Abstract
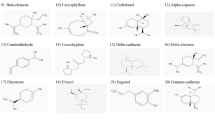
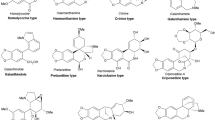
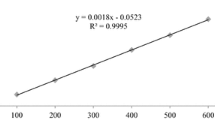
Acetylcholinesterase is an important target for control of neurodegenerative diseases causing cholinergic signaling deficit. Traditionally, galanthamine has been used as an Amaryllidaceae-derived acetylcholinesterase inhibitor, although new Amaryllidaceae plants could serve as source for better acetylcholinesterase inhibitors. Therefore, the objective of this study was to characterize the alkaloid composition from bulbs of Rhodolirium andicola (Poepp.) Traub, a native Chilean Amaryllidaceae specie, and assess their inhibitory activity on acetylcholinesterase by in vitro and in silico methodologies. Alkaloidal extracts from R. andicola exhibited an inhibitory activity with IC50 values between 11.25 ±0.04 and 57.78 ± 1.92 μg/ml that included isolated alkaloid, galanthamine (2.3 ± 0.18 μg/ml), Additionally, 12 alkaloids were detected using gas chromatography-mass spectrometry and identified by comparing their mass fragmentation patterns with literature and database NIST vs.2.0. To better understand the bioactivity of isolated compounds and alkaloidal extracts against acetylcholinesterase, a molecular docking approach was performed. Results suggested that alkaloids such as lycoramine, norpluvine diacetate and 6α-deoxy-tazettine expand the list of potential acetylcholinesterase inhibitors to not only galanthamine. The role of R. andicola as a source for acetylcholinesterase inhibitors is further discussed in this study.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Atanasova, M., Yordanov, N., Dimitrov, I., Berkov, S., Doytchinova, I., 2005. Molecular docking study on galantamine derivatives as cholinesterase inhibitors. Mol. Inform. 34, 394–403.
Barnard, E.A., 1974. Neuromusculartransmission- enzymatic destruction of acetylcholine. In: Hubbard, J.I. (Ed.), The Peripheral Nervous System. Plenum Press, New York, pp. 201–224.
Bartolucci, C., Perola, E., Pilger, C., Fels, G., Lamba, D., 2001. Three-dimensional structure of a complex of galanthamine (Nivalin) with acetylcholinesterase from Torpedo californica: implications for the design of new anti-Alzheimer drugs. Proteins 42, 182–191.
Berkov, S., Codina, C., Viladomat, F., Bastida, J., 2008. N-alkylated galanthamine derivatives: potent acetylcholinesterase inhibitors from Leucojum aestivum. Bioorg. Med. Chem. Lett. 18, 2263–2266.
Berkov, S., Evstatieva, L., Popov, S., 2004. Alkaloids in Bulgarian Pancratium mariti-mum L. Z. Naturforsch. 59c, 65–69.
Berkov, S., Viladomat, F., Codina, C., Suárez, S., Ravelo, A., Bastida, J., 2012. GC-MS of amaryllidaceous galanthamine-type alkaloids. J. Mass. Spectrom. 47, 1065–1073.
Bulger, P.G., Bagal, S.K., Marquez, R., 2008. Recent advances in biomimetic natural product synthesis. Nat. Prod. Rep. 25, 254–297.
