Abstract
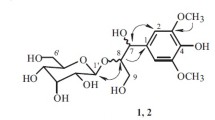
Two new monoterpene glycosides, perillanolides A and B, together with a known compound reported from the genus Perilla for the first time were isolated and characterized from the leaves of Perilla frutescens (L.) Britton, Lamiaceae, a garnish and colorant for foods as well as commonly used for traditional medicine. The structures of the isolated compounds were elucidated on the basis of extensive spectroscopic evidences derived from nuclear magnetic resonance experiments, mass spectrometry and by comparing their physical and spectroscopic data of literature. These compounds, together with the previously isolated secondary metabolites of this species, were investigated for their inhibitory effects on xanthine oxidase in vitro. Of the compounds, luteolin showed the strongest inhibitory activity with an IC50 value of 2.18 μM. Esculetin and scutellarein moderately inhibited the enzyme, while perillanolides A and B, and 4-(3,4-dihydroxybenzoyloxymethyl)phenyl-O-β-d-glucopyranoside exerted weak activities.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bae, J.S., Han, M., Shin, H.S., Kim, M.K., Shin, C.Y., Lee, D.H., Chung, J.H., 2017. Perilla frutescens leaves extract ameliorates ultraviolet radiation-induced extracellular matrix damage in human dermal fibroblasts and hairless mice. J. Ethnopharmacol. 195, 334–342.
Bassoli, A., Borgonovo, G., Morini, G., De Petrocellis, L., Schiano Moriello, A., Di Marzo, V., 2013. Analogues of perillaketone as highly potent agonists of TRPA1 channel. Food Chem. 141, 2044–2051.
Bierstedt, A., Stölting, J., Fröhlich, R., Metz, P., 2001. Enzymatic kinetic resolution of l-(3’-furyl)-3-buten-l-ol and 2-(2’-furyl)-propan-l-ol. Tetrahedron 12, 3399–3407.
Choi, U.K., Lee, O.H., Lim, S.I., Kim, Y.C., 2010. Optimization of antibacterialactivity of Perilla frutescens var. acuta leaf against Pseudomonas aeruginosa using the evolutionary operation-factorial design technique. Int. J. Mol. Sci. 11, 3922–3932.
Feng, MX., Wang, S.F., Zhang, X.X., 2013. Chemical constituents in fruits of Lycium barbarum. Chin. Tradit. Herb. Drugs 44, 265–268.
Fu, C., Chen, C., Zhou, G.X., Ye, W.C., 2011. Chemical constituents from fruits of Amomum villosum. Chin. Tradit. Herb. Drugs 42, 2410–2412.
Gao, H., Wang, W., Chu, W.X., Liu, K., Liu, Y., Liu, X.H., Yao, H.L., Gao, Q,, 2017. Paniculatumoside G, a new C21 steroidal glycoside from Cynanchum paniculatum. Rev. Bras. Farmacogn. 27, 54–58.
Heci, Y., 2001. Valuable ingredients from herb Perilla: a mini review. Innov. Food Technol. 29–30, 32–33.
Huo, L.N., Wang, W., Zhang, C.Y., Shi, H.B., Liu, Y., Liu, X.H., Guo, B.H., Zhao, D.M., Gao, H., 2015. Bioassay-guided isolation and identification of xanthine oxidase inhibitory constituents from the leaves of Perilla frutescens. Molecules 20, 17848–17859.
Huo, L.N., Wang, W., Liu, Y., Liu, X.H., Zhang, L., Cheng, K., Liu, K., Gao, H., 2016. Chemical constituents from Perilla frutescens. Chin. Tradit. Herb. Drugs 47, 26–31.
Jiang, H., Gschwend, B., Albrecht, L., Jørgensen, K.A., 2010. Organocatalytic preparation of simple β-hydroxy and β-amino esters: low catalyst loadings and gram-scale synthesis. Org. Lett. 12, 5052–5055.
Kim, E.Y., Choi, H.J., Chung, T.W., Choi, J.Y., Kim, H.S., Jung, Y.S., Lee, S.O., Ha, K.T., 2016. Water-extracted Perilla frutescens increases endometrial receptivity though leukemia inhibitory factor-dependent expression of integrins. J. Pharmacol. Sci. 131, 259–266.
Kimura, J., Maki, N., 2002. New loliolide derivatives from the brown alga Undaria pinnatifida. J. Nat. Prod. 65, 57–58.
Kotkar, S.P., Suryavanshi, G.S., Sudalai, A., 2007. Ashort synthesis of (+)-harzialactone A and (R)-(+)-4-hexanolide via proline-catalyzed sequential α-aminooxylation and Horner-Wadsworth-Emmons olefination of aldehydes. Tetrahedron 18, 1795–1798.
