Abstract
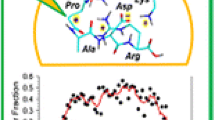
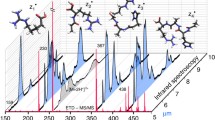
First results are reported on the application of ECD in analysis of 2+ and 3+ ions of stereoisomers of Trp-cage (NLYIQWLKDGGPSSGRPPPS), the smallest and fastest-folding protein, which exhibits a tightly folded tertiary structure in solution. The chiral recognition based on the ratios of the abundances of z 18 and z 19 fragments in ECD of 2+ ions was excellent even for a single amino acid (Tyr) D-substitution (R chiral=8.6). The chiral effect decreased with an increase of temperature at the electrospray ion source, as well as at a higher degree of ionization, 3+ ions (R chiral=1.5). A general approach is suggested for charge localization in n+ ions by analysis of ECD mass spectra of (n+1)+ ions. Application of this approach to 3+ Trp-cage ions revealed the protonation probability order in 2+ ions: Arg16≫Gln5 >≈ N-terminus. The ECD results for native form of the 2+ ions favor the preservation of the solution-phase tertiary structure, and chiral recognition through the interaction between the charges and the neutral bond network. Conversely, ECD of 3+ ions supports the dominance of ionic hydrogen bonding which determines a different gas-phase structure than found in solution. Vibrational activation of 2+ ions indicated greater stability of the native form, but the fragmentation patterns did not provide stereoisomer differentiation, thus underlying the special position of ECD among other MS/MS fragmentation techniques. Further ECD studies should yield more structural information as well as quantitative single-amino acid D/l content measurements in proteins.
Article PDF
Similar content being viewed by others
References
Fenn, J. B.; Mann, M.; Chin Kai Meng, S. F. W.; Whitehouse, C. M. Electrospray Ionization for Mass Spectrometry of Large Biomolecules. Science 1989, 246, 64–71.
Eigen, M.; de Maeyer, L. Relaxation Methods; Interscience: New York, NY, 1963; 895–1054.
Oneil, K. T.; Degrado, W. F. A Thermodynamic Scale for the Helix-Forming Tendencies of the Commonly Occurring Amino-Acids. Science 1990, 250, 646–651.
Pace, C. N.; Shirley, B. A.; McNutt, M.; Gajiwala, K. Forces Contributing to the Conformational Stability of Proteins. Faseb J. 1996, 10, 75–83.
Sturtevant, J. M. The Thermodynamic Effects of Protein Mutations. Curr. Opin. Struct. Biol. 1994, 4, 69–78.
Kaltashov, I. A.; Eyles, S. J. Studies of Biomolecular Conformations and Conformational Dynamics by Mass Spectrometry. Mass Spectrom. Rev. 2002, 21, 37–71.
Lorenz, S. A.; Maziarz, E. P.; Wood, T. D. Electrospray Ionization Fourier Transform Mass Spectrometry of Macromolecules: The First Decade. Appl. Spectroscop. 1999, 53, 18A-36A.
Smith, R. D.; Loo, J. A.; Loo, R. R. O.; Busman, M.; Udseth, H. R. Principles and Practice of Electrospray Ionization-Mass-Spectrometry for Large Polypeptides and Proteins. Mass Spectrom. Rev. 1991, 10, 359–451.
Pramanik, B. N.; Bartner, P. L.; Mirza, U. A.; Liu, Y.; Ganguly, A. K. Electrospray Ionization Mass Spectrometry for the Study of Non-Covalent Complexes: An Emerging Technology. J. Mass Spectrom. 1998, 33, 911–920.
Loo, J. A. Electrospray Ionization Mass Spectrometry: A Technology for Studying Noncovalent Macromolecular Complexes. Int. J. Mass Spectrom. 2000, 200, 175–186.
Miranker, A. D. Protein Complexes and Analysis of Their Assembly by Mass Spectrometry. Curr. Opin. Struct. Biol. 2000, 10, 601–606.
Sobott, F. Protein Complexes Gain Momentum. Curr. Opin. Struct. Biol. 2002, 12, 729–734.
Jurchen, J. C.; Williams, E. R. Origin of Asymmetric Charge Partitioning in the Dissociation of Gas-phase Protein Homodimers. J. Am. Chem. Soc. 2003, 125, 2817–2826.
McLafferty, F. W.; Guan, Z. Q.; Haupts, U.; Wood, T. D.; Kelleher, N. L. Gaseous Conformational Structures of Cytochrome. C. J Am. Chem. Soc. 1998, 120, 4732–4740.
