Abstract
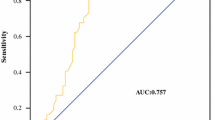
The prediction of survival of patients with pancreatic cancer is usually based on tumor staging and grading and on the level of tumor markers. However, accurate tumor staging can be obtained only after resection, and still there is a great difference in survival rates among patients with the same clinicopathologic parameters. Recently the uptake of 18-fluorodeoxyglucose (FDG) by positron emission tomography (PET) has been found to be correlated with survival in patients with pancreatic cancer. This study evaluated the role of 18FDG PET as a prognostic factor for patients with pancreatic cancer. From June 1996 to July 2002, a total of 118 patients underwent PET for pancreatic cancer. The standardized uptake value (SUV) of 18FDG was calculated in 60 of them, and these patients were divided into high (>4) and low (≦4) SUV groups. They were also evaluated according to the tumor node metastasis (TNM) classification system of the International Union Against Cancer, and by tumor grade, medical or surgical treatment, diabetes, age, sex, and CA19-9 serum levels. Twenty-nine cancers showed high and 31 showed low SUVs. Survival was significantly influenced by tumor stage (P = 0.0001), tumor grade (P = 0.01), and SUV (P = 0.005). Multivariate analysis showed that only stage (P = 0.001) and SUV (P = 0.0002)were independent predictors of survival. When patients who were analyzed for SUV were stratified according to the other variables, FDG uptake was related to survival also after stratification for the following: stage III to IVa (P = 0.002), stage IVb (P = 0.01), tumor resection (P = 0.006), moderately differentiated tumors (P = 0.01), age less than 65 years (P = 0.006), CA 19–9 levels greater than 300 kU/L (P = 0.002), and absence of diabetes (P = 0.0001). The SUV calculated with 18FDG PET is an important prognostic factor for patients with pancreatic cancer and may be useful in selecting patients for therapeutic management.
Similar content being viewed by others
References
Bray F, Sankila R, Ferlay J, Parkin DM. Estimates of cancer incidence and mortality in Europe in 1995. Eur J Cancer 2002;38:99–166.
Lim JE, Chien MW, Earle CC. Prognostic factors following curative resection for pancreatic adenocarcinoma. A population-based, linked database analysis of 396 patients. Ann Surg 2003;237:74–85.
Niedergethmann M, Hildebrand R, Wostbrock B, Hartel M, Sturm JW, Richter A, Post S. High expression of vascular endothelial growth factor predicts early recurrence and poor prognosis after curative resection for ductal adenocarcinoma of the pancreas. Pancreas 2002;25:122–129.
Wittekind C, Compton C, Greene FL, Sobin LH. TNM residual tumor classification revisited. Cancer 2002;94:2511–2519.
Sperti C, Pasquali C, Catalini S, Cappellazzo F, Bonadimani B, Behboo R, Pedrazzoli S. CA 19–9 as a prognostic index after resection for pancreatic cancer. J Surg Oncol 1993; 52:137–141.
Vogel I, Kalthoff H, Henne-Bruns D, Kremer B. Detection and prognostic impact of disseminated tumor cells in pancreatic carcinoma. Pancreatology 2002;2:79–88.
Nakata B, Nishimura S, Ishikawa T, Ohira M, Nishino H, Kawabe J, Ochi H, Hirakawa K Prognostic predictive value of 18-Fluorodeoxyglucose positron emission tomography for patients with pancreatic cancer. Int J Oncology 2001;19:53–58.
Delbeke D, Martin WH. Positron emission tomography imaging in oncology. Radiol Clin N Am 2001;39:883–917.
Hustinx R, Benard F, Alavi A Whole-body imaging in the management of patients with cancer. Sem Nucl Med 2002; 32:35–46.
Rose M, Delbeke D, Beauchamp D, Chapman WC, Sandler MP, Sharp KW, Richards WO, Write JK, Frexes ME, Pinson CW, Leach SD. 18-Fluorodeoxyglucose positron emission tomography in the management of patients with suspected pancreatic cancer. Ann Surg 1998;229:729–738.
