Abstract
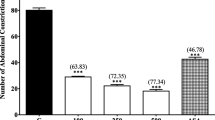
Muntingia calabura L, Muntingiaceae, is a medicinal plant for various pain-related diseases. The aims of the present study were to determine the antinociceptive profile and to elucidate the possible mechanisms of antinociception of petroleum ether partition obtained from crude methanol extract of M. calabura leaves using various animal models. The antinociceptive profile of petroleum ether fraction (given oral; 100, 250 and 500mg/kg) was established using the in vivo chemicals (acetic acid-induced abdominal constriction and formalin-induced paw licking test) and thermal (hot plate test) models of nociception. The role of glutamate, TRPV1 receptor, bradykinin, protein kinase C, potassium channels, and various opioid and non-opioid receptors in modulating the partition’s antinociceptive activity was also determined. The results obtained demonstrated that petroleum ether partition exerted significant (p<0.05) antinociception in all the chemicals-, thermal-, capsaicin-, glutamate-, bradykinin, and phorbol 12-myristate 13-acetate (PMA)-induced nociception models. The antinociceptive activity was reversed following pretreatment with opioid antagonists (i.e. naloxone, ß-funaltrexamine, naltrindole and nor-binaltorphimine), and the non-opioid receptor antagonists (i.e. pindolol (a ß-adrenoceptor), haloperidol (a non-selective dopaminergic), atropine (a non-selective cholinergic receptor), caffeine (a non-selective adenosinergic receptor), and yohimbine (an α2-noradrenergic)). In addition, pretreatment with l-arginine (a nitric oxide (NO) donor), NG-nitro-l-arginine methyl esters (l-NAME; an inhibitor of NO synthase (NOS)), methylene blue (MB; an inhibitor of cyclic-guanosine monophosphate (cGMP) pathway), or their combination failed to inhibit petroleum ether partition’s antinociception. In conclusion, petroleum ether partition exerts antinociceptive activity at the peripheral and central levels via the modulation of, partly, the opioid (i.e. μ, κ and δ) and several non-opioids (i.e. ß-adrenergic, dopaminergic, cholinergic, adenosinergic, and α2-noradrenergic) receptors, glutamatergic, TRPV1 receptors, PKC and K+ channels systems, but not L-arg/NO/cGMP pathway.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alves, D., Duarte, I., 2002. Involvement of ATP-sensitive K(+) channels in the peripheral antinociceptive effect induced by dipyrone. Eur. J. Pharmacol. 444, 47–52.
Aruna Sindhe, M., Bodke, Y.D., Chandrashekar, A., 2013. Antioxidant and in vivo anti-hyperglycemic activity of Muntingia calabura leaves extracts. Der Pharmacia Lettre 5, 427–435.
Astin, JA., 1998. Why patients use alternative medicine: results of a national study. J. Am. Med. Assoc. 279, 1548–1553.
Aznam, N., Atun, S., Arianingrum, R., Nurestri, S., 2012. Isolation, identification and antiviral activity of bioactive compounds of Kampheria rotunda. Int. Proc. Chem. Biol. Environ. Eng. 38, 27–30.
Balan, T., Mohd Sani, M.H., Suppaiah, V., Mohtarrudin, N., Suhaili, Z., Ahmad, Z., Zakaria, Z.A., 2014. Antiulcer activity of Muntingia calabura leaves and the possible mechanisms of action involved. Pharm. Biol. 52, 410–418.
Beirith, A., Santos, A.R.S., Calixto, J.B., 2002. Mechanisms underlying the nociception and paw oedema caused by injection of glutamate into the mouse paw. Brain Res. 924, 219–228.
Beirith, A., Santos, A.R.S., Calixto, J.B., Hess, S.C., Messana, I., Ferrari, F., Yunes, RA., 1999. Study of the antinociceptive action of the ethanolic extract and the triterpene 24-hydroxytormentic acid isolated from the stem bark of Ocotea suaveolens. Planta Med. 65, 50–55.
Chen, J.J., Lee, H.H., Duh, C.Y., Chen, I.S., 2005. Cytotoxic chalcones and flavonoids from the leaves of Muntingia calabura. Planta Med. 71, 970–973.
Chen, J.J., Lee, H.H., Shih, CD., Liao, C.H., Chen, I.S., Chou, H.T., 2007. New dihydrochal-cones and anti-platelet aggregation constituents from the leaves of Muntingia calabura. Planta Med. 73, 572–577.
Chen, Y.F., Tsai, H.Y., Wu, T.S., 1995. Antiinflammatory and analgesic activities from roots of Angelica pubescens. Planta Med. 61, 2–8.