Wood, T. D.; Chorush, R. A.; Wampler, F. M. III.; Little, D. P.; O’Connor, P.B.; McLafferty, F.W. Gas-Phase Folding and Unfolding of CytochromeC Cations. Proc. Natl. Acad. Sci. U.S.A. 1995, 92, 2451–2454.
Lorenz, S. A.; Maziarz, E. P.; Woods, T. D. Using Solution Phase Hydrogen/Deuterium (H/D) Exchange to Determine the Origin of Non-Covalent Complexes Observed by Electrospray Ionization Mass Spectrometry: In Solution or in Vacuo? J. Am. Soc. Mass Spectrom. 2001, 12, 795–804.
Wang, F.; Freitas, M. A.; Marshall, A. G.; Sykes, B. D. Gas-Phase Memory of Solution-phase Protein Conformation: H/D Exchange and Fourier Transform Ion Cyclotron Resonance Mass Spectrometry of the N-terminal Domain of Cardiac Troponin. C. Int. J. Mass Spectrom. 1999, 192, 319–325.
Green, M. K.; Lebrilla, C. B. Ion-Molecule Reactions as Probes of Gas-phase Structures of Peptides and Proteins. Mass Spectrom. Rev. 1997, 16, 53–71.
Loo, R. R. O.; Smith, R. D. Investigation of the Gas-Phase Structure of Electrosprayed Proteins Using Ion-Molecule Reactions. J. Am. Soc. Mass Spectrom. 1994, 5, 207–220.
Iavarone, A. T.; Williams, E. R. Collisionally Activated Dissociation of Supercharged Proteins Formed by Electrospray Ionization. Anal. Chem. 2003, 75, 4525–4533.
Laskin, J.; Futrell, J. H. Collisional Activation of Peptide Ions in FT-ICR Mass Spectrometry. Mass Spectrom. Rev. 2003, 22, 158–181.
Dufresne, C. P.; Wood, T. D.; Hendrickson, C. L. High-Resolution Electrospray Ionization Fourier Transform Mass Spectrometry with Infrared Multiphoton Dissociation of Glucokinase from. Bacillus stearothermophilus. J. Am. Soc. Mass Spectrom. 1998, 9, 1222–1225.
Charlebois, J. P.; Patrie, S. M.; Kelleher, N. L. Electron Capture Dissociation and C-13, N-15 Depletion for Deuterium Localization in Intact Proteins After Solution-Phase Exchange. Anal. Chem. 2003, 75, 3263–3266.
Kjeldsen, F.; Haselmann, K. F.; Sorensen, E. S.; Zubarev, R. A. Distinguishing of Ile/Leu Amino Acid Residues in the PP3 Protein by (Hot) Electron Capture Dissociation in Fourier Transform Ion Cyclotron Resonance Mass Spectrometry. Anal. Chem. 2003, 75, 1267–1274.
Szilagyi, Z.; Drahos, L.; Vekey, K. Conformation of Doubly Protonated Peptides Studied by Charge-Separation Reactions in Mass Spectrometry. J. Mass Spectrom. 1997, 32, 689–696.
Haselmann, K. F.; Jorgensen, T. J. D.; Budnik, B. A.; Jensen, F.; Zubarev, R. A. Electron Capture Dissociation of Weakly Bound Polypeptide Polycationic Complexes. Rapid Commun. Mass Spectrom. 2002, 16, 2260–2265.
Oh, H.; Breuker, K.; Sze, S. K.; Ge, Y.; Carpenter, B. K.; McLafferty, F. W. Secondary and Tertiary Structures of Gaseous Protein Ions Characterized by Electron Capture Dissociation Mass Spectrometry and Photofragment Spectroscopy. Proc. Natl. Acad. Sci. U.S.A. 2002, 99, 15863–15868.
Breuker, K.; Oh, H. B.; Horn, D. M.; Cerda, B. A.; McLafferty, F. W. Detailed Unfolding and Folding of Gaseous Ubiquitin Ions Characterized by Electron Capture Dissociation. J. Am. Chem. Soc. 2002, 124, 6407–6420.
Horn, D. M.; Breuker, K.; Frank, A. J.; McLafferty, F. W. Kinetic Intermediates in the Folding of Gaseous Protein Ions Characterized by Electron Capture Dissociation Mass Spectrometry. J. Am. Chem. Soc. 2001, 123, 9792–9799.
Budnik, B. A.; Nielsen, M. L.; Olsen, J. V.; Haselmann, K. F.; Horth, P.; Haehnel, W.; Zubarev, R. A. Can Relative Cleavage Frequencies in Peptides Provide Additional Sequence Information? Int. J. Mass Spectrom. 2002, 219, 283–294.
