Abstract
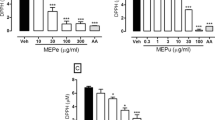
Passiflora alata Curtis, Passifloraceae, is a liana popularly known in Brazil as ‘maracujá-doce’ that has been used for treating different illnesses. Its leaves are described in the Brazilian Pharmacopoeia, but the gastroprotective activity has never been investigated. In the present study a freeze-dried crude 60% ethanol–water extract of P. alata aerial parts was prepared. Total flavonoid content, expressed as vitexin, was 0.67% ±0.01. The hemolytic activity was 32 units for P. alata, using Saponin (Merck®) as reference. P. alata presented EC50 of 1061.2 ±8.5 μg/ml in the 2,2-diphenyl-1-picryhydrazyl assay and 1076 ±85 μmol Trolox/g in the Oxygen Radical Absorbance Capacity assay. P. alata, its solvent fractions and a P. alata nanopreparation were investigated for gastroprotective activity. The test samples exhibited gastroprotective activity on HCl/ethanol induced gastric mucosal lesions in rats. P. alata at doses of 100, 200 and 400 mg/kg, using the necrotizing agent at 150 mmol/l, inhibited 100% of ulcer formation (compared to the negative control), while lansoprazole (30 mg/kg) 77%. When tested against a more concentrated necrotizing agent (300 mmol/l), fractions of P. alata at 100 mg/kg reduced 57% (n-hexane), 34% (ethyl acetate) and 72% (aqueous fraction) the ulcer formation. In this assay, lansoprazole (30 mg/kg) inhibited 47%. When encapsulated, P. alata inhibited ulcer formation at 55%, 94% and 90% for dosages of 25, 50 and 100 mg/kg. These results suggest the potential use of P. alata as a gastroprotective herbal medicine.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amaral, K.M., Schenkel, E.P., Langeloh, A., 2001. Avaliação da toxicidade reprodutiva dos extratos aquosos liofilizados de Passiflora alata Dryander e Passiflora edulis Sims em ratas Wistar. Lat. Am. J. Pharm. 20. 215–220.
Baggio, C.H., Freitas, C.S., Otofuji, G.M., Cipriani, T.R., Souza, L.M., Sassaki, G.L., Iacomini, M., Marques, M.C.A., Mesia-Vela, S., 2007. Flavonoid-rich fraction of Maytenus ilicifolia Mart. ex Reiss. protects the gastric mucosa of rodents through inhibition of both H+, K+- ATPase activity and formation of nitric oxide. J. Ethnopharmacol. 113. 433–440.
Bernacci, L.C., Cervi, A.C., Milward-de-Azevedo, M.A., Nunes, T.S., Imig, D.C., Mezzonato, A.C., 2015. Passifloraceae in Lista de Espécies da Flora do Brasil. Jardim Botânico do Rio de Janeiro, http://reflora.jbrj.gov.br/jabot/floradobrasil/FB12506 (accessed May 2015).
Birk, C.D., Provensi, G., Reginatto, F.H., Schenkel, E.P., Gosmann, G., 2005. TLC fingerprints of flavonoids and saponins 2285–2291.
Blumenthal, M. (Ed.), 1998. The Complete German Commission E Monographs. American Botanical Council, Austin/Integrative Medicine Communications, Boston.
Boeira, J.M., Fenner, R., Betti, A.H., Provensi, G., Lacerda, L., De, A., Barbosa, P.R., Gonzalez, F.H.D., Correa, A.M.R., Driemeier, D., Dall’alba, M.P., Pedroso, A.P., Gosmann, G., Da Silva, J., Rates, S.M.K., 2010. Toxicity and genotoxicity evaluation of Passiflora alata Curtis (Passifloraceae). J. Ethnopharmacol. 128. 526–532.
Bonifácio, B.V., da Silva, P.B., dos Santos Ramos, M.A., Negri, K.M.S., Bauab, T.M., Chorilli, M., 2014. Nanotechnology-based drug delivery systems and herbal medicines: a review. Int. J. Nanomed. 9. 1.
Braga, A., Stein, A.C., Stolz, E.D., Dallegrave, E., Buffon, A., Rego, J-C., Gosmann, G., Viana, A.F., Rates, S.M.K., 2013. Repeated administration of an aqueous spraydried extract of the leaves of Passiflora alata Curtis (Passifloraceae) inhibits body weight gain without altering mice behavior. J. Ethnopharmacol. 145. 59–66.
Brunton, L.L. (Ed.), 2006. Goodman & Gilman’s The pharmacological Basis of Therapeutics. 11th ed. McGraw-Hill, New York.