Choi, S.S., Han, K.J., Lee, H.K., Han, E.J., Suh, H.W., 2003. Possible antinociceptive mechanisms of opioid receptor antagonists in the mouse formalin test. Pharmacol. Biochem. Behav. 75, 121–124.
Collier, H.O.J., Dinneen, J.C., Johnson, CA., Schneider, C., 1968. The abdominal constriction response and its suppression by analgesic drugs in the mouse. Br. J. Pharmacol. Chemother. 32, 295–310.
Cui, M., Honore, P., Zhong, C., Gauvin, D., Mikusa, J., Hernandez, G., Faltynek, C.R., 2006. TRPV1 receptors in the CNS play a keyrole in broad-spectrum analgesia of TRPV1 antagonists. J. Neurosci. 26, 9385–9393.
Craig, A.D., Sorkin, L.S., 2011. Pain and analgesia. In: Encyclopedia of Life Sciences. John Wiley, Chichester, England, https://doi.org/10.1002/9780470015902.a0000275.pub3.
de Vasconcelos, D.I.B., Leite, J.A., Carneiro, L.T., Piuvezam, M.R., de Lima, M.R.V., de Morais, L.C.L., Rumjanek, V.M., Rodrigues-Mascarenhas, S., 2011. Antiinflammatory and antinociceptive activity of ouabain in mice. Med. Inflamm. 2011, 1–11, https://doi.org/10.1155/2011/912925.
Deraedt, R., Jouquey, S., Delevallee, F., Flahaut, M., 1980. Release of prostaglandins E and F in an algogenic reaction and its inhibition. Eur. J. Pharmacol. 61, 17–24.
Ferreira, J., da Silva, G.L., Calixto, J.B., 2004. Contribution of vanilloid receptors to the overt nociception induced by B2 kinin receptor activation in mice. Br. J. Pharmacol. 141, 787–794.
Giglio, CA., Defino, H.LA, da-Silva, CA., de-Souza, A.S., del Bel, EA., 2006. Behavioral and physiological methods for early quantitative assessment of spinal cord injury and prognosis in rats. Braz. J. Med. Biol. Res. 39, 1613–1623.
Gomathi, R., Anusuya, N., Manian, S., 2013. A dietary antioxidant supplementation of Jamaican cherries (Muntingia calabura L.) attenuates inflammatory related disorders. Food Sci. Biotechnol. 22, 787–794.
Goncalves, CE., Araldi, D., Panatieri, R.B., Rocha, J.B., Zeni, G., Nogueira, C.W., 2005. Antinociceptive properties of acetylenicthiophene and furan derivatives: evidence forthe mechanism of action. Life Sci. 76, 2221–2234.
Holt, GA., Chandra, A., 2002. Herbs in the modern healthcare environment: an overview of uses, legalities and the role of the healthcare professional. Clin. Res. Regul. Aff. 19, 83–107.
Hosseinzadeh, H., Younesi, H.M., 2002. Antinociceptive and antiinflammatory effects of Crocus sativus L. stigma and petal extracts in mice. BMC Pharmacol. 2, 7, https://doi.org/10.1186/1471-2210-2-7.
Ikeda, Y., Ueno, A., Naraba, H., Oh-Ishi, S., 2001. Involvement of vanilloid receptor VR1 and prostanoids in the acid-induced writhing responses of mice. Life Sci. 69, 2911–2919.
Isnarianti, R., Wahyudi, I.A., Puspita, R.M., 2013. Muntingia calabura L. leaves extract inhibits glucosyltransferase activity of Streptococcus mutans. J. Dent. Indonesia 20, 59–63.
Kaneda, N., Pezzuto, J.M., Soejarto, D.D., Kinghorn, A.D., Farnsworth, N.R., Santisuk, T., Tuchinda, P., Udchachon, J., Reutrakul, V., 1991. Plant anticancer agents, XLVIII. New cytotoxic flavonoids from Muntingia calabura roots. J. Nat. Prod. 54, 196–206.
Karumi, Y., Onyeyili, P., Ogugbuaja, V.O., 2003. Anti-inflammatory and antinociceptive (analgesic) properties of Momordical balsamina Linn. (Balsam apple) leaves in rats. Pak. J. Biol. Sci. 6, 1515–1518.
Le Bars, D., Gozariu, M., Cadden, S.W., 2001. Animal models of nociception. Pharmacol. Rev. 53, 597–652.