Cooper, H. J.; Hudgins, R. R.; Håkansson, K.; Marshall, A. G. Characterization of Amino Acid Side Chain Losses in Electron Capture Dissociation. J. Am. Soc. Mass Spectrom. 2002; 13, 241–249.
Haselmann, K. F.; Budnik, B. A.; Kjeldsen, F.; Polfer, N. C.; Zubarev, R. A. Can the (·-X) Region in Electron Capture Dissociation Provide Reliable Information on Amino Acid Composition of Polypeptides? Eur. J. Mass Spectrom. 2002, 8, 461–469.
Cooper, H. J.; Håkansson, K.; Marshall, A. G.; Hudgins, R. R.; Haselmann, K. F.; Kjeldsen, F.; Budnik, B. A.; Polfer, N. C.; Zubarev, R. A. The Diagnostic Value of Amino Acid Side-Chain Losses in Electron Capture Dissociation of Polyepeptides. Comment on: “Can the (Mo-X) Region in Electron Capture Dissociation Provide Reliable Information on Amino Acid Composition of Polypeptides?”. Eur. J. Mass Spectrom. 2003, 9, 221–222.
Meot-Ner, M. The Ionic Hydrogen Bond. 2. Intramolecular and Partial Bonds. Protonation of Polyethers, Crown Ethers, and Diketones. J. Am. Chem. Soc. 1983, 105, 4906–4911.
Neidigh, J. W.; Fesinmeyer, R. M.; Andersen, N.H. Designing a 20-Residue Protein. Nat. Struct. Biol. 2002, 9, 425–430.
Snow, C. D.; Zagrovic, B.; Pande, V.S. The Trp Cage: Folding Kinetics and Unfolded State Topology via Molecular Dynamics Simulations. J. Am. Chem. Soc. 2002, 124, 14548–14549.
Qiu, L. L.; Pabit, S. A.; Roitberg, A. E.; Hagen, S. J. Smaller and Faster: The 20-Residue Trp-cage Protein Folds in 4 mu s. J. Am. Chem. Soc. 2002, 124, 12952–12953.
Chowdhury, S.; Lee, M. C.; Xiong, G. M.; Duan, Y. Ab Initio Folding Simulation of the Trp-cage Mini-Protein Approaches NMR Resolution. J. Mol. Biol. 2003, 327, 711–717.
Carnevali, P.; To’th, G.; Toubassi, G.; Meshkat, S. N. Fast Protein Structure Prediction Using Monte Carlo Simulations with Modal Moves. J. Am. Chem. Soc. 2003, 125, 14244–14245.
Schug, A. H. T.; Wenzel, W. Reproducible Protein Folding with the Stochastic Tunneling Method. Phys. Rev. Lett. 2003, 91, Art. No. 158102.
Barua, B.; Andersen, N.H. Determinants of Miniprotein Stability: Can Anything Replace a Buried H-Bonded Trp Sidechain? Lett. Pept. Sci. 2001, 8, 221–226.
Krause, E.; Beyermann, M.; Dathe, M.; Rothemund, S.; Bienert, M. Location of an Amphipathic α-Helix in Peptides Using Reversed-Phase HPLC Retention Behavior of D-Amino-Acid Analogs. Anal. Chem. 1995, 67, 252–258.
Mitchell, J.B. D-Amino Acid Residues in Peptides and Proteins. Proteins: Structure, Function, and Genomics. 2003, 50, 563–571.
Haselmann, K. F.; Budnik, B. A.; Olsen, J. V.; Nielsen, M. L.; Reis, C. A.; Clausen, H.; Johnsen, A. H.; Zubarev, R. A. Advantages of External Accumulation for Electron Capture Dissociation in Fourier Transform Mass Spectrometry. Anal. Chem. 2001, 73, 2998–3005.
Tsybin, Y. O.; Hakansson, P.; Budnik, B. A.; Haselmann, K. F.; Kjeldsen, F.; Gorshkov, M.; Zubarev, R. A. Improved Low-Energy Electron Injection Systems for High Rate Electron Capture Dissociation in Fourier Transform Ion Cyclotron Resonance Mass Spectrometry. Rapid Commun. Mass Spectrom. 2001, 15, 1849–1854.
Boeckmann, B.; Bairoch, A.; Apweiler, R.; Blatter, M. C.; Estreicher, A.; Gasteiger, E.; Martin, M. J.; Michoud, K.; O’Donovan, C.; Phan, I.; Pilbout, S.; Schneider, M. The SWISS-PROT Protein Knowledgebase and Its Supplement TrEMBL in 2003. Nucl. Acids Res. 2003, 31, 365–370.
Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual Molecular Dynamics. J. Mol. Graphics 1996, 14, 33.
Williams, E.R. Proton Transfer Reactivity of Large Multiply Charged Ions. J. Mass Spectrom. 1996, 31, 831–842.
Zubarev, R. A.; Haselmann, K. F.; Budnik, B.; Kjeldsen, F.; Jensen, F. Towards an Understanding of the Mechanism of Electron-capture Dissociation: A Historical Perspective and Modern Ideas. Eur. J. Mass Spectrom. 2002, 8, 337–349.
Tsaprailis, G.; Nair, H.; Somogyi, A.; Wysocki, V. H.; Zhong, W.Q.; Futrell, J. H.; Summerfield, S. G.; Gaskell, S. J. Influence of Secondary Structure on the Fragmentation of Protonated Peptides. J. Am. Chem. Soc. 1999, 121, 5142–5154.
Kjeldsen, F.; Haselmann, K. F.; Budnik, B. A.; Jensen, F.; Zubarev, R. A. Dissociative Capture of Hot (3–13 eV) Electrons by Polypeptide Polycations: An Efficient Process Accompanied by Secondary Fragmentation. Chem. Phys. Lett. 2002, 356, 201–206.
Kjelden, F.; Zubarev, R. A. Secondary Losses via ?-Lactam Formation in Hot Electron Capture Dissociation: A Missing Link to Complete de Novo Sequencing of Proteins? J. Am. Chem. Soc. 2003, 125, 6628–6629.
Polfer, N. C. Structural Elucidation of Peptides and Proteins by Fourier Transform Ion Cyclotron Resonance Mass Spectrometry; Thesis, University of Edinburgh: Edinburgh, UK, 2003, 95–226.
Wu, L. M.; Cooks, R. G. Chiral Analysis Using the Kinetic Method with Optimized Fixed Ligands: Applications to Some Antibiotics. Anal. Chem. 2003, 75, 678–684.
Wu, C.; Siems, W. F.; Klasmeier, J.; Hill, H. H. Separation of Isomeric Peptides Using Electrospray Ionization/High-Resolution Ion Mobility Spectrometry. Anal. Chem. 2000, 72, 391–395.
Tao, W. A.; Zhang, D. X.; Nikolaev, E. N.; Cooks, R. G. Copper(II)-Assisted Enantiomeric Analysis of D,L-Amino acids Using the Kinetic Method: Chiral Recognition and Quantification in the Gas Phase. J. Am. Chem. Soc. 2000, 122, 10598–10609.
Salem, L. C. X.; Segal, G.; Hiberty, C.; Minot, C.; Leforestier, C.; Sautet, P. Chirality Forces. J. Am. Soc. Mass Spectrom. 1987, 109, 2887–2894.
Handman, M.; Curcuruto, O. Probing Conformational Changes in Some Proteins by Positive-Ion Electrospray Mass Spectrometry. Rapid Commun. Mass Spectrom. 1994, 8, 144–147.
Jarrold, M. Unfolding, Refolding, and Hydration of Proteins in the Gas Phase. Acc. Chem. Res. 1999, 32, 360–367.
Schnier, P. D.; Gross, D. S.; Williams, E. R.; Electrostatic Forces and Dielectric Polarizability of Multiply Protonated Gas-Phase Cytochrome-C Ions Probed by Ion/Molecule Chemistry. J. Am. Chem. Soc. 1995, 117, 6747–6757.
Wu, Z.; Fenselau, C. Proton Affinity of Arginine Measured by the Kinetic Approach. Rapid Commun. Mass Spectrom. 1992, 6, 403–405.
McLafferty, F. W. Tandem Mass Spectrometry of Large Molecules Mass Spectrometry in the Analysis of Large Molecules, C. J. McNeal, ed.; J. Wiley: New York, 1986, 107–120.
Zubarev, R. A.; Kelleher, N. L.; McLafferty, F. W. Electron Capture Dissociation of Multiply Charged Protein Cations. A Nonergodic Process. J. Am. Chem. Soc. 1998, 120, 3265–3266.
Adams, C. M.; Orekhov, V.; Zubarev, R. A., unpublished NMR data
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online June 10, 2004
Supplementary material available: Rotating 3-D model of the Trp-cage molecule (PowerPoint).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Adams, C.M., Kjeldsen, F., Zubarev, R.A. et al. Electron capture dissociation distinguishes a single D-amino acid in a protein and probes the tertiary structure. J Am Soc Mass Spectrom 15, 1087–1098 (2004). https://doi.org/10.1016/j.jasms.2004.04.026
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2004.04.026