Abstract
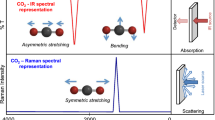
Fourier Transform Ion Cyclotron Resonance Mass Spectrometry (ESI-FTICR-MS) coupled with infrared multiphoton dissociation (IRMPD) is potentially a powerful method for rapid phosphopeptide mapping of complex proteolytic digests. The dissociation of deprotonated phosphopeptides by IRMPD is energetically favorable over unmodified deprotonated peptides because of a lower energy of activation and a higher internal energy under identical irradiation conditions. The energies of activation for dissociation are determined for model peptides phosphorylated on an aliphatic side chain (serine) and an aromatic side chain (tyrosine). The determination of phosphorylation location provides important biochemical information identifying the kinase involved in specific phosphorylation mechanisms. The data presented in this manuscript also support the theory that for phosphopeptides, the phosphate moiety’s P-O stretch is in direct resonance with the infrared laser (10.6 µm), thus increasing the relative absorptivity of the modified species. A greater extinction coefficient affords more extensive photon absorption and subsequently a greater internal energy at the rapid exchange limit.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Krebs, E. G. The Growth of Research on Protein Phosphorylation Trends Biochem. Sci. 1994, 19, 439.
Hunter, T. Protein Kinases and Phosphatases: The Yin and Yang of Protein Phosphorylation and Signaling Cell 1995, 80, 225–236.
Posada, J.; Cooper, J. A. Molecular Signal Integration. Interplay between Serine, Threonine, and Tyrosine Phosphorylation Mol. Biol. Cell 1992, 3, 583–592.
Kennelly, P. J.; Krebs, E. G. Consensus Sequences as Substrate Specificity Determinants for Protein Kinases and Protein Phosphatases J. Biol. Chem. 1991, 266, 15555–15558.
Songyang, Z.; Cantley, L. C. Recognition and Specificity in Protein Tyrosine Kinase-Mediated Signalling Trends Biochem. Sci. 1995, 20, 470–475.
Davis, M. T.; Lee, T. D. Rapid Protein Identification Using a Microscale Electrospray LC/MS System on an Ion Trap Mass Spectrometer J. Am. Soc. Mass Spectrom. 1998, 9, 194–201.
Zhou, W.; Merrick, B. A.; Khaledi, M. G.; Tomer, K. B. Detection and Sequencing of Phosphopeptides Affinity Bound to Immobilized Metal Ion Beads by Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry J. Am. Soc. Mass Spectrom 1999, 11, 273–282.
Posewitz, M. C.; Tempst, P. Immobilized Gallium(III) Affinity Chromatography of Phosphopeptides Anal. Chem. 1999, 71, 2883–2892.
McLachlin, D. T.; Chait, B. T. Analysis of Phosphorylated Proteins and Peptides by Mass Spectrometry Curr. Opin. Chem. Biol. 2001, 5, 591–602.
Oda, Y.; Nagasu, T.; Chait, B. T. Enrichment Analysis of Phosphorylated Proteins as a Tool for Probing the Phosphoproteome Nat. Biotechnol. 2001, 19, 379–382.
Zhou, H.; Watts, J. D.; Aebersold, R. A Systematic Approach to the Analysis of Protein Phosphorylation Nat. Biotechnol. 2001, 19, 375–378.
Goshe, M. B.; Conrads, T. P.; Panisko, E. A.; Angell, N. H.; Veenstra, T. D.; Smith, R. D. Phosphoprotein Isotope-Coded Affinity Tag Approach for Isolating and Quantitating Phosphopeptides in Proteome-Wide Analyses Anal. Chem. 2001, 73, 2578–2586.
Kalo, M. S.; Pasquale, E. B. Multiple in Vivo Tyrosine Phosphorylation Sites in Ephb Receptors Biochemistry 1999, 38, 14396–14408.
Watty, A.; Neubauer, G.; Dreger, M.; Zimmer, M.; Wilm, M.; Burden, S. J. The in Vitro and in Vivo Phosphotyrosine Map of Activated Musk Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 4585–4590.
Okamura, H.; Aramburu, J.; Garcia-Rodriguez, C.; Viola, J. P.; Raghavan, A.; Tahiliani, M.; Zhang, X.; Qin, J.; Hogan, P. G.; Rao, A. Concerted Dephosphorylation of the Transcription Factor Nfat1 Induces a Conformational Switch that Regulates Transcriptional Activity Mol. Cell 2000, 6, 539–550.
Knotts, T. A.; Orkiszewski, R. S.; Cook, R. G.; Edwards, D. P.; Weigel, N. L. Identification of a Phosphorylation Site in the Hinge Region of the Human Progesterone Receptor and Additional Amino-Terminal Phosphorylation Sites J. Biol. Chem. 2001, 276, 8475–8483.
Larsen, M. R.; Sorensen, G. L.; Fey, S. J.; Larsen, P. M.; Roepstorff, P. Phospho-Proteomics: Evaluation of the Use of Enzymatic De-Phosphorylation and Differential Mass Spectrometric Peptide Mass Mapping for Site Specific Phosphorylation Assignment in Proteins Separated by Gel Electrophoresis Proteomics 2001, 1, 223–238.
