Abstract
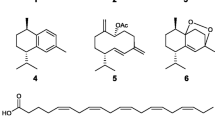
Biofilm has a primary role in the pathogenesis of diseases and in the attachment of multicellular organisms to a fouled surface. Because of that, the control of bacterial biofilms has been identified as an important target. In the present study, five lipid compounds isolated from soft coral Eunicea sp. and three terpenoids together with a mixture of sterols from Eunicea fusca collected at the Colombian Caribbean Sea showed different effectiveness against biofilm formation by three marine bacteria associated with immersed fouled surfaces, Ochrobactrum pseudogringnonense, Alteromona macleodii and Vibrio harveyi, and against two known biofilm forming bacteria, Pseudomonas aeruginosa ATCC 27853 and Staphylococcus aureus ATCC 25923. The pure compounds were characterized by NMR, HRESI-MS, HRGC-MS and optical rotation. The most effective compounds were batyl alcohol (1) and fuscoside E peracetate (6), acting against four strains without affecting their microbial growth. Compound 1 showed biofilm inhibition greater than 30% against A. macleodii, and up to 60% against O. pseudogringnonense, V. harveyi and S. aureus. Compound 6 inhibited O. pseudogringnonense and V. harveyi between 25 and 50%, and P. aeruginosa or S. aureus up to 60% at 0.5 mg/ml. The results suggest that these compounds exhibit specific biofilm inhibition with lower antimicrobial effect against the bacterial species assayed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Almeida, J.G.L., Maia, A.I.V., Wilke, D.V., Silveira, E.R., Braz-Filho, R., La Clair, J.J., Costa-Lotufo, L.V., Pessoa, O.D., 2012. Palyosulfonoceramides A and B: unique sulfonylated ceramides from the Brazilian zoanthids Palythoa caribaeorum and Protopalyhtoa variabilis. Mar. Drugs 10, 2846–2860.
Al-Lihaibi, S.S., Ayyad, S.E.N., Shaher, F., Alarif, W.M., 2010. Antibacterial sphingolipid and steroids from the black coral Antipathes dichotoma. Chem. Pharm. Bull. 58, 1635–1638.
Al-Sohaibani, S., Murugan, K., 2012. Anti-biofilm activity of Salvadorapersica on cariogenic isolates of Streptococcus mutans: in vitro and molecular docking studies. Biofouling 28, 29–38.
Beermann, Ch., Winterling, N., Green, A., Möbius, M., Schmitt, J.J., Boehm, G., 2007. Comparison ofthe structures oftriacylglycerols from native transgenic mediumchain fatty acid-enriched rape seed oil by liquid chromatography-atmospheric pressure chemical ionization ion-trap mass spectrometry (LC-APCI-ITMS). Lipids 42, 383–394.
Blunt, J.W., Copp, B.R., Keyzers, R.A., Munro, M.H.G., Prinsep, M.R., 2014. Marine natural products. Nat. Prod. Rep. 31, 160–258.
Butovich, I.A., Uchiyama, E., McCulley, J.P., 2007. Lipids of human meibum: massspectrometric analysis and structural elucidation. J. Lipid Res. 48, 2220–2235.
Castellanos, L.H., Mayorga, H.W., Duque, C.B., 2010. Study ofthe chemical composition and antifouling activity ofthe marine sponge Cliona delitrix extract. Vitae 17, 209–224.
Cuadrado, C.T.S., Castellanos, L., Arévalo-Ferro, C., Osorno, O.E., 2013. Detection of quorum sensing systems of bacteria isolated from fouled marine organisms. Biochem. Syst. Ecol. 46, 101–107.
Deniau, A.L., Mosset, P., Pédrono, F., Mitre, R., Le Bot, D., Legrand, A.B., 2010. Multiple beneficial health effects of natural alkylglycerols from shark liver oil. Mar. Drugs 8, 2175–2184.
Dobretsov, S., Abed, R.M.M., Teplitski, M., 2013. Mini-review: Inhibition of biofouling by marine microorganisms. Biofouling 29, 423–441.
Dusane, D.H., Pawar, V.S., Nancharaiah, Y.V., Venugopalan, V.P., Kumar, A.R., Zinjarde, S.S., 2011. Anti-biofilm potential ofaglycolipid surfactant produced by a tropical marine strain of Serratia marcescens. Biofouling 27, 645–654.
Fusetani, N., 2011. Antifouling marine natural products. Nat. Prod. Rep. 28, 400–410.
Han, A.R., Song, J.I., Jang, D.S., Min, H.Y., Lee, S.K., Seo, E.K., 2005. Cytotoxic constituents ofthe octocoral Dendronephthya gigantea. Arch. Pharm. Res. 28, 290–293.
Huigens, R.W., Richards, J.J., Parise, G., Ballard, T.E., Zeng, W., Deora, R., Melander, C., 2007. Inhibition of Pseudomonas aeruginosa biofilm formation with bromoageliferin analogues. J. Am. Chem. Soc. 129, 6966–6967.