Colomeu, T.C., Figueiredo, D., Cazarin, C.B.B., Schumacher, N.S.G., Maróstica Jr., M.R., Meletti, L.M.M., Zollner, R.L., 2014. Antioxidant and anti-diabetic potential of Passiflora alata Curtis aqueous leaves extract in type 1 diabetes mellitus (NODmice). Int. Immunopharmacol. 18. 106–115.
Dai, F., Miao, Q., Zhou, B., Yang, L., Liu, Z.L., 2006. Protective effects of flavonols and their glycosides against free radical-induced oxidative hemolysis of red blood cells. Life Sci. 78. 2488–2493.
De-Paris, F., Petry, R.D., Reginatto, F., Gosmann, G., Quevedo, J., Salgueiro, J.B., Kapczinski, F., González-Ortega, G., Schenkel, E.P., 2002. Pharmacochemical study of aqueous extracts of Passiflora alata Dryander and Passiflora edulis Sims. Lat. Am. J. Pharm. 21. 5–8.
Doyama, J.T., Rodrigues, H.G., Novelli, E.L.B., Cereda, E., Vilegas, W., 2005. Chemical investigation and effects of the tea of Passiflora alata on biochemical parameters in rats. J. Ethnopharmacol. 96. 371–374.
Dudonné, S., Vitrac, X., Coutière, P., Woillez, M., Mérillon, J-M., 2009. Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J. Agric. Food Chem. 57. 1768–1774.
Duke, J.A., 2002. Handbook of Medicinal Herbs, 2nd ed. CRC Press, Boca Raton.
European Medicines Agency (EMA), 2014. Committee for herbal medicinal products. Assessment report on Passiflora incarnata L., herba, http://www.ema.europa.eu/ema (accessed March 2015).
2011. European Pharmacopoeia, 7th ed. Council of Europe, Strasbourg.
1977. Farmacopeia Brasileira. Organização Andrei Editora S.A., São Paulo.
Farmacopeia Brasileira, vol. 2., 5th ed. Agência Nacional de Vigilância Sanitária, Brasília, http://www.anvisa.gov.br/hotsite/cd_farmacopeia/index.htm (accessed 5.05.15).
1959. Farmacopeia dos Estados Unidos do Brasil. Gráfica Siqueira, São Paulo.
Glavin, G.B., Szabo, S., 1992. Experimental gastric mucosal injury: laboratory models reveal mechanisms of pathogenesis and new therapeutic strategies. FASEB J. 6. 825–831.
Gosmann, G., Provensi, G., Comunello, L.N., Rates, S.M.K., 2011. Composição química e aspectos farmacológicos de espécies de Passiflora L. (Passifloraceae). Rev. Bras. Biocienc. 9. 88–99.
Gracioso, J.S., Vilegas, W., Hiruma-Lima, C.A., Souza Brito, A.R.M., 2002. Effects of tea from Turnera ulmifolia L. on mouse gastric mucosa support the Turneraceae as a new source of antiulcerogenic drugs. Biol. Pharm. Bull. 25. 487–491.
Gruenwald, J.G., Brendler, T., Jaenicke, C. (Eds.), 2000. PDR for Herbal Medicines. 2nd ed. Medical Economics Company, Montvale.
Harborne, J.B., 1978. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis. Chapman and Hall, London.
Hostettmann, K., Marston, A., 1995. Saponins. Cambridge University Press, Cambridge.
Huang, D., Ou, B., Hampch-Woodill, M., Flanagan, J.A., Prior, R., 2002. Highthroughput assay of oxygen radical aborbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 50. 4437–4444.
Inatomi, N., et al., 1991. Effects of a proton pump inhibitor, AG-1749 (lansoprazole), on reflux esophagitis and experimental ulcers in rats. Jpn. J. Pharmacol. 55. 437–451.
Isbrucker, R.A., Burdock, G.A., 2006. Risk and safety assessment on the consumption of Licorice root (Glycyrrhiza sp.), its extract and powder as a food ingredient, with emphasis on the pharmacology and toxicology of glycyrrhizin. Regul. Toxicol. Pharm. 46. 167–192.
Kawano, S., Tsuji, S., 2000. Role of mucosal blood flow: a conceptional review in gastric mucosal injury and protection. J. Gastroenterol. Hepatol. 15. 1–6.
Klein, N., Gazola, A.C., Lima, T.C.M., Schenkel, E., Nieber, K., Butterweck, V., 2014. Assessment of sedative effects of Passiflora edulis f. flavicarpa and Passiflora alata extracts in mice, measured by telemetry. Phytother. Res. 28. 706–713.
