Abstract
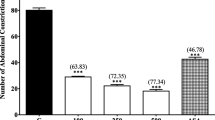
Melastoma malabathricum L., Melastomaceae, has been traditionally used to relieve diverse pain-related ailments. The objectives of the present study were to determine the antinociceptive activity of methanol extract of M. malabathricum leaves and to elucidate the possible mechanisms of antinociception involved using various rats’ models. The extract (100, 250, and 500 mg/kg) was administered orally 60 min prior to subjection to the respective test. The in vivo acetic acid-induced abdominal constriction, formalininduced paw licking, and hot plate tests were used as the models of nociception to evaluate the extract antinociceptive activity. Further studies were carried out to determine the role of opioid and vanilloid receptors, glutamate system and nitric oxide/cyclic guanosine phosphate (NO/cGMP) pathway in modulating the extract antinociceptive activity. From the results obtained, M. malabathricum exhibited significant (p < 0.05) antinociceptive activity in all the chemical- and thermal-induced nociception models. Naloxone (5 mg/kg), a non-selective opioid antagonist, failed to significantly affect the antinociceptive activity of MEMM when assessed using the abdominal constriction-, hot plate- and formalin-induced paw licking-test. M. malabathricum also significantly (p < 0.05) reversed the nociceptive response in capsaicinand glutamate-induced paw licking test. Furthermore, only l-arginine (a nitric oxide precursor) alone, but not, NG-nitro-l-arginine methyl esters (l-NAME; an inhibitor of NO synthase), methylene blue (MB; an inhibitor of cGMP), or their combination thereof, significantly (p < 0.05) block the antinociceptive activity of M. malabathricum. In conclusion, M. malabathricum exerted a non-opioid antinociceptive activity at the central and peripheral levels partly via the inhibition of vanilloid receptors and glutamatergic system, and activation of the NO-mediated/cGMP-independent pathway.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barrière, D.A., Mallet, C., Blomgren, A., Simonsen, C., Daulhac, L., Libert, F., Chapuy, E., Etienne, M., Högestätt, E.D., Zygmunt, P.M., Eschalier, A., 2013. Fatty acid amide hydrolase-dependent generation of antinociceptive drug metabolites acting on TRPV1 in the brain. PLOS ONE 8, e70690, https://doi.org/10.1371/journal.pone.0070690.
Beirith, A., Santos, A.R.S., Calixto, J.B., 2002. Mechanisms underlying the nociception and paw oedema caused by injection of glutamate into the mouse paw. Brain Res. 924, 219–228.
Beirith, A., Santos, A.R., Rodrigues, A.L., Creczynski-Pasa, T.B., Calixto, J.B., 1998. Spinal and supraspinal antinociceptive action of dipyrone in formalin, capsaicin and glutamate tests. Study of the mechanism of action. Eur. J. Pharmacol. 345, 233–245.
Bentley, G.A., Newton, S.H., Starr, J., 1981. Evidence for an action of morphine and the enkephalins on sensory nerve endings in the mouse peritoneum. Br. J. Pharmacol. 73, 325–332.
Bhave, G., Karim, F., Carlton, S.M., Gereau, I.V.R.W., 2001. Peripheral group I metabotropic glutamate receptors modulate nociception in mice. Nat. Neurosci. 4, 417–423.
Caterina, M.J., Julius, D., 2001. The vanilloid receptor: a molecular gateway to the pain pathway. Annu. Rev. Neurosci. 24, 487–517.
Collier, H.O., Dinneen, L.C., Johnson, C.A., Schneider, C., 1968. The abdominal constriction response and its suppression by analgesic drugs in the mouse. Br. J. Pharmacol. 32, 295–310.
Comalada, M., Camuesco, D., Sierra, S., Ballester, I., Xaus, J., Gálvez, J., Zarzuelo, A., 2005. In vivo quercitrin anti-inflammatory effect involves release of quercetin, which inhibits inflammation through down-regulation of the NF-kappa B pathway. Eur. J. Immunol. 35, 584–592.
Cortright, D.N., Szallasi, A., 2004. Biochemical pharmacology of the vanilloid receptor TRPV1. An update. Eur. J. Biochem. 271, 1814–1819.
Cragg, G.M., Newman, D.J., Snader, K.M., 2003. Natural product in drug discovery and development. J. Nat. Prod. 66, 1022–1037.