Iannitti, T., Palmieri, B., 2010. An update on the therapeutic role of alkylglycerols. Mar. Drugs 8, 2267–2300.
Ivars-Martinez, E., Martin-Cuadrado, A.B., D’Auria, G., Mira, A., Ferriera, St., Johnson, J., Friedman, R., Rodriguez-Valera, F., 2008. Comparative genomics of two ecotypes ofthe marine planktonic copiotroph Alteromonas macleodii suggests alternative lifestyles associated with different kinds of particulate organic matter. ISMEJ. 2, 1194–1212.
Jadhav, S., Shah, R., Bhave, Mr., Palombo, E.A., 2013. Inhibitory activity of yarrow essential oil on Listeria planktonic cells and biofilms. Food Control 29, 125–130.
Jung, J.E., Pandit, S., Jeon, J.G., 2014. Identification of linoleic acid, a main component ofthe n-hexane fraction from Dryopteris crassirhizoma, as an anti-Streptococcus mutans biofilm agent. Biofouling 30, 789–798.
Kind, C.A., Bergmann, W., 1942. Contributions to the study of marine products. XI. The occurrence of octadecyl alcohol, batyl alcohol, and cetyl palmitate in gorgonias. J. Org. Chem. 7, 424–427.
Majik, M.S., Parvatkar, P.T., 2014. Next generation biofilm inhibitors for Pseudomonas aeruginosa: synthesis and rational design approaches. Curr. Top. Med. Chem. 14, 81–109.
Mayorga, H.W., Urrego, N.F., Castellanos, L.H., Duque, C.B., 2011. Cembradienes from the Caribbean Sea whip Eunicea sp. Tetrahedron Lett. 52, 2515–2518.
McAnoy, A.M., Wu, Ch.C., Murphy, R.C., 2005. Direct qualitative analysis oftriacylglycerols by electrospray mass spectrometry using a linear ion trap. J. Am. Soc. Mass Spectrom. 16, 1498–1509.
Montgomery, D.C., 2007. Design and Analysis of Experiments. Limusa, Mexico.
Mora-Cristancho, J.A., Arévalo-Ferro, C., Ramos, F.A., Tello, E., Duque, C., Lhullier, C., Falkenberg, M., Schenkel, E.P., 2011. Antifouling activities against colonizer marine bacteria of extracts from marine invertebrates collected in the Colombian Caribbean Sea and on the Brazilian coast (Santa Catarina). Z. Naturforsch. 66c, 515–526.
Myers, B.L., Crews, Ph., 1983. Chiral ether glycerides from a marine sponge. J. Org. Chem. 48, 3583–3585.
O’toole, G., Kaplan, H.B., Kolter, R., 2000. Biofilm formation as microbial development. Annu. Rev. Microbiol. 54, 49–79.
Qi, S.H., Zhang, S., Yang, L.H., Qian, P.Y., 2008. Antifouling and antibacterial compounds from the gorgonians Subergorgia suberosa and Scirpearia gracilis. Nat. Prod. Rep. 22, 154–166.
Quijano, L., Cruz, F., Navarrete, I., Gómez, P., Rios, T., 1994. Alkyl glycerol monoethers in the marine sponge Desmapsamma anchorata. Lipids 29, 731–734.
Quinn, G.A., Maloy, A.P., McClean, St., Carney, B., Slater, J.W., 2012. Lipopeptide biosurfactants from Paenibacillus polymyxa inhibit single and mixed species biofilms. Biofouling 28, 1151–1166.
Radhika, P., 2006. Chemical constituents and biological activities ofthe soft corals of genus Cladiella: a review. Biochem. Syst. Ecol. 34, 781–789.
Ramos, F., Takaishi, Y., Kawazoe, K., Osorio, C., Duque, C., Acuña, R., Fujimoto, Y., Sato, M., Okamoto, M., Oshikawa, T., Ahmed, Sh.U., 2006. Immunosuppressive diacetylenes, ceramides and cerebrosides from Hydrocotyle leucocephala. Phytochemistry 67, 1143–1150.
Raut, J.S., Shinde, R.B., Chauhan, N.M., Karuppayil, S.M., 2013. Terpenoids of plant origin inhibit morphogenesis, adhesion, and biofilm formation by Candida albicans. Biofouling 29, 87–96.
Reina, E., Puentes, C., Rojas, J., García, J., Ramos, F.A., Castellanos, L, Aragón, M., Ospina, L.F., Fuscoside, E., 2011. A strong antiinflammatory diterpene from Caribbean octocoral Eunicea fusca. Bioorg. Med. Chem. Lett. 21, 5888–5891.
Seo, Ch.W., Yamada, Y., Okada, H., 1982. Synthesis of fatty acid esters by Corynebacterium sp. S-401. Agric. Biol. Chem. 46, 405–409.
Shin, J., Fenical, W., 1991. Fuscosides A-D: antiinflammatory diterpenoid glycosides of new structural classes from the Caribbean gorgonian Eunicea fusca. J. Org. Chem. 56, 3153–3158.