Cortes, N., Alvarez, R., Osorio, E.H., Alzate, F., Berkov, S., Osorio, E., 2015. Alkaloids metabolite profiles by GC-MS and acetylcholinesterase inhibitory activities with bending-mode predictions of five Amaryllidaceae plants. J. Pharm. Biomed. Anal. 102, 222–228.
de Andrade, J.P., Guo, Y., Font-Bardia, M., Calvet, T., Dutilh, J., Villadomat, F., Codina, C., Nair, J.J., Zuanazzi, J.A.S., Bastidas, J., 2014. Crinine-type alkaloids from Hippeastrum alicum and H. calyptratum. Phytochemistry 103, 188–195.
de Andrade, J.P., Giordani, R.P., Torras-Claviera, L., Pigni, N.B., Berkov, S., Font-Bardia, M., Calvet, T., Konrath, E., Bueno, K., Sachett, L.G., Dutil, J.H., de Sousa Borgues, W., Villadomat, F., Henriques, A.T., Nair, J.J., Zuanazzi, J.A.S., Bastidas, J., 2016. The Brazilian Amaryllidaceae as source of acetylcholinesterase inhibitory alkaloids. Phytochem. Rev. 15, 147–160.
Edgar, R.C., 2004. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797.
Elisha, I.L., Elgorashi, E.E., Hussein, A.A., Duncan, G., Eloff, J.N., 2013. Acetylcholinesterase inhibitory effects of the bulb of Ammocharis coranica (Amaryllidaceae) and its active constituent lycorine. S. Afr. J. Bot. 85, 44–47.
Ellman, G.L., Courtney, D.K., Valentino Jr., A., Featherstone, R.M., 1961. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 7, 88–95.
Gotti, R., Fiori, J., Bartolini, M., Cavrini, V., 2006. Analysis of Amaryllidaceae alkaloids from Narcissus by GC-MS and capillary electrophoresis. J. Pharm. Biomed. Anal. 42, 17–24.
Greenblatt, H.M., Kryger, G., Lewis, T., Silman, I., Sussman, J.L., 1999. Structure of acetylcholinesterase complexedwith(-)-galanthamineat 2.3 A resolution. FEBS Lett. 463, 321–326.
Greig, N.H., Utsuki, T., Yu, Q., Zhul, X., Holloway, H.W., Perry, T., Lee1, B., Ingram, D.K., Lahir, D.K., 2001. A new therapeutic target in Alzheimer’s disease treatment: attention to butyrylcholinesterase. Curr. Med. Res. Opin. 17, 159–165.
Heinrich, M., Teoh, H.L., 2004. Galanthamine from snowdrop - the development of a modern drug against Alzheimer’s disease from local Caucasian knowledge. J. Ethnopharmacol. 92, 147–162.
Houghton, P.J., Rena, Y., Howes, M.J., 2006. Acetylcholinesterase inhibitors from plants and fungi. Nat. Prod. Rep. 23, 181–199.
Lee, S.S., Venkatesham, U., Rao, C.P., Lam, S.H., Lin, H.M., 2007. Preparation of sec-olycorines against acetylcholinesterase. Bioorg. Med. Chem. 15, 1034–1043.
López, S., Bastida, J., Viladomat, F., Codina, C., 2002. Acetylcholinesterase inhibitory activity of some Amaryllidaceae alkaloids and Narcissus extracts. Life Sci. 71, 2521–2529.
Marco, L., Carreiras, M.D., 2006. Galanthamine, a natural product for the treatment of Alzheimer’s disease. Recent Pat. CNS Drug. Discov. 1, 105–111.
Mehndiratta, M.M., Pandey, S., Kuntzer, T., 2011. Acetylcholinesterase inhibitor treatment for myasthenia gravis. Cochrane Database Syst. Rev. 2, 1–20.
Mehta, M., Adem, A., Sabbagh, M., 2012. New acetylcholinesterase inhibitors for Alzheimer’s disease. Int. J. Alzheimers Dis., https://doi.org/10.1155/2012/728983.
Morris, G.M., Goodsell, S.D., Halliday, R.S., Huey, R.S., Hart, R., Belew, R.K., Olson, J., 1998. Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J. Comput. Chem. 19, 1639–1662.
Mukherjee, P.K., Kumar, V., Mal, M., Houghton, P.J., 2007. Acetylcholinesterase inhibitors from plants. Phytomedicine 14, 289–300.