Cui, X., Zhang, J., Ma, P., Myers, D.E., Goldberg, I.G., Sittler, K.J., Barb, J.J., Munson, P.J., Cintron Adel, P., McCoy, J.P., Wang, S., Danner, R.L., 2005. cGMPindependent nitric oxide signaling and regulation of the cell cycle. BMC Genomics, https://doi.org/10.1186/1471-2164-6-151.
Déciga-Campos, M., Montiel-Ruiz, R.M., Navarrete-Vázquez, G., López-Muñoz, F.J., 2007. Palmitic acid analogues exhibiting antinociceptive activity in mice. Proc. West Pharmacol. Soc. 50, 75–77.
Deraedt, R., Jougney, S., Delevalcese, F., Falhout, M., 1980. Release of prostaglandins E and F in an algogenic reaction and it inhibition. Eur. J. Pharmacol. 51, 17–24.
Dray, A., Dickenson, A., 1991. Systemic capsaicin and olvanil reduce the acute algogenic and the late inflammatory phase following formalin injection into rodent paw. Pain 47, 79–83.
Faravani, M., (PhD thesis) 2009. The population biology of Straits Rhododendron (Melastoma malabathricum L.). University of Malaya, Kuala Lumpur, Malaysia.
Ferreira, S.H., Duarte, I.D., Lorenzetti, B.B., 1991. The molecular mechanism of action of peripheral morphine analgesia: stimulation of the cGMP system via nitric oxide release. Eur. J. Pharmacol. 201, 121–122.
Gadotti, V.M., Schmeling, L.O., Machado, C., Liz, F.H., Meyre-Silva, C., Santos, A.R., 2005. Antinociceptive action of the extract and the flavonoid quercitrin isolated from Bauhinia microstachya leaves. J. Pharm. Pharmacol. 57, 1345–1351.
Garthwaite, J., Boulton, C.L., 1995. Nitric oxide signaling in the central nervous system. Annu. Rev. Physiol. 57, 683–706.
Giglio, C.A., Defino, H.L.A., Da-Silva, C.A., De-Souza, A.S., Del Bel, E.A., 2006. Behavioral and physiological methods for early quantitative assessment of spinal cord injury and prognosis in rats. Braz. J. Med. Biol. Res. 39, 1613–1623.
Grosvenor, P.W., Gothard, P.K., McWilliam, N.C., Supriono, A., Gray, D.O., 1995. Medicinal plants from Riau Province, Sumatra, Indonesia. Part 1: Uses. J. Ethnopharmacol. 45, 75–95.
Hosseinzadeh, H., Younesi, H.M., 2002. Antinociceptive and anti-inflammatory effects of Crocus sativus L. stigma and petal extracts in mice. BMC Pharmacol., https://doi.org/10.1186/1471-2210-2-7.
Hunskaar, S., Berge, O.G., Hole, K., 1986. A modified hot-plate test sensitive to mild analgesics. Behav. Brain Res. 21, 101–108.
Hunskaar, S., Hole, K., 1987. The formalin test in mice: dissociation between inflammatory and non-inflammatory pain. Pain 30, 103–114.
Hussain, F., Abdulla, M.A., Noor, S.M., Ismail, S., Ali, H.M., 2008. Gastroprotective effects of Melastoma malabathricum aqueous leaf extract against ethanolinduced gastric ulcer in rats. Am. J. Biochem. Biotechnol. 4, 438–441.
Ichinose, F., Mi, W.D., Miyazaki, M., Onouchi, T., Goto, T., Morita, S., 1998. Lack of correlation between the reduction of sevoflurane MAC and the cerebellar cyclic GMP concentrations in mice treated with 7-nitroindazole. Anesthesiology 89, 143–148.
Kamisan, F.H., Yahya, F.A., Mamat, S.S., Kamarolzaman, M.F.F., Suhaili, Z., Mohtarrudin, N., Ching, S.M., Teh, L.K., Salleh, M.Z., Abdullah, M.N.H., Zakaria, Z.A., 2014. Effect of methanol extract of Dicranopteris linearis against carbon tetrachloride-induced acute liver injury in rats. BMC Complim. Alt. Med. https://doi.org/10.1186/1472-6882-14-123.
Larson, A.A., Kovacs, K.J., Cooper, J.C., Kitto, K.F., 2000. Transient changes in the synthesis of nitric oxide result in long-term as well as short-term changes in acetic acid-induced writhing in mice. Pain 86, 103–111.