Liao, P. C.; Leykam, J.; Andrews, P. C.; Gage, D. A.; Allison, J. An Approach to Locate Phosphorylation Sites in a Phosphoprotein: Mass Mapping by Combining Specific Enzymatic Degradation with Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry Anal. Biochem. 1994, 219, 9–20.
Jensen, O. N.; Larsen, M. R.; Roepstorff, P. Mass Spectrometric Identification and Microcharacterization of Proteins from Electrophoretic Gels: Strategies and Applications. Proteins 1998(Suppl 2), 74–89.
Amankwa, L. N.; Harder, K.; Jirik, F.; Aebersold, R. High-Sensitivity Determination of Tyrosine-Phosphorylated Peptides by on-Line Enzyme Reactor and Electrospray Ionization Mass Spectrometry Protein Sci 1995, 4, 113–125.
Wind, M.; Edler, M.; Jakubowski, N.; Linscheid, M.; Wesch, H.; Lehmann, W. D. Analysis of Protein Phosphorylation by Capillary Liquid Chromatography Coupled to Element Mass Spectrometry with 31p Detection and to Electrospray Mass Spectrometry Anal. Chem. 2001, 73, 29–35.
Covey, T.; Shushan, B.; Bonner, R.; Schröder, W.; Hucho, F. LC/MS and LC/MS/MS Screening for the Sites of Post-Translational Modification in Proteins. In Methods in Protein Sequence Analysis (Proceedings of the 8th International Conference), Jörnvall, H.; Höög, J.-O.; Gustavsson, A.-M., Eds.; Birkhäuser Verlag: Basel, 1991; pp. 249–256.
Tholey, A.; Reed, J.; Lehmann, W. D. Electrospray Tandem Mass Spectrometric Studies of Phosphopeptides and Phosphopeptide Analogues J. Mass Spectrom. 1999, 34, 117–123.
Annan, R. S.; Huddleston, M. J.; Verma, R.; Deshaies, R. J.; Carr, S. A. A Multidimensional Electrospray MS-Based Approach to Phosphopeptide Mapping Anal. Chem. 2001, 73, 393–404.
DeGnore, J. P.; Qin, J. Fragmentation of Phosphopeptides in an Ion Trap Mass Spectrometer J. Am. Soc. Mass Spectrom. 1998, 9, 1175–1188.
Wilm, M.; Neubauer, G.; Mann, M. Parent Ion Scans of Unseparated Peptide Mixtures Anal. Chem. 1996, 68, 527–533.
Carr, S. A.; Huddleston, M. J.; Annan, R. S. Selective Detection and Sequencing of Phosphopeptides at the Femtomole Level by Mass Spectrometry Anal. Biochem. 1996, 239, 180–192.
Steen, H.; Kuster, B.; Fernandez, M.; Pandey, A.; Mann, M. Detection of Tyrosine Phosphorylated Peptides by Precursor Ion Scanning Quadrupole TOF Mass Spectrometry in Positive Ion Mode Anal. Chem. 2001, 73, 1440–1448.
Borchers, C.; Parker, C. E.; Deterding, L. J.; Tomer, K. B. Preliminary Comparison of Precursor Scans and Liquid Chromatography-Tandem Mass Spectrometry on a Hybrid Quadrupole Time-of-Flight Mass Spectrometer J. Chromatogr. A 1999, 854, 119–130.
Verma, R.; Annan, R. S.; Huddleston, M. J.; Carr, S. A.; Reynard, G.; Deshaies, R. J. Phosphorylation of Sic1p by G1 Cdk Required for Its Degradation and Entry into S Phase Science 1997, 278, 455–460.
Beuvink, I.; Hess, D.; Flotow, H.; Hofsteenge, J.; Groner, B.; Hynes, N. E. Stat5a Serine Phosphorylation. Serine 779 is Constitutively Phosphorylated in the Mammary Gland, and Serine 725 Phosphorylation Influences Prolactin-Stimulated in Vitro DNA Binding Activity J. Biol. Chem. 2000, 275, 10247–10255.
Schlosser, A.; Pipkorn, R.; Bossemeyer, D.; Lehmann, W. D. Analysis of Protein Phosphorylation by a Combination of Elastase Digestion and Neutral Loss Tandem Mass Spectrometry Anal. Chem. 2001, 73, 170–176.
Huddleston, M. J.; Annan, R. S.; Bean, M. F.; Carr, S. A. Selective Detection of Phosphopeptides in Complex Mixtures by Electrospray Liquid Chromatography Mass Spectrometry J. Am. Soc. Mass Spectrom. 1993, 4, 710–717.
Ding, J.; Burkhart, W.; Kassel, D. B. Identification of Phosphorylated Peptides from Complex Mixtures Using Negative-Ion Orifice-Potential Stepping and Capillary Liquid Chromatography/Electrospray Ionization Mass Spectrometry Rapid Commun. Mass Spectrom. 1994, 8, 94–98.