Ortiz, J.E., Berkov, S., Pigni, N.B., Theoduloz, C., Roitman, G., Tapia, A., Bastida, J., Feresin, G., 2012. Wild Argentinian Amaryllidaceae, a new renewable source of the acetylcholinesterase inhibitor galanthamine and other alkaloids. Molecules 17, 13473–13482.
Ortiz, J.E., Pigni, N.B., Andujar, S.A., Roitman, G., Suvire, F.D., Enriz, R.D., Tapia, A., Bastida, J., Feresin, G.E., 2016. Alkaloids from Hippeastrum argentinum and their cholinesterase-inhibitory activities: an in vitro and in silico study. J. Nat. Prod. 79, 1241–1248.
Perry, E.K., 1986. The cholinergic hypothesis, ten years on. Br. Med. Bull. 42, 63–69.
Rahman, A.U., Choudhary, M.I., 2001. Bioactive natural products as a potential source of new pharmacophores a theory of memory. Pure Appl. Chem. 73, 555–560.
Ravena, P., 2003. Elucidation and systematics of the Chilean genera of Amaryllidaceae. Bot. Aust. 2, 1–21.
Rhee, I.K., Appels, N., Hofte, B., Karabatak, B., Erkelens, C., Stark, L.M., Flippin, L.A., Ver-poorte, R., 2004. Isolation of the acetylcholinesterase inhibitor ungeremine from Nerine bowdenii by preparative HPLC coupled on-line to a flow assay system. Biol. Pharm. Bull. 27, 1804–1809.
Rhee, I.K., Van Rijn, R.M., Verpoorte, R., 2003. Qualitative determination of false-positive effects in the acetylcholinesterase assays using thin layer chromatography. Phytochem. Anal. 14, 127–131.
Schulz, V., 2003. Ginkgo extract or cholinesterase inhibitors in patients with dementia: what clinical trial and guidelines fail to consider. Phytomedicine 10, 74–79.
Sheng-Dian, H., Yul, Z., Hong-Ping, H., Shi-Fei, L., Gui-Hua, T., Duo-Zhi, Ch., Ming-Ming, C., Ying-Tong, D., Xiao-Jiang, H., 2013. A new Amarylliceae alkalolid from the bulbs ofLycoris radiate. Chin. J. Nat. Med. 11, 406–410.
Stryer, L., 1995. Biochemistry, 4th ed. W.H. Freeman & Co., New York.
Tram, N., Mitova, M., Bankova, V., Handjieva, N., Popov, S.S., 2014. GC-MS ofCrinum latifolium L. alkaloids. Z. Naturforsch. C 57, 239–242.
Wang, Y.H., Wan, Q.L., Gu, Ch.D., Luo, H.R., Long Ch., L., 2012. Synthesis and biological evaluation of lycorine derivatives as dual inhibitors of human acetylcholinesterase and butyrylcholinesterase. Chem. Cent. J., https://doi.org/10.1186/1752-153X-6-96.
Waterhouse, A.M., Procter, J.B., Martin, D.M.A., Clamp, M., Barton, G.J., 2009. Jalview Version 2 - a multiple sequence alignment editor and analysis work bench. Bioin-formatics 25, 1189–1191.
World Alzheimer Report, 2015. The Global Impact of Dementia, http://www.worldalzreport2015.org/(accessed 11.08.17).
Zarotsky, V., Sramek, J.J., Cutler, N.R., 2003. Galanthamine hydrobromide: an agent for Alzheimer’s disease. Am. J. Health-Syst. Pharmacist. 60, 446–452.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moraga-Nicolás, F., Jara, C., Godoy, R. et al. Rhodolirium andicola: a new renewable source of alkaloids with acetylcholinesterase inhibitory activity, a study from nature to molecular docking. Rev. Bras. Farmacogn. 28, 34–43 (2018). https://doi.org/10.1016/j.bjp.2017.11.009
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2017.11.009