Abstract
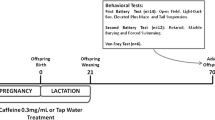
Drugs used in the treatment of depression can cross the placenta giving rise to questions regarding the effects these drugs exert on the fetus. Hypericum perforatum L., Hypericaceae, is a natural product used to treat depression. However, information about its toxicity and the occurrence of alterations in the central nervous system development of the offspring is scarce. This work assessed the behavior of adult male rats born from mothers treated with Hypericum extract during gestation and analyzed the fluorescence of the extract in different organs of mothers and fetuses. Male pups were divided into three treated groups, corresponding to the administration of the Hypericum extract to mothers at the dose levels of 36 mg/kg, 72 mg/kg and 144 mg/kg, and one control group in which the mothers received distilled water. At 90 days of age, the offspring underwent the following tests: rotarod, pentobarbital-induced sleep time, elevated plus maze, hole-board and forced swimming test. The observed fluorescence indicated the presence of the extract in all tissues analyzed. The obtained results suggest lasting changes in the performances displayed in the CNS, depression and anxiety tests, indicating that the use of Hypericum during gestation could interfere with the behavioral development of the offspring reducing anxiety and depression when they become adults. We suggest that these alterations are associated with the reprogramming of the brain regions related to changes in emotional reactivity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bahls, S.C., 2001. Tratamento fltoterápico da depressão. J. Bras. Psiquiatr. 50, 389–396.
Beck, A.T., Brad, A.A., 2008. Depression: Cause and Treatment, 2nd ed. University of Pennsylvania Press, Philadelphia.
Bennett, H.A., Einarson, A., Taddio, A., Koren, G., Einarson, T.R., 2004. Prevalence of depression during pregnancy: systematic review. Obstet. Gynecol. 103, 698–709.
Bergonzi, M.C., Bilia, A.R., Gallori, S., Guerrini, D., Vincieri, F.F., 2001. Variability in the content of the constituents of Hypericum perforatum L. and some commercial extracts. Drug Dev. Ind. Pharm. 27, 491–497.
Borges, L.V., Cancino, J.C.C., Peters, V.M., Las Casas, L., Guerra, M.O., 2005. Evaluation of Hypericum perforatum toxicity when administered to pregnant rats. Rev. Assoc. Med. Bras. 51, 206–208.
Borrelli, F., Izzo, A.A., 2009. Herb-drug interactions with St John’s wort (Hypericum perforatum): an update on clinical observations. AAPS J. 11, 710–727.
Bouron, A., Lorrain, E., 2014. Effets cellulaires et moléculaires de l’hyperforine, un antidépresseur végétal: revue de la littérature. L’Encéphale 40, 108–113.
Brattstrom, A., 2009. Long-term effects of St. John’s wort (Hypericum perforatum) treatment: a 1-year safety study in mild to moderate depression. Phytomedicine 16, 277–283.
Cada, A.M., Hansen, D.K., LaBorde, J.B., Ferguson, S.A., 2001. Minimal effects from developmental exposure to St. John’s wort (Hypericum perforatum) in Sprague-Dawley rats. Nutr. Neurosci. 4, 135–141.
Camacho, R.S., Cantinelli, F.S., Ribeiro, C.S., Cantilino, A., Gonsales, B.K., Braguittoni, E., Rennó, J.R., 2006. Transtornos psiquiátricos na gestação e no puerpério: classificação, diagnóstico e tratamento. Rev. Psiquiatr. Clin. 33, 92–102.
Campagne, D.M., 2004. The obstetrician and depression during pregnancy. Eur. J. Obstet. Gynaecol. Reprod. Biol. 116, 125–130.
Carlini, E.A., Mendes, F.R., 2011. Protocolos em psicofarmacologia comportamental: um guia para a pesquisa de drogas com ação sobre o SNC, com ênfase nas plantas medicinais. Fap-Unifesp, Sao Paulo.
Crupi, R., Mazzon, E., Marino, A., La Spada, G., Bramanti, P., Battaglia, F., Cuzzocrea, S., Spina, E., 2011. Hypericum perforatum treatment: effect on behaviour and neurogenesis in a chronic stress model in mice. BMC Complement. Altern. Med., https://doi.org/10.1186/1472-6882-11-7.