Shin, J., Seo, Y.W., 1995. Isolation of new ceramides from the gorgonian Acabaria undulata. J. Nat. Prod. 58, 948–953.
Shinzawa-Itoh, K., Aoyama, H., Muramoto, K., Terada, H., Kurauchi, Ts., Tadehara, Y., Yamasaki, A., Sugimura, T., Kurono, S., Tsujimoto, K., Mizushima, Ts., Yamashita, E., Tsukihara, T., Yoshikawa, Sh., 2007. Structures and physiological roles of 13 integral lipids of bovine heart cytochrome c oxidase. EMBO J. 26, 1713–1725.
Stamatov, St.D., Stawinski, J., 2007. Regioselective and stereospecific acylation across oxiraneand silyloxy systems as a novel strategy to the synthesis of enantiomerically pure mono-, di- and triglycerides. Org. Biomol. Chem. 5, 3787–3800.
Sun, X.F., Wu, Q., Wang, N., Cai, Y., Lin, X.F., 2005. Novel mutual pro-drugs of 2;3-dideoxyinosine with 3-octadecyloxy-propane-1, 2-diol by straightforward enzymatic regioselective synthesis in acetone. Biotechnol. Lett. 27, 113–117.
Tello, E., Castellanos, L, Arevalo-Ferro, C., Rodríguez, J., Jimenez, C., Duque, C., 2011. Absolute stereochemistry of antifouling cembranoid epimers at C-8 from the Caribbean octocoral Pseudoplexaura flagellosa. Revised structures of plexaurolones. Tetrahedron 67, 9112–9121.
Tello, E., Castellanos, L., Arévalo-Ferro, C., Duque, C., 2012. Disruption in quorumsensing systems and bacterial biofilm inhibition by cembranoid diterpenes isolated from the octocoral Eunicea knighti. J. Nat. Prod. 75, 1637–1642.
Ved, H.S., Gustow, E., Mahadevans, V., Pieringer, R.A., 1984. Dodecylglycerol a new type of antibacterial agent which stimulates autolysin activity in Streptococcus faecium ATCC 9790. J. Biol. Chem. 259, 8115–8121.
Viano, Y., Bonhomme, D., Camps, M., Briand, J.F., Ortalo-Magné, A., Blache, Y., Piovetti, L., Culioli, G., 2009. Diterpenoids from the Mediterranean brown alga Dictyota sp. evaluated as antifouling substances against a marine bacterial biofilm. J. Nat. Prod. 72, 1299–1304.
Vieville, C., Mouloungui, Z., Gaset, A., 1995. Synthesis and analysis of the C1-C18 alkyl oleates. Chem. Phys. Lipids 75, 101–108.
Vikram, A., Jesudhasan, P.R., Jayaprakasha, G.K., Pillai, S.D., Patil, Bh.S., 2011. Citrus limonoids interfere with Vibrio harveyi cell-cell signalling and biofilm formation by modulating the response regulator LuxO. Microbiology 157, 99–110.
Watschinger, K., Werner, E.R., 2013. Orphan enzymes in ether lipid metabolism. Biochimie 95, 59–65.
Wilkins, T.D., Holdeman, L.V., Abramson, I.J., Moore, W.E.C., 1972. Standardized single-disc method for antibiotic susceptibility testing of anaerobic bacteria. Antimicrob. Agents Chemother. 1, 451–459.
Yang, J.L., Li, X., Liang, X., Bao, W.Y., Shen, H.D., Li, J.L., 2014. Effects of natural biofilms on settlement of plantigrades ofthe mussel Mytilus coruscus. Aquaculture, 424-425, 228–233.
Acknowledgment
This work was supported by Universidad Nacional de Colombia [grant number 5815/2012 and 20406/2014], and Colciencias [grant number 1101-521-28468/2011]. The Ministerio de Ambiente, Vivienda y Desarrollo Territorial granted permission for research on marine organisms [No. 4 of 10/02/2010, and No. 0306 of 22/02/2011]. The authors are grateful to professors Sven Zea, Monica Puyana and Edisson Tello.
Author information
Authors and Affiliations
Contributions
YRMD (MSc student) performed inhibitions assays and biofilm formation. GPVL and LERG were responsible for running the experimental work. HMW supervised the laboratory work and contributed to preparation of the paper. CAF supervised the laboratory work and contributed to the biological studies. FARR supervised the laboratory work and contributed to critical reading of the manuscript. CDB contributed with study design and supervised the research project. LCH supervised the research project as head of the research team, and contributed to critical reading of the manuscript. All the authors have read the final manuscript and approved its submission.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Díaz, Y.M., Laverde, G.V., Gamba, L.R. et al. Biofilm inhibition activity of compounds isolated from two Eunicea species collected at the Caribbean Sea. Rev. Bras. Farmacogn. 25, 605–611 (2015). https://doi.org/10.1016/j.bjp.2015.08.007
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2015.08.007