Mahmood, D., Mamat, S.S., Kamisan, F.H., Yahya, F., Kamarolzaman, M.F.F., Nasir, N., Suhaili, Z., Mohtarrudin, N., Zakaria, Z.A., 2014. Amelioration of paracetamol-induced hepatotoxicity in rat by the administration of methanol extract of Muntingia calabura L. leaves. Biomed. Res. Int. 2014, 1–10, https://doi.org/10.1155/2014/695678.
Martínez-Vázquez, M., Estrada-Reyes, R., Martínez-Laurrabaquio, A., López-Rubalcava, C., Heinze, G., 2012. Neuropharmacological study of Dracocephalum moldovica L. (Lamiaceae) in mice: sedative effect and chemical analysis of an aqueous extract. J. Ethnopharmacol. 141, 908–917.
Melo, M.S., Santana, M.T., Guimarães, A.G., Siqueira, R.S., Sousa, D.P., Santos, M.R.V., Bonjardim, L.R., Araújo, A.A.S., Onofre, A.S.C., Lima, J.T., Almeida, J.R.G.S., Quintans-Júnior, L.J., 2011. Bioassay-guided evaluation of central nervous system effects of citronellal in rodents. Braz. J. Pharmacogn. 21, 697–703.
Millan, M.J., 1999. The induction of pain: an integrative review. Prog. Neurobiol. 57, 1–164.
Mohamad Yusof, M.I., Salleh, M.Z., Teh, L.K., Ahmat, N., NikAzmin, N.F., Zakaria, Z.A., 2013. Activity-guided isolation of bioactive constituents with antinociceptive activity from Muntingia calabura L. leaves using the formalin test. Evid. Based Complement. Altern. Med., 715074, https://doi.org/10.1155/2013/715074.
Mohd Sani, M.H., Zakaria, Z.A., Balan, T., Teh, L.K., Salleh, M.Z., 2012. Antinociceptive activity of methanol extract of Muntingia calabura leaves and the mechanisms of action involved. Evid. Based Complement. Altern. Med. 2012, 890361, https://doi.org/10.1155/2012/890361.
Mohd Sani, M.H., Taher, M., Susanti, D., The, L.K., Salleh, M.Z., Zakaria, Z.A., 2014. Mechanisms of a-mangostin-induced antinociception in a rodent model. Biol. Res. Nurs. 17, 68–77.
Musa, A.M., Aliyu, A.B., Yaro, A.H., Magaji, M.G., Hassan, H.S., Abdullahi, M.I., 2009. Preliminary phytochemical, analgesic and anti-inflammatory studies of the methanol extract of Anisopus mannii in rodents. Afr. J. Pharm. Pharmacol. 3, 374–378.
Neto Bandeira, G., da Camara, C.A.G., de Moraes, M.M., Barros, R., Muhammad, S., Akhtar, Y., 2013. Insecticidal activity of Muntingia calabura extracts against larvae and pupae of diamondback, Plutellaxylostella (Lepidoptera, Plutellidae). J. King Saud Univ. 25, 83–89.
Nivethetha, M., Jayasari, J., Brindha, P., 2009. Effects of Muntingia calabura L. on isoproterenol-induced myocardial infarction. Singapore Med. J. 50, 300–302.
Nshimo, CM., Pezzuto, J.M., Kinghorn, A.D., Farnsworth, N.R., 1993. Cytotoxic constituents of Muntingia calabura leaves and stems collected in Thailand. Int. J. Pharmacogn. 31, 77–81.
Ocana, M., Cendan, CM., Cobos, E.J., Entrena, J.M., Baeyens, J.M., 2004. Potassium channels and pain: present realities and future opportunities. Eur. J. Pharmacol. 500, 203–219.
Osikowicz, M., Mika, J., Przewlocka, B., 2013. The glutamatergic system as a target for neuropathic pain relief. Exp. Physiol. 98, 372–384.
Patel, N.K., Bhutani, K.K., 2014. Pinostrobin and cajanus lactone isolated from Cajanus cajan(L.) leaves inhibits TNF-α and IL-1ß production: in vitro and in vivo experimentation. Phytomed. 21, 946–953.
Perez-Arbelaez, E., 1975. In: Salazar, H. (Ed.), Plantas Medicinales y Venenosas de Colombia., p. 192, Medellin, Colombia.
Preethi, K., Vijayalakshmi, N., Shamna, R., Sasikumar, J.R., 2010. In vitro antioxidant activity of extracts from fruits of Muntingia calabura Linn. from India. Pharmacogn. J. 14, 11–18.
Preethi, K., Premasudha, P., Keerthana, K., 2012. Anti-inflammatory activity of Muntingia calabura fruits. Pharmacogn. J. 4, 51–56.