Draves, A.H., Walker, S.E., 2000. Determination of hypericin and pseudohypericin in pharmaceutical preparations by liquid chromatography with fluorescence detection. Br. J. Biomed. Sci. 749, 57–66.
Dugoua, J.J., Mills, E., Perri, D., Koren, G., 2006. Safety and efficacy of St. John’s wort (Hypericum) during pregnancy and lactation. Can. J. Clin. Pharmacol. 13, e268–e276.
Field, T., Diego, M., Hernandez-Reif, M., Figueiredo, B., Schanberg, S., Kuhn, C., Deeds, O., Contogeorgos, J., Ascencio, A., 2008. Chronic prenatal depression and neonatal outcome. Int. J. Neurosci. 118, 95–103.
Franklin, M., 2003. Sub-chronic treatment effects of an extract of Hypericum perforatum (St. John’s Wort, Li 160) on neuroendocrine responses to the 5-T2A agonist, DOI in the rat. Pharmacopsychiatry 36, 161–164.
Galeotti, N., Farzad, M., Bianchi, E., Ghelardini, C., 2014. PKC-mediated potentiation of morphine analgesia by St. John’s Wort in rodents and humans. J. Pharmacol. Sci. 124, 409–417.
Galeotti, N., Ghelardini, C., 2013. Reversal of NO-induced nociceptive hypersensitivity by St. John’s wort and hypericin: NF-KB, CREB and STAT1 as molecular targets. Psychopharmacology 227, 149–163.
Galeotti, N., Vivoli, E., Bilia, A.R., Vincieri, F.F., Ghelardini, C., 2010. St. John’s Wort reduces neuropathic pain through a hypericin-mediated inhibition of the protein kinase Cgamma and epsilon activity. Biochem. Pharmacol. 79, 1327–1336.
Gaspar, P., Cases, O., Maroteaux, L., 2003. The developmental role of serotonin: news from mouse molecular genetics. Nat. Rev. Neurosci. 4, 1002–1012.
Gregoretti, B., Stebel, M., Candussio, L., Crivellato, E., Bartoli, F., Decorti, G., 2004. Toxicity of Hypericum perforatum (St. John’s wort) administered during pregnancy and lactation in rats. Toxicol. Appl. Pharmacol. 200, 201–205.
Grundmann, O., Nakajima, J., Kamata, It, Seo, S., Butterweck, V., 2009. Kaempferol from the leaves of Apocynum venetum possesses anxiolytic activities in the elevated plus maze test in mice. Phytomedicine 16, 295–302.
Grundmann, O., Nakajima, J.I., Seo, S., Butterweck, V., 2007. Anti-anxiety effects of Apocynum venetum L. in the elevated plus maze test. J. Ethnopharmacol. 110, 406–411.
Han, H., Ma, Y., Eun, J.S., Li, R., Hong, J.T., Lee, M.K., Oh, K.W., 2009. Anxiolytic-like effects of sanjoinine A isolated from Zizyphispinosi semen: possible involvement of GABAergic transmission. Pharmacol. Biochem. Behav. 92, 206–213.
Herrera-Ruiz, M., Roman-Ramos, R., Zamilpa, A., Tortoriello, J., Jimenez-Ferrer, J.E., 2008. Flavonoids from Tilia americana with anxiolytic activity in plus-maze test. J. Ethnopharmacol. 118, 312–317.
Hiremath, S.P., Badami, S., Swamy, H.K.S., Patil, S.B., Londonkar, R.L., 1997. Antian-drogenic effect of Striga orobanchioides. J. Ethnopharmacol. 56, 55–60.
Hostetter, A., Ritchie, J.C., Stowe, Z.N., 2000. Amniotic fluid and umbilical cord blood concentrations of antidepressants in three women. Biol. Psychiatry 48, 1032–1034.
Howland, R.H., 2010. Update on St. John’s Wort. J. Psychosoc. Nurs. Ment. Health Serv. 48, 20–24.