Kwiecien´, S., Brzozowski, T., Konturek, S.J., 2002. Effects of reactive oxygen species action on gastric mucosa in various models of mucosal injury. J. Physiol. Pharmacol. 53. 39–50.
Leontiadis, G.I., Nyrén, O., 2014. Epidemiology of Helicobacter pylori infection, peptic ulcer disease and gastric cancer. In: Talley, N.J., Locke, G.R., Moayyedi, P., West, J., Ford, A.C., Saito, Y.A. (Eds.), GI Epidemiology: Diseases and Clinical Methodology. 2nd ed. Wiley Blackwell, Oxford, pp. 135–157.
Lorenzi, H., Sartori, S.F., Bacher, L.B., Lacerda, M.T.C., 2006. Frutas Brasileiras e Exóticas Cultivadas. Plantarum, Nova Odessa.
Mayer, C., 2005. Nanocapsules as drug delivery systems. Int. J. Artif. Organs 28. 1163–1171.
Mensor, L.L., Menezes, F.S., Leitão, G.G., Reis, A.S., Santos, T.C., Coube, C.S., Leitão, S.G., 2001. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother. Res. 15. 127–130.
Mizui, T., Doteuchi, M., 1983. Effect of polyamines on acidified ethanol-induced gastric lesion in rats. Jpn. J. Pharmacol. 33. 939–945.
Mora-Huertas, C.E., Fessi, H., Elaissari, A., 2010. Polymer-based nanocapsules for drug delivery. Int. J. Pharm. 385. 113–142.
Morikawa, T., Li, N., Nagatomo, A., Matsuda, H., Li, X., Yoshikawa, M., 2006. Triterpene saponins with gastroprotective effects from tea seed (seeds of Camellia sinensis). J. Nat. Prod. 69. 185–190.
Müller, S.D., Vasconcelos, S.B., Coelho, M., Biavatti, M.W., 2005. LC and UV determination of flavonoids from Passiflora alata medicinal extracts and leaves. J. Pharm. Biomed. Anal. 37. 399–403.
Noriega, P., Mafud, D.F., Strasser, M., Kato, E.T.M., Bacchi, E.M., 2011. Passiflora alata Curtis: a Brazilian medicinal plant. Bol. Latinoam. Caribe 10. 398–413.
Oga, S., Freitas, P.C.D., Silva, A.C.G., Hanada, S., 1984. Pharmacological trials of crude extracts of Passiflora alata. Planta Med. 51. 303–306.
Okawa, M., Kinjo, J., Nohara, T., Ono, M., 2001. DPPH (1,1-diphenyl-2-picrylhydrazyl) radical scavenging activity of flavonoids obtained from some medicinal plants. Biol. Pharm. Bull. 24. 1202–1205.
Petry, R.D., Reginatto, F.H., De-Paris, F., Gosmann, G., Salgueiro, J.B., Quevedo, J., Kapczinski, F., Ortega, G.G., Schenkel, E.P., 2001. Comparative pharmacological study on hydroethanol extracts of Passiflora alata and Passiflora edulis leaves. Phytother. Res. 15. 162–164.
Plotze, R.O., Falvo, M., Padua, J.G., Bernacci, L.C., Vieira, M.L.C., Oliveira, G.C.X., Bruno, O.M., 2005. Leaf shape analysis using the multiscale Minkowski fractal dimension, a new morphometric method: a study with Passiflora (Passifloraceae). Can. J. Bot. 83. 287–301.
Pohlmann, A.R., 2013. Poly (ε-caprolactone) microcapsules and nanocapsules in drug delivery. Expert Opin. Drug Deliv. 10. 623–638.
Prior, R.L., Hoang, H., Gu, L., Wu, X., Bacchiocca, M., Howard, L., Hampsch-Woodill, M., Huang, D., Ou, B., Jacob, R., 2003. Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORAC)) of plasma and other biological and food samples. J. Agric. Food Chem. 51. 3273–3279.
Priya, G., Parminder, N., Jaspreet, S., 2012. Oxidative stress induced ulcer protected by natural antioxidants: a review. Int. Res. J. Pharm. 3. 76–81.
Provensi, G., Noel, F., Lopes, D.V.S., Fenner, R., Betti, A.H., Costa, F., Morais, E.C., Gosmann, G., Rates, S.M.K., 2008. Participation of GABA-benzodiazepine receptor complex in the anxiolytic effect of Passiflora alata Curtis (Passifloraceae). Lat. Am. J. Pharm. 27. 845–851.