Lee, J.K., Kim, N.S., 2007. Genetic diversity and relationships of cultivated and weedy types of Perilla frutescens collected from East Asia revealed by SSR markers. Korean J. Breed. Sci. 39, 491–499.
Liang, Q., Yang, S.X., Kuang, Y., Liu, L., 2015. Chemical constituents from stems of Pleioblastus amarus. Chin. Tradit. Herb. Drugs 46, 1125–1128.
Liu, K., Wang, W., Guo, B.H., Gao, H., Liu, Y., Liu, X.H., Yao, H.L., Cheng, K., 2016. Chemical evidence for potent xanthine oxidase inhibitoryactivity of ethylacetate extract of Citrus aurantium L. dried immature fruits. Molecules 21, 302.
Makino, T., Ono, T., Muso, E., Honda, G., 2002. Effect of Penila frutescens on nitric oxide production and DNA synthesis in cultured murine vascular smooth muscle cells. Phytother. Res. 16 (Suppl. 1), S19–S23.
Nakatani, N., Kikuzaki, H., 1987. A new antioxidative glucoside isolated from Oregano (Origanum vulgare L.). Agric. Biol. Chem. 51, 2727–2732.
Park, J.H., Lee, D.G., Yeon, S.W., Kwon, H.S., Ko, J.H., Shin, D.J., Park, H.S., Kim, Y.S., Bang, M.H., Baek, N.I., 2011. Isolation of megastigmane sesquiterpenes from the silkworm (Bombyx mori L.) droppings and their promotionactivity on HO-1 and SIRT1. Arch. Pharm. Res. 34, 533–542.
Ponou, B.K., Teponno, R.B., Ricciutelli, M., Quassinti, L., Bramucci, M., Lupidi, G., Barboni, L., Tapondjou, A.L., 2010. Dimeric antioxidant and cytotoxic triterpenoid saponins from Terminalia ivorensis A. Chev. Phytochemistry 71, 2108–2115.
Shin, T.Y., Kim, S.H., Kim, S.H., Kim, Y.K., Park, HJ., Chae, B.S., Jung, HJ., Kim, H.M., 2000. Inhibitory effect of mast cell-mediated immediate-type allergic reactions in rats by Penlla frutescens. Immunopharmacol. Immunotoxicol. 22, 489–500.
Takeda, H., Tsuji, M., Matsumiya, T., Kubo, M., 2002. Identification of rasmarinicacid as a novel antidepressive substance in the leaves of Perilla fritescens Britton var. acuta Kudo (Perillae Herba). Nihon Shinkei Seishin Yakurigaku Zasshi 22, 15–22.
Wang, Y., Huang, X., Han, J., Zheng, W., Ma, W., 2013. Extract of Perilla frutescens inhibits tumor proliferation of HCC via PI3K/AKT signal pathway. Afr. J. Tradit. Complement Altera Med. 10, 251–257.
Yoon, H., Eom, S., Hyun, J., Jo, G., Hwang, D., Lee, S., Yong, Y., Park, J.C., Lee, Y.H., Lim, Y., 2011. 1H and 13C NMR data on hydroxy/methoxy flavonoids and the effects of substituents on chemical shifts. Bull. Korean Chem. Soc. 32, 2101–2104.
Yu, H., Qiu, J.F., Ma, L.J., Hu, Y.J., Li, P., Wan, J.B., 2016. Phytochemical and phytopharmacological review of Penlla frutescens L. (Labiatae), a traditional edible-medicinal herb in China. Food Chem. Toxicol., https://doi.org/10.1016/j.fct.2016.11.023.
Zekonis, G., Zekonis, J., Sadzeviciene, R., Simoniene, G., Kevelaitis, E., 2008. Effect of Penlla frutescens aqueous extract on free radical production by human neutrophil leukocytes. Medicina (Kaunas) 44, 699–705.
Zhang, L.Y., Ren, LZ., Wang, T.H., Dong, X., Wan, M.Z., Wu, H.F., Mei, Q.X., Gao, Y.H., 2014. Chemical constituents from Lepidogrammitis drymoglossoides. Chin. Tradit. Herb. Drugs 45, 2890–2894.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
YL, XHL, GLL, and WJG performed the extraction, isolation, and elucidation of the constituents. SZ and XYF were involved in the biological evaluations. HG confirmed all the isolation and structural elucidation procedures. WW designed the study, supervised the laboratory work, and contributed to critical reading of the manuscript. All the authors have read the final manuscript and approved the submission.
Conflicts of interest
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, Y., Liu, XH., Zhou, S. et al. Perillanolides A and B, new monoterpene glycosides from the leaves of Perilla frutescens. Rev. Bras. Farmacogn. 27, 564–568 (2017). https://doi.org/10.1016/j.bjp.2017.06.003
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2017.06.003