diphosphate Kinase Studied by Nano-Electrospray Mass Spectrometry. J. Biol. Chem. 1999, 274, 35337–35342.
Wu Q.; Gao J.; Joseph-McCarthy D.; Sigal G. B.; Bruce J. E.; Whitesides G. M.; Smith R. D. Carbonic Anhydrase-Inhibitor Binding: From Solution to the Gas Phase. J. Am. Chem. Soc. 1997, 119, 1157–1158.
Light-Wahl K. J.; Schwartz B. L.; Smith R. D. Observation of the Noncovalent Quaternary Associations of Proteins by Electrospray Ionization Mass Spectrometry. J. Am. Chem. Soc. 1994, 116, 5271–5278.
Loo J. A.; Hu P.; McConnell P.; Mueller W. T. A Study of Src SH2 Domain Protein-Phosphopeptide Binding Interactions by Electrospray Ionization Mass Spectrometry. J. Am. Soc. Mass Spectrom. 1997, 8, 234–243.
Zhu M. M.; Rempel D. L.; Gross M. L. Modeling Data from Titration, Amide H/D Exchange, and Mass Spectrometry to Obtain Protein-Ligand Binding Constants. J. Am. Soc. Mass Spectrom. 2004, 15, 388–397.
Zhu M. M.; Rempel D. L.; Du Z.; Gross M. L. Quantification of Protein-Ligand Interactions by Mass Spectrometry, Titration, and H/D Exchange: PLIMSTEX. J. Am. Chem. Soc. 2003, 125, 5252–5253.
Powell K. D.; Ghaemmaghami S.; Wang M. Z.; Liyuan M.; Oas T. G.; Fitzgerald M. C. A General Mass Spectrometry-Based Assay for the Quantitation of Protein-Ligand Binding Interactions in Solution. J. Am. Chem. Soc. 2002, 124, 10256–10257.
Powell K. D.; Fitzgerald M. C. The Accuracy and Precision of a New H/D Exchange- and Mass Spectrometry-Based Technique for Measuring the Thermodynamic Properties of Protein-Peptide Complexes. Biochemistry. 2003, 42, 4962–4970.
Grunwell J. R.; Rath V. L.; Rasmussen J.; Cabrilo Z.; Bertozzi C. R. Characterization and Mutagenesis of Gal/GlcNAc-6-O-Sulfotransferases. Biochemistry. 2002, 41, 15590–15600.
Cormier E. G.; Persuh M.; Thompson D. A. D.; Lin S. W.; Sakmar T. P.; Olson W. C.; Dragic T. Specific Interaction of CCR5 Amino-Terminal Domain Peptides Containing Sulfotyrosines with HIV-1 Envelope Glycoprotein gp120. Proc. Natl. Acad. Sci. U.S.A. 2000, 97, 5762–5767.
Seibert C.; Cadene M.; Sanfiz A.; Chait B. T.; Sakmar T. P. Tyrosine Sulfation of CCR5 N-Terminal Peptide by Tyrosylprotein Sulfotransferases 1 and 2 Follows a Discrete Pattern and Temporal Sequence. Proc. Natl. Acad. Sci. U.S.A. 2002, 99, 11031–11036.
Dong J.; Li C. Q.; Lopez J. A. Tyrosine Sulfation of the Glycoprotein Ib-IX Complex: Identification of Sulfated Residues and Effect on Ligand Binding. Biochemistry. 1994, 33, 13946–13953.
Ehrhardt D. W.; Atkinson E. M.; Faull K. F.; Freedberg D. I.; Sutherlin D. P.; Armstrong R.; Long S. R. In Vitro Sulfotransferase Activity of NodH, a Nodulation Protein of Rhizobium meliloti Required for Host-Specific Nodulation. J. Bacteriol. 1995, 177, 6237–6245.
Pi N.; Armstrong J. I.; Bertozzi C. R.; Leary J. A. Kinetic Analysis of NodST Sulfotransferase Using an Electrospray Ionization Mass Spectrometry Assay. Biochemistry. 2002, 41, 13283–13288.
Pi N.; Yu Y.; Leary J. A. Observation of a Hybrid Random Ping-Pong Mechanism of Catalysis for NodST: A Mass Spectrometry Approach. Protein Sci. 2004, 13, 903–912.
Burkart M. D.; Izumi M.; Chapman E.; Lin C. -H.; Wong C. -H. Regeneration of PAPS for the Enzymatic Synthesis of Sulfated Oligosaccharides. J. Org. Chem. 2000, 65, 5565–5574.
Gao J.; Wu Q.; Carbeck J.; Lei Q. P.; Smith R. D.; Whitesides G. M. Probing the Energetics of Dissociation of Carbonic Anhydrase-Ligand Complexes in the Gas Phase. Biophys. J. 1999, 76, 3253–3260.
Loo J. A.; Loo R. R. O.; Udseth H. R.; Edmonds C. G.; Smith R. D. Solvent-Induced Conformational Changes of Polypeptides Probed by Electrospray-Ionization Mass Spectrometry. Rapid Commun. Mass. Spectrom. 1991, 5, 101–105.
Cheng X.; Harms A. C.; Goudreau P. N.; Terwilliger T. C.; Smith R. D. Direct Measurement of Oligonucleotide Binding Stoichiometry of Gene V Protein by Mass Spectrometry. Proc. Natl. Acad. Sci. U.S.A. 1996, 93, 7022–7027.
Negishi M.; Pedersen L. G.; Petrotchenko E.; Shevtsov S.; Gorokhov A.; Kakuta Y.; Pedersen L. C. Structure and Function of Sulfotransferases. Arch. Biochem. Biophys. 2001, 390, 149–157.
Davidson W.; Hopkins J. L.; Jeanfavre D. D.; Barney K. L.; Kelly T. A.; Grygon C. A. Characterization of the Allosteric Inhibition of a Protein-Protein Interaction by Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2003, 14, 8–13.
Wicki J.; Rose D. R.; Withers S. G. Trapping Covalent Intermediates on β-Glycosidases. Methods Enzymol. 2002, 354, 84–105.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online August 28, 2004
Rights and permissions
About this article
Cite this article
Yu, Y., Kirkup, C.E., Pi, N. et al. Characterization of noncovalent protein-ligand complexes and associated enzyme intermediates of GlcNAc-6-O-sulfotransferase by electrospray ionization FT-ICR mass spectrometry. J Am Soc Mass Spectrom 15, 1400–1407 (2004). https://doi.org/10.1016/j.jasms.2004.06.002
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2004.06.002