Le Bars, D., Gozariu, D., Cadden, M.S.W., 2001. Animal models of nociception. Pharmacology 53, 597–652.
Luiz, A.P., Moura, J.D., Meotti, F.C., Guginski, G., Guimaraes, C.L., Azevedo, M.S., Rodrigues, A.L., Santos, A.R., 2007. Antinociceptive action of ethanolic extract obtained from roots of Humirianthera ampla Miers. J. Ethnopharmacol. 114, 355–363.
Manicam, C., Abdullah, J.O., Mohd Tohit, E.R., Seman, Z., Sieo, C.C., Hamid, M., 2010. In vitro anticoagulant activities of Melastoma malabathricum L. aqueous leaf extract: a preliminary novel finding. J. Med. Plants Res. 4, 1464–1472.
Machelska, H., Labuz, D., Przewlocki, R., Przewlocka, B., 1997. Inhibition of nitric oxide synthase enhances antinociception mediated by Mu, Delta and Kappa opioid receptors in acute and prolonged pain in the rat spinal cord. J. Pharm. Exp. Ther. 282, 977–984.
Matsumoto, K., Horie, S., Ishikawa, H., Takayama, H., Aimi, N., Ponglux, D., Watanabe, K., 2004. Antinociceptive effect of 7-hydroxymitragynine in mice: discovery of an orally active opioid analgesic from the Thai medicinal herb Mitragyna speciosa. Life Sci. 74, 2143–2155.
McNamara, C.R., Mandel-Brehm, J., Bautista, D.M., Siemens, J., Deranian, K.L., Zhao, M., 2007. TRPA1 mediates formalin-induced pain. Proc. Natl. Acad. Sci. 104, 13525–13530. Mohd.
Sani, M.H., Zakaria, Z.A., Balan, T., Teh, L.K., Salleh, M.Z., 2012. Antinociceptive activity of methanol extract of Muntingia calabura leaves and the mechanisms of action involved. Evid. Based Complim. Altern. Med., https://doi.org/10.1155/2012/890361.
Nazlina, I., Norha, S., Noor Zarina, A.W., Ahmad, I.B., 2008. Cytotoxicity and antiviral activity of Melastoma malabathricum extracts. Malays. J. Appl. Biol. 37, 53–55.
Neugebauer, V., 2001a. Metabotropic glutamate receptors: novel targets for pain relief. Expert Rev. Neurother. 1, 207–224.
Neugebauer, V., 2001b. Peripheral metabotropic glutamate receptors: fight the pain where it hurts. Trends Neurosci. 24, 550–552.
Neugebauer, V., 2002. Metabotropic glutamate receptors–important modulators of nociception and pain behavior. Pain 98, 1–8.
Numazaki, M., Tominaga, M., 2004. Nociception and TRP channels. Curr. Drug Targets CNS Neurol. Disord. 3, 479–485.
Ong, H.C., Nordiana, M., 1990. Malay ethno-medico botany in Machang, Kelantan, Malaysia. Fitoterapia 70, 502–513.
Parada, C.A., Tambeli, C.H., Cunha, F.Q., Ferreira, S.H., 2001. The major role of peripheral release of histamine and 5-hydroxytryptamine in formalin-induced nociception. Neuroscience 102, 937–944.
Raghav, S.K., Gupta, B., Agrawal, C., Goswami, K., Das, H.R., 2006. Anti-inflammatory effect of Ruta graveolens L. in murine macrophage cells. J. Ethnopharmacol. 104, 234–239.
Rajenderan, M.T., 2010. Ethno medicinal uses and antimicrobial properties of Melastoma malabathricum. SEGi Rev. 3, 34–44.
Rang, H.P., Dale, M.M., Ritter, J.M., Flower, R.J., Henderson, G., 2011. Pharmacology, 7th ed. Elsevier Churchill Livingstone, Edinburgh, UK.
Rebolledo, R., Abarzúa, J., Zavala, A., Quiroz, A., Alvear, M., Aguilera, A., 2012. The effects of the essential oil and hydrolate of canelo (Drimys winteri) on adults of Aegorhinus superciliosus in the laboratory. Cien. Inv. Agr. 39, 481–488.