Quintans-Júnior, L.J., Silva, D.A., Siqueira, J.S., Araújo, A.A.S., Barreto, R.S.S., Bonjardim, LR., DeSantana, J.M., De Lucca Júnior, W., Souza, M.F.V., Gutierrez, S.J.C., Barbosa-Filho, J.M., Santana-Filho, V.J., Araújo, D.A.M., Almeida, R.N., 2010. Bioassay-guided evaluation of antinociceptive effect of N-salicyloyltryptamine: a behavioral and electrophysiological approach. J. Biomed. Biotechnol. 2010, 1–6, https://doi.org/10.1155/2010/230745.
Rates, S.M.K., 2001. Plants as source of drugs. Toxicon 29, 603–613.
Reeta, K., Mediratta, P.K., Rathi, N., Jain, H., Chugh, C., Sharma, K.K., 2006. Role of kappa and delta opioid receptors in the antinociceptive effect of oxytocin in formalin-induced pain response in mice. Regul. Pept. 135, 85–90.
Rodella, L.F., Borsani, E., Rezzani, R., 2010. New perspective in pain treatment. Open Conf. Proc. J. 1, 61–65.
Savegnago, L., Pinto, L.G., Jesse, CR., Alves, D., Rocha, J.B.T., Noguira, C.W., Zeni, G., 2007. Antinociceptive properties of diphenyldiselenide: evidences for the mechanism of action. Eur. J. Pharmacol. 555, 129–138.
Schmidtko, A., Tegeder, I., Geisslinger, G., 2009. No NO, no pain? The role of nitric oxide and cGMP in spinal pain processing. Trends Neurosci. 32, 339–346.
Shih, CD., 2009. Activation of nitric oxide/cGMP/PKG signaling cascade mediates antihypertensive effects of Muntingia calabura in anesthetized spontaneously hypertensive rats. Am. J. Chin. Med. 37, 1045–1058.
Shih, CD., Chen, J.J., Lee, H.H., 2006. Activation of nitric oxide signaling pathway mediates hypotensive effect of Muntingia calabura L. (Tiliaceae) leaf extract. Am. J. Chin. Med. 34, 857–872.
Siddiqua, A., Premakumari, K.B., Sultana, R., Savitha, V., 2010. Antioxidant activity and estimation of total phenolic content of Muntingia calabura by calorimetry. Int. J. Chem Tech. Res. 2, 205–208.
Starec, M., Waitzov’a, D., Elis, J., 1988. Evaluation of the analgesic effect of RG-tannin using the “hot plate” and “tail flick” method in mice. Cesk. Farm. 37, 319–321.
Sufian, A.S., Ramasamy, K., Ahmat, N., Zakaria, Z.A., Mohd. Yusof, M.I., 2013. Isolation and identification of antibacterial and cytotoxic compounds from the leaves of Muntingia calabura L J. Ethnopharmacol. 146, 198–204.
Su, B.N., Jung Park, E., Vigo, J.S., Graham, J.G., Cabieses, F., Fong, H.H., Pezzuto, J.M., Kinghorn, A.D., 2003. Activity-guided isolation of the chemical constituents of Muntingia calabura using a quinone reductase induction assay. Phytochemistry 63, 335–341.
Tsimogiannis, D., Samiotaki, M., Panayotou, G., Oreopoulou, V., 2007. Characterization of flavonoid subgroups and hydroxy substitution by HPLC-MS/MS. Molecules 12, 593–606.
Tuchinda, P., Reutrakul, V., Claeson, P., Pongprayoon, U., Sematong, T., Santisuk, T., Taylor, W., 2002. Anti-inflammatory cyclohexenyl chalcone derivatives in Boesenbergia pandurata. Phytochemistry 59, 169–173.
Velázquez, K.T., Mohammad, H., Sweitzer, S.M., 2007. Protein kinase C in pain: involvement of multiple isoforms. Pharmacol. Res. 55, 578–589.
Vellani, V., Mapplebeck, S., Moriondo, A., Davis, J.B., McNaughton, P.A., 2001. Protein kinase C activation potentiates gating of the vanilloid receptor VR1 by capsaicin, protons, heat and anandamide. J. Physiol. Lond. 543, 813–825.
Verma, P.R., Joharapurkar, A.A., Chatpalliwar, V.A., Asnani, A.J., 2005. Antinociceptive activity of alcoholic extract of Hemidesmus indicus R. Br. in mice. J. Ethnopharmacol. 102, 298–301.
Vogel, H.G., Vogel, W.H., 1997. Pharmacological assays. In: Drug Discovery and Evaluation. J.A. Majors Company, Lewisville, TX., USA, pp. 360–418.
