Abstract
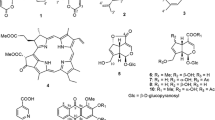
Roccella montagnei Bél. belongs to lichen family Roccelleceae growing luxuriantly along the coastal regions of India. As Roccella has been shown to be bioactive, we prepared methanolic extract and assessed its anticancer potential. The methanolic extract showed significant in vitro cytotoxic activity against four human cancer cell lines such as colon (DLD-1, SW-620), breast (MCF-7), head and neck (FaDu). This prompted us to isolate bioactive compounds through column chromatography. Two compounds roccellic acid and everninic acid have been isolated, out of which everninic acid is reported for the first time. Both the compounds have been tested for in vitro cytotoxic activity in which roccellic acid showed strong anticancer activity as compared to the everninic acid. Cyclin Dependent Kinase (CDK-10) contributes to proliferation of cancer cells, and aberrant activity of these kinases has been reported in a wide variety of human cancers. These kinases therefore constitute biomarkers of proliferation and attractive pharmacological targets for development of anticancer therapeutics. Therefore both the isolated compounds were tested for in silico molecular docking study against Cyclin Dependent Kinase isomer enzyme to support the cytotoxic activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Balaji, P., Bharath, P., Satyan, R., Hariharan, G., 2006. In vitro antimicrobial activity of Roccella montagnei thallus extracts. J. Trop. Med. Plant 7, 169–173.
Cetin, H., Tufan-Cetin, O., Turk, A.O., Tay, T., Candan, M., Yanikoglu, A., Sumbul, H., 2008. Insecticidal activity of major lichen compounds, (−)-and (+)-usnic acid, against the larvae of house mosquito, Culex pipiens L. Parasitol. Res. 102, 1277–1279.
Hawksworth, D., Honegger, R., 1994. The lichen thallus: a symbiotic phenotype of nutritionally specialized fungi and its response to gall producers. In: Williams, M.A.J. (Ed.), Plant Galls. Organisms, Interactions, Populations, The Systematics Association Special Volume, vol. 49. Clarendon Press, Oxford, pp. 77–98.
Huneck, S., Yoshimura, I., 1996. Identification of Lichen Substances. Springer, Berlin.
Krishna, S., Singh, D.K., Meena, S., Datta, D., Siddiqi, M.I., Banerjee, D., 2014. Pharmacophore-based screening and identification of novel human ligase I inhibitors with potential anticancer activity. J. Chem. Inf. Model. 54, 781–792.
Marshak, A., Kuschner, M., 1950. The action of streptomycin and usnic acid on the development of tuberculosis in guinea pigs. Public Health Rep. (1896–1970), 131–144.
Mishra, T., Pal, M., Meena, S., Datta, D., Dixit, P., Kumar, A., Meena, B., Rana, T., Upreti, D., 2016. Composition and in vitro cytotoxic activities of essential oil of Hedychium spicatum from different geographical regions of western Himalaya by principal components analysis. Nat. Prod. Res. 30, 1224–1227.
Mittal, O., Neelakantan, S., Seshaadri, T., 1952. Chemical investigation of Indian lichens. XIV. Chemical components of Ramalina calicaris and Ramalina sinensis. J. Sci. Ind. Res. India B 11, 386–387.
Müller, K., 2001. Pharmaceutically relevant metabolites from lichens. Appl. Microbiol. Biotechnol. 56, 9–16.
Nanayakkara, C., Bombuwala, K., Kathirgamanathar, S., Adikaram, N., Wijesundara, D., Hariharan, G., Wolseleys, P., Karunaratne, V., 2010. Effect of some lichen extracts from Sri Lanka on larvae Ofaedes aegypti and the fungus Cladosporium cladosporioides. J. Natl. Sci. Found. Sri Lanka 33, 147–149.
Peyressatre, M., Prével, C., Pellerano, M., Morris, M.C., 2015. Targeting cyclindependent kinases in human cancers: from small molecules to peptide inhibitors. Cancers 7, 179–237.
Tehler, A., Dahlkild, Å., Eldenäs, P., Feige, G.B., 2004. The phylogeny and taxonomy of Macaronesian, European and Mediterranean Roccella (Roccellaceae, Arthoniales). Symb. Bot. Upsal. 34, 405–428.
Williams, D.E., Bombuwala, K., Lobkovsky, E., de Silva, E.D., Karunatne, V., Allen, T.M., Clardy, J., Andersen, R.J., 1998. Ambewelamides A and B, antineoplastic epidithiapiperazinediones isolated from the lichen Usnea sp. Tetrahedron Lett. 39, 9579–9582.
Yusof, H., Azahar, H., Din, L.B., Ibrahim, N., 2015. Chemical constituents of the lichens Cladonia multiformis and cryptothecia SP. Malay. J. Anal. Sci. 19, 930–934.
Zhang, Y., 2007. Template-based modeling and free modeling by I-TASSER in CASP7. Proteins: Struct. Funct. Bioinform. 69, 108–117.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
Conceived and design the experiments: MP DD DKU. Performed the experiments: TM SS, SM, and RS. Analyzed the data: MP DD. Contributed reagents/material/analysis tools: DD DKU. Wrote the paper: MP TM SS SM.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mishra, T., Shukla, S., Meena, S. et al. Isolation and identification of cytotoxic compounds from a fruticose lichen Roccella montagnei, and it’s in silico docking study against CDK-10. Rev. Bras. Farmacogn. 27, 724–728 (2017). https://doi.org/10.1016/j.bjp.2017.07.006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2017.07.006