Annan, R. S.; Carr, S. A. Phosphopeptide Analysis by Matrix-Assisted Laser Desorption Time-of-Flight Mass Spectrometry Anal. Chem. 1996, 68, 3413–3421.
Schnölzer, M.; Lehmann, W. D. Identification of Modified Peptides by Metastable Fragmentation in MALDI Mass Spectrometry Int. J. Mass Spectrom. Ion Processes 1997, 169/170, 263–271.
Metzger, S.; Hoffmann, R. Studies on the Dephosphorylation of Phosphotyrosine-Containing Peptides During Post-Source Decay in Matrix-Assisted Laser Desorption/Ionization J. Mass Spectrom. 2000, 35, 1165–1177.
Shi, S. D.; Hemling, M. E.; Carr, S. A.; Horn, D. M.; Lindh, I.; McLafferty, F. W. Phosphopeptide/Phosphoprotein Mapping by Electron Capture Dissociation Mass Spectrometry Anal. Chem. 2001, 73, 19–22.
Fenn, J. B.; Mann, M.; Meng, C. K.; Wong, S. F.; Whitehouse, C. M. Electrospray Ionization for Mass Spectrometry of Large Biomolecules Science 1989, 246, 64–71.
Comisarow, M. B.; Marshall, A. G. Fourier Transform Ion Cyclotron Resonance Mass Spectrometry Chem. Phys. Lett. 1974, 25, 282–283.
Little, D. P.; Speir, J. P.; Senko, M. W.; O’Connor, P. B.; McLafferty, F. W. Infrared Multiphoton Dissociation of Large Multiply Charged Ions for Biomolecule Sequencing Anal. Chem. 1994, 66, 2809–2815.
Flora, J. W.; Muddiman, D. C. Selective, Sensitive, and Rapid Phosphopeptide Identification in Enzymatic Digests Using ESI-FT-ICR-MS with Infrared Multiphoton Dissociation Anal. Chem. 2001, 73, 3305–3311.
Flora, J. W.; Muddiman, D. C. Gas-Phase Ion Unimolecular Dissociation for Rapid Phosphopeptide Mapping by IRMPD in a Penning Ion Trap: An Energetically Favored Process J. Am. Chem. Soc. 2002, 124, 6546–6547.
Jockusch, R. A.; Paech, K.; Williams, E. R. Energetics from Slow Infrared Multiphoton Dissociation of Biomolecules J. Phys. Chem. A 2000, 104, 3188–3196.
Freitas, M. A.; Hendrickson, C. L.; Marshall, A. G. Determination of Relative Ordering of Activation Energies for Gas-Phase Ion Unimolecular Dissociation by Infrared Radiation for Gaseous Multiphoton Energy Transfer J. Am. Chem. Soc. 2000, 122, 7768–7775.
Hannis, J. C.; Muddiman, D. C. Nanoelectrospray Mass Spectrometry Using Non-Metalized, Tapered (50–10 Mm) Fused-Silica Capillaries Rapid Commun. Mass Spectrom. 1998, 12, 443–448.
Senko, M. W.; Hendrickson, C. L.; Emmett, M. R.; Shi, S. D.-H.; Marshall, A. G. External Accumulation of Ions for Enhanced Electrospray Ionization Fourier Transform Ion Cyclotron Resonance Mass Spectrometry J. Am. Soc. Mass Spectrom. 1997, 8, 970–976.
Dunbar, R. C. Kinetics of Thermal Unimolecular Dissociation by Ambient Infrared Radiation J. Phys. Chem. 1994, 98, 8705–8712.
Dunbar, R. C.; McMahon, T. B. Activation of Unimolecular Reactions by Ambient Blackbody Radiation Science 1998, 279, 194.
Hannis, J. C.; Muddiman, D. C. Tailoring the Gas-Phase Dissociation and Determining the Relative Energy of Activation for Dissociation of 7-Deaza Purine Modified Oligonucleotides Containing a Repeating Motif Int. J. Mass. Spectrom. 2002, 219, 139–150.
Dunbar, R. C. Kinetics of Low-Intensity Infrared Laser Photodissociation. The Thermal Model and Application of the Tolman Theorem J. Chem. Phys. 1991, 95, 2537.
Affolter, M.; Watts, J. D.; Krebs, D. L.; Aebersold, R. Evaluation of Two-Dimensional Phosphopeptide Maps by Electrospray Ionization Mass Spectrometry of Recovered Peptides Anal. Biochem. 1994, 223, 74–81.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online December 2, 2003
Rights and permissions
About this article
Cite this article
Flora, J.W., Muddiman, D.C. Determination of the relative energies of activation for the dissociation of aromatic versus aliphatic phosphopeptides by ESI-FTICR-MS and IRMPD. J Am Soc Mass Spectrom 15, 121–127 (2004). https://doi.org/10.1016/j.jasms.2003.10.004
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/j.jasms.2003.10.004