Van Heertum RL, Fawrwaz RA The role of nuclear medicine in the evaluation of pancreatic disease. Surg Clin N Am 2001;82:345–348.
Di Chiro G, Delapaz RL, Brooks RA, Sokoloff L, Kornblith PL, Smith BH, Patronas NJ, Kufta CV, Kessler RM, Johnston GS, Manning RG, Wolf AP. Glucose utilization of Cerebral Gliomas measured by (18F) fluorodeoxyglucose and positron emission tomography. Neurology 1982;32:1323–1329.
Patronas NJ, Di Chiro G, Kufta C, Bairamian D, Kornblith PL, Simon R, Larson SM. Prediction of survival in glioma patients by means of positron emission tomography. J Neurosurg 1985;62:816–822.
Nakata B, Chung YS, Nishimura S, Nishihara T, Sakurai Y, Sawada T, Okamura T, Kawabe J, Ochi H, Sowa M. 18-Fluorodeoxyglucose positron emission tomography and the prognosis of patients with pancreatic adenocarcinoma. Cancer 1997;79:695–699.
Maisey NR, Webb A, Flux GD, Padhani A, Cunningham DC, Ott RJ, Norman A. FDG-PET in the prediction of survival of patients with cancer of the pancreas: a pilot study. Br J Cancer 2000;83:287–293.
Zimny M, Fass J, Bares R, Cremerius U, Sabri O, Buechin P, Schumpelick V, Buell U. Fluorodeoxyglucose positron emission tomography and the prognosis of pancreatic carcinoma. ScandJ Gastroenterology 2000;35:883–888.
Sobin LH, Wittekind C. UICC: TNM Classification of malignant tumors. Eight edition. New York: John Wiley and sons, Inc, 1997.
Higashi T, Tamaki N, Honda T, Torizuka T, Kimura T, Inokuma T, Ohshio G, Hosotani R, Imamura M, Konishi J. Expression of glucose transporters in human pancreatic tumours compared with increased FDG accumulation in PET. J Nucl Med 1997;38:1337–1344.
Reske SN, Grillenberger KG, Glatting G, Port M, Hildebrandt M, Gansauge F, Beger HG. Overexpression of glucose transporter 1 and increased FDG uptake in pancreatic carcinoma. J Nucl Med 1997;38:1344–1348.
Scheck N, Hall BL, Finn OJ. Increased glyceraldehyde-3-phosphate dehydrogenase gene expression in human pancreatic adenocarcinoma. Cancer Res 1988;48:6354–6359.
Minn H, Joensuu H, Ahonen A, Klemi P. Fluorodeoxyglucose imaging: a method to assess the proliferative activity of human cancer in vivo. Comparison with DNA flow cytometry in head and neck tumours. Cancer 1988;61:1776–1781.
Higashi K, Clavo AC, Wahl RL. Does FDG uptake measure proliferative activity of human cancer cells? In vivo comparison with DNA flow cytometry and tritiated thymidine uptake. J Nucl Med 1993;34:414–419.
Sperti C, Pasquali C, Piccoli A, Pedrazzoli S. Survival after resection for ductal adenocarcinoma of the pancreas. Br J Surg 1996;83:625–631.
Benassai G, Mastrorilli M, Quarto G, Coppiello A, Giani U, Forestieri P, Mazzeo F. Factors influencing survival after resection for ductal adenocarcinoma of the head of the pancreas. J Surg Oncol 2000;73:212–218.
Townsend DW, Beyer T. A combined PET/CT scanner: the path to true image fusion. Brit J Radiol 2002;75:S24-S30.
Hany TF, Steinert HC, Goerres GW, Buck A, von Schulthess GK. PET diagnostic accuracy: improvement with inline PET-CT system: initial results. Radiology 2002;225:575–581.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sperti, C., Pasquali, C., Chierichetti, F. et al. 18-fluorodeoxyglucose positron emission tomography in predicting survival of patients with pancreatic carcinoma. J Gastrointest Surg 7, 953–960 (2003). https://doi.org/10.1016/j.gassur.2003.09.002
Issue Date:
DOI: https://doi.org/10.1016/j.gassur.2003.09.002