Isacchi, B., Galeotti, N., Bergonzi, M.C., Ghelardini, C., Bilia, A.R., Vincieri, F.F., 2009. Pharmacological in vivo test to evaluate the bioavailability of some St John’s Wort innovative oral preparations. Phytother. Res. 23, 197–205.
Ito, T., Suzuki, T., Wellman, S.E., Ho, I.K., 1996. Pharmacology of barbiturate tolerance/dependence: GABAA receptors and molecular aspects. Life Sci. 59, 169–195.
Kannan, S., Varkey, D., Tyagi, M.G., 2013. Influence of ATP sensitive potassium channels on intravenous thiopentone aodium: a rotarod test to evaluate the motor coordination of rodents. Indian J. Pharmacol. 45, S245.
Laakmann, G., Schule, C., Baghai, T., Kieser, M., 1998. St. John’s wort in mild to moderate depression: the relevance of hyperforin for the clinical efficacy. Pharmacopsychiatry 31 (Suppl 1), 54–59.
Liang, D.Y., Shi, X., Li, X., Li, J., Clark, J.D., 2007. The beta2 adrenergic receptor regulates morphine tolerance and physical dependence. Behav. Brain Res. 181, 118–126.
Linde, K., 2009. St. John’s wort - an overview. Forsch Komplementmed 16, 146–155.
Linde, K., Berner, M.M., Kriston, L., 2008. St John’s wort for major depression. Cochrane Db Syst. Rev., 147.
Linde, K., Ramirez, G., Mulrow, C.D., Pauls, A., Weidenhammer, W., Melchart, D., 1996. St John’s wort for depression: an overview and meta-analysis of randomised clinical trials. Brit. Med. J. 313, 253–258.
Moretti, M.E., Maxson, A., Hanna, F., Koren, G., 2009. Evaluating the safety of St. John’s Wort in human pregnancy. Reprod. Toxicol. 28, 96–99.
Muller, M.J., Seifritz, E., Hatzinger, M., Hemmeter, U., HolsboerTrachsler, E., 1997. Side effects of adjunct light therapy in patients with major depression. Eur. Arch. Psy. Clin. N. 247, 252–258.
Murrin, L.C., Sanders, J.D., Bylund, D.B., 2007. Comparison of the maturation of the adrenergic and serotonergic neurotransmittersystems in the brain: implications for differential drug effects on juveniles and adults. Biochem. Pharmacol. 73, 1225–1236.
Nahas, R., Sheikh, O., 2011. Complementary and alternative medicine for the treatment of major depressive disorder. Can. Fam. Physician 57, 659–663.
Nepomuceno, F., Casas, L.L., Peters, V.M., Guerra, M.O., 2005. Desenvolvimento embrionario em ratas tratadas com Hypericum perforatum durante o periodo de implantação. Rev. Bras. Farmacogn. 15, 224–228.
Oberlander, T.F., Warburton, W., Misri, S., Aghajanian, J., Hertzman, C., 2006. Neonatal outcomes after prenatal exposure to selective serotonin reuptake inhibitor antidepressants and maternal depression using population-based linked health data. Arch. Gen. Psychiat. 63, 898–906.
Ott, M., Huls, M., Cornelius, M.G., Fricker, G., 2010. St. John’s Wort constituents modulate P-glycoprotein transport activity at the blood-brain barrier. World J. Pharm. Res. 27, 811–822.
Pereira, P.K., Lovisi, G.M., 2008. Prevalence of gestacional depression and associated factors. Rev. Psiq. Clin-Brazil 35, 144–153.
Petit-Demouliere, B., Chenu, F., Bourin, M., 2005. Forced swimming test in mice: a review of antidepressant activity. Psychopharmacology 177, 245–255.
Porsolt, R.D., Le Pichon, M., Jalfre, M., 1977. Depression: a new animal model sensitive to antidepressant treatments. Nature 266, 730–732.
Reichling, J., Hostanska, It, Sailer, R., 2003. St. John’s Wort (Hypericum perforatum L.) - multicompound preparations versus single substances. Forsch. Komp. Klas. Nat 10, 28–32.