Ribeiro, R.A., Vale, M.L., Thomazzi, S.M., Paschoalato, A.B., Poole, S., Ferreira, S.H., 2000. Involvement of resident macrophages and mast cells in the writhing nociceptive response induced by zymosan and acetic acid in mice. Eur. J. Pharmacol. 387, 111–118.
Roosita, K., Kusharto, C.M., Sekiyama, M., Fachrurozi, M., Ohtsuka, R., 2008. Medicinal plants used by the villagers of a Sundanese community in West Java, Indonesia. J. Ethnopharmacol. 115, 72–81.
Sakurada, T., Katsumata, K., Tan-No, K., Sakurada, S., Kisara, K., 1992. The capsaicin test in mice for evaluating tachykinin antagonists in the spinal cord. Neuropharmacology 31, 1279–1285.
Sharma, H.K., Chhangte, L., Dolui, A.K., 2001. Traditional medicinal plants in Mizoram, India. Fitoterapia 72, 146–161.
Shibata, M., Ohkubo, M., Takahashi, T., Inoki, H., 1989. Modified formalin test; characteristic biphasic pain response. Pain 38, 347–352.
Simanjuntak, M., (M.S. thesis) 2008. Ekstraksi, fraksinasi: komponen ekstrak daun tumbuhan senduduk (Melsatoma malabathricum) serta pengujian efek sediaan krim terhadap penyembuhan luka bakar. Universitas Sumatera Utara, Sumatera, Indonesia.
Sulaiman, M.R., Somchit, M.N., Israf, D.A., Ahmad, Z., Moin, S., 2004. Antinociceptive effect of Melastoma malabathricum ethanolic extract in mice. Fitoterapia 75, 667–672.
Sunilson, J.A.J., Anandarajagopal, K., Kumari, A.V.A.G., Mohan, S., 2009. Antidiarrhoeal activity of leaves of Melastoma malabathricum L. Indian J. Pharm. Sci. 71, 691–695.
Susanti, D., Sirat, H.M., Ahmad, F., Mat Ali, R., 2008. Bioactive constituents from the leaves of Melastoma malabathricum L. J. Ilmiah Farmasi. 5, 1–8.
Tandon, O.P., Malhotra, T.V., Tandon, S., D’silva, I., 2003. Neurophysiology of pain: insight to orofacial pain. Indian J. Physiol. Pharmacol. 47, 247–269.
Tjolsen, A., Berge, O.G., Hunskaar, S., Rosland, J.H., Hole, K., 1992. The formalin test: an evaluation of the method. Pain 51, 5–17.
Verpoorte, R., 1998. Exploration of nature’s chemodiversity–the role of secondary metabolites as leads in drug development. Drug Discov. Today 3, 232–238.
Wiart, C., Mogana, S., Khalifah, S., Mahan, M., Ismail, S., Buckle, M., Narayana, A.K., Sulaiman, M., 2004. Antimicrobial screening of plants used for traditional medicine in the state of Perak, Peninsular Malaysia. Fitoterapia 75, 68–73.
Zakaria, Z.A., Raden Mohd Nor, R.N.S., Abdul Ghani, Z.D.F., Hanan Kumar, G., Sulaiman, M.R., Fatimah, C.A., 2006. Antinociceptive and anti-inflammatory properties of Melastoma malabathricum leaves chloroform extract in experimental animals. J. Pharmacol. Toxicol. 1, 337–345.
Zakaria, Z.A., Raden Mohd Nor, R.N.S., Hanan Kumar, G., Abdul Ghani, Z.D.F., Sulaiman, M.R., Rathna Devi, G., Mat Jais, A.M., Somchit, M.N., Fatimah, C.A., 2008. Antinociceptive, anti-inflammatory and antipyretic properties of Melastoma malabathricum leaves aqueous extract in experimental animals. Can. J. Physiol. Pharmacol. 84, 1291–1299.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contribution
ESJ carried out the experiments and drafted the manuscript. SAR and AKA participated in the experimental design, and helped to draft the manuscript. SMC and MND involved in the statistical analysis and manuscript preparation. ZAZ conceived of the study, participated in its design and helped to draft the manuscript. All authors read and approved the final manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jaios, E.S., Rahman, S.A., Ching, S.M. et al. Possible mechanisms of antinociception of methanol extract of Melastoma malabathricum leaves. Rev. Bras. Farmacogn. 26, 586–594 (2016). https://doi.org/10.1016/j.bjp.2016.01.011
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2016.01.011