Vongtau, H.O., Abbah, J., Ngazal, I.E., Kunle, O.F., Chinbo, B.A., Otsapa, P.B., Gamaniel, K.S., 2004. Antinociceptive and anti-inflammatory activity of the methanolic extract of Parinari polyandra stem bark in rats and mice. J. Ethnopharmacol. 90, 115–121.
Wirth, J.H., Craig Hudgins, J., Paice, J.A., 2005. Use of herbal therapies to relieve pain: a review of efficacy and adverse effects. Pain Manage. Nurs. 6, 145–167.
Zakaria, Z.A., Balan, T., Suppaiah, V., Ahmad, S., Jamaludin, F., 2014a. Mechanism(s) of action involved in the gastroprotective activity of Muntingia calabura. J. Ethnopharmacol. 3, 1184–1193.
Zakaria, Z.A., Fatimah, CA., Mat Jais, A.M., Zaiton, H., Henie, E.F.P., Sulaiman, M.R., Somchit, M.N., Thenamuha, M., Kasthuri, D., 2006a. The in vitro antibacterial activity of Muntingia calabura extracts. Int. J. Pharmacol. 2, 439–442.
Zakaria, Z.A., Kumar, G.H., Mohd Zaid, S.N.H., Abdul Ghani, M., Hassan, M.H., Mohd Nor Hazalin, N.A., Khamis, M.M., Devi, R.G., 2007a. Analgesic and antipyretic actions of Muntingia calabura leaves chloroform extract in animal models. Orient. Pharm. Exp. Med. 7, 34–40.
Zakaria, Z.A., Mohammad, A.M., Mohd Jamil, N.S., Rofiee, M.S., Hussain, M.K., Sulaiman, M.R., Teh, LK., Salleh, M.Z., 2011. In vitro antiproliferative and antioxidant activities of the extracts of Muntingia calabura leaves. Am. J. Chin. Med. 39, 183–200.
Zakaria, Z.A., Mohd Nor Hazalin, N.A., Mohd Zaid, S.N.H., Abdul Ghani, M., Hassan, M.H., Gopalan, H.K., Sulaiman, M.R., 2007b. Antinociceptive, anti-inflammatory and antipyretic effects of Muntingia calabura aqueous extract in animal models. J. Nat. Med. 61, 443–448.
Zakaria, Z.A., Mohd Sani, M.H., Cheema, M.S., Arifah, A.K., Teh, L.K., Salleh, M.Z., 2014b. Antinociceptive activity of methanolic extract of Muntingia calabura leaves: further elucidation of the possible mechanisms. BMC Complement. Altern. Med. 63, https://doi.org/10.1186/1472-6882-14-63.
Zakaria, Z.A., Sufian, A.S., Ramasamy, K., Ahmat, N., Sulaiman, M.R., Arifah, A.K., Zuraini, A., Somchit, M.N., 2010. In vitro antimicrobial activity of Muntigia calabura extracts and fractions. Afr. J. Microbiol. Res. 4, 304–308.
Zakaria, Z.A., Sulaiman, M.R., Mat Jais, A.M., Somchit, M.N., Kogilla, V.J., Ganesh, R., Fatimah, CA., 2006b. The antinociceptive activity of Muntingia calabura aqueous extract and the involvement of L-arginine/nitric oxide/cyclic guanosine monophosphate pathway in its observed activity in mice. Fundam. Clin. Pharmacol. 20, 365–372.
Zimmermann, M., 1983. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16, 109–110.
Acknowledgements
This study was supported by the Science Fund Research Grant (Reference no. 06-01-04-SF1127) awarded by the Ministry of Science Technology and Innovation (MOSTI), Malaysia and the Research University Grant Scheme (Reference no. 04-02-12-2019RU) from the Universiti Putra Malaysia, Malaysia. The authors thanked the Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, Malaysia for providing the facilities to carry out this study.
Author information
Authors and Affiliations
Contributions
ZAZ conceived of the study, participated in its design and helped to draft the manuscript. All authors read and approved the final manuscript. MHMS carried out the experiments and drafted the manuscript. AKA participated in the experimental design, and helped to draft the manuscript. TLK and MZS involved in the statistical analysis and manuscript preparation.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zakaria, Z.A., Sani, M.H.M., Kadir, A.A. et al. Antinociceptive effect of semi-purified petroleum ether partition of Muntingia calabura leaves. Rev. Bras. Farmacogn. 26, 408–419 (2016). https://doi.org/10.1016/j.bjp.2015.12.007
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2015.12.007