Ruedeberg, C., Wiesmann, U.N., Brattstroem, A., Honegger, U.E., 2010. Hypericum perforatum L. (St John’s wort) extract Ze 117 inhibits dopamine re-uptake in rat striatal brain slices. An implication for use in smoking cessation treatment? Phytother. Res. 24, 249–251.
Russo, E., Scicchitano, F., Whalley, B.J., Mazzitello, C., Ciriaco, M., Esposito, S., Patane, M., Upton, R., Pugliese, M., Chimirri, S., Mammi, M., Palleria, C., De Sarro, G., 2014. Hypericum perforatum: pharmacokinetic, mechanism of action, tolerability, and clinical drug-drug interactions. Phytother. Res. PTR 28, 643–655.
Sarris, J., Panossian, A., Schweitzer, I., Stough, C., Scholey, A., 2011. Herbal medicine for depression, anxiety and insomnia: a review of psychopharmacology and clinical evidence. Eur. Neuropsychopharmacol. 21, 841–860.
Schmidt, M., Butterweck, V., 2015. The mechanisms of action of St. John’s wort: an update. Wien Med. Wochenschr. 165, 229–235.
Sokal, R.R., Rolf, F.J., 1994. Biometry. The Principles and Practice of Statistics in Biological Research, 4th ed. W.H. Freeman & Company, New York.
Souza, F.G.M., 1999. Tratamento da depressao. Rev. Bras. Psiquiatr. 21, 18–23.
Stahl, S.M., 1998. Mechanism of action of serotonin selective reuptake inhibitors. J. Affect Disord. 51, 215–235.
Stemmelin, J., Cohen, C., Terranova, J.P., Lopez-Grancha, M., Pichat, P., Bergis, O., Decobert, M., Santucci, V., Francon, D., Alonso, R., Stahl, S.M., Keane, P., Avenet, P., Scatton, B., le Fur, G., Griebel, G., 2008. Stimulation of the beta(3)-adrenoceptor as a novel treatment strategy for anxiety and depressive disorders. Neuropsy-chopharmacology 33, 574–587.
Tschudin, S., Lapaire, O., 2005. Antidepressants and pregnancy. Ther. Umsch. 62, 17–22.
Vieira, V.A., Campos, L.V., Silva, L.R., Guerra, M.G., Peters, V.M., Sá, R.C.S., 2013. Evaluation of postpartum behaviour in rats treated with Hypericum perforatum during gestation. Rev. Bras. Farmacogn 23, 796–801.
Wambebe, C., 1985. Influence of some agents that affect 5-hydroxytryptamine metabolism and receptors on nitrazepam-induced sleep in mice. Br. J. Pharmacol. 84, 185–191.
Woelk, H., Burkard, G., Grunwald, J., 1994. Benefits and risks ofthe hypericum extract LI 160: drug monitoring study with 3250 patients. J. Geriatr. Psychiatry. Neurol. 7 (Suppll), S34–S38.
Wolfensohn, S., Lloyd, M., 1994. Handbook of Laboratory Animal Management an Welfare, 4th ed. Oxford University Press, New York.
Xia, Y., Haddad, G.G., 1992. Ontogeny and distribution of GABAA receptors in rat brainstem and rostral brain regions. J. Neurosci. 49, 973–989.
Xu, L., Wei, C.E., Zhao, M.B., Wang, J.N., Tu, P.F., Liu, J.X., 2005. Experimental study ofthe total flavonoid in Hypericum perforatum on depression. Zhongguo Zhong YaoZaZhi 30, 1184–1188.
Author information
Authors and Affiliations
Corresponding author
Additional information
In memoriam.
Authors contributions
Participated in research design: RCSS, VAV, VMP and MOG; Conducted experiments: VAV, LVC, LRS and JJ; Contributed new reagents or analytic tools: VMP and MOG; Performed data analysis: RCSS, VAV, LVC and JJasmin; Wrote or contributed to the writing of the manuscript: RCSS and LVC.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Campos, L.V., Vieira, V.A., Silva, L.R. et al. Rats treated with Hypericum perforatum during pregnancy generate offspring with behavioral changes in adulthood. Rev. Bras. Farmacogn. 27, 361–368 (2017). https://doi.org/10.1016/j.bjp.2017.01.004
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2017.01.004