Reginatto, F.H., De-Paris, F., Petry, R.D., Quevedo, J., González-Ortega, G., Gosmann, G., Schenkel, E.P., 2006. Evaluation of anxiolytic activity of spray dried powders of two south Brazilian Passiflora species. Phytother. Res. 20. 348–351.
Reginatto, F.H., Gosmann, G., Schripsema, J., Schenkel, E.P., 2004. Assay of quadranguloside, the major saponin of leaves of Passiflora alata, by HPLC–UV. Phytochem. Anal. 15. 195–197.
Reginatto, F.H., Kauffmann, C., Schripsema, J., Guillaue, D., Gosmann, G., Schenkel, E.P., 2001. Steroidal and triterpenoidal glucosides from Passiflora alata. J. Braz. Chem. Soc. 12. 32–36.
Rocha, T.D., de Brum Vieira, P., Gnoatto, S.C., Tasca, T., Gosmann, G., 2012. Anti- Trichomonas vaginalis activity of saponins from Quillaja, Passiflora, and Ilex species. Parasitol. Res. 110. 2551–2556.
Saitoh, T., Kinoshita, T., Shibata, S., 1976. New isoflavan and flavanone from Licorice root. Chem. Pharm. Bull. 24. 752–755.
Satoh, H., 2013. Discovery of lansoprazole and its unique pharmacological properties independent from anti-secretory activity. Curr. Pharm. Des. 19. 67–75.
Satoh, H., Inatomi, N., Nagaya, H., Inada, I., Nohara, A., Nakamura, N., Maki, Y., 1989. Antisecretory and antiulcer activities of a novel proton pump inhibitor AG-1749 in dogs and rats. J. Pharmacol. Exp. Ther. 248. 806–815.
Silva, R.A.D., 1926. Pharmacopeia dos Estados Unidos do Brasil. Companhia Editora Nacional, São Paulo.
Sinha, V.R., et al., 2004. Poly-ε-caprolactone microspheres and nanospheres: an overview. Int. J. Pharm. 278. 1–23.
Sung, J.J.Y., Kuipers, E.J., El-Serag, H.B., 2009. Systematic review: the global incidence and prevalence of peptic ulcer disease. Aliment. Pharmacol. Ther. 29. 938–946.
Ulubelen, A., Oksuz, S., Mabry, T.J., Dellamonica, G., Chopin, J., 1982. Cglycosylflavonoids from Passiflora pittieri, Passiflora alata, Passiflora ambigua and Adenia mannii. J. Nat. Prod. 45. 783.
Vargas, A.J., Geremias, D.S., Provensi, G., Fornari, P.E., Reginatto, F.H., Gosmann, G., Schenkel, E.P., Fröde, T.S., 2007. Passiflora alata and Passiflora edulis spray-dried aqueous extracts inhibited inflammation in mouse model of pleurisy. Fitoterapia 78. 112–119.
Vasić, S.M., Stefanović, O.D., Liˇcina, B.Z., Radojević, I.D., ˇComić, L.R., 2012. Biological activities of extracts from cultivated granadilla Passiflora alata. EXCLI J. 11. 208–218.
World Health Organization, 2011. Quality Control Methods for Medicinal Plant Materials. WHO Library, Geneva, Switzerland.
Yamaguchi, F., Saito, M., Ariga, T., Yoshimura, Y., Nakazawa, H., 2000. Free radical scavenging activity and antiulcer activity of garcinol from Garcinia indica fruit rind. J. Agric. Food Chem. 48. 2320–2325.
Yuldasheva, L.N., Carvalho, E.B., Catanho, M.-T.J.A., Krasilnikov, O.V., 2005. Cholesterol-dependent hemolytic activity of Passiflora quadrangularis leaves. Braz. J. Med. Biol. Res. 38. 1061–1070.
Acknowledgments
This project (number 04/07933-3) was supported by FAPESP. A. Wasicky thanks CNPq for scholarship (134740/2005-0) during the period of his M.Sc. The authors thank M.O.M. Marques, L.M.M. Melletti, L.C. Bernacci and M.D.S. Scott (Instituto Agronômico de Campinas) for the plant material used in this research and I.M. Cuccovia for the nanoparticles analysis by Zetasizer.
Author information
Authors and Affiliations
Contributions
MY is the coordinator of the Passiflora project; PRHM, EMB and ETMK are participants of the Passiflora project; AW and AV-N conducted the experimental study; LSH is the main responsible for the organization and writing of the manuscript.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wasicky, A., Hernandes, L.S., Vetore-Neto, A. et al. Evaluation of gastroprotective activity of Passiflora alata. Rev. Bras. Farmacogn. 25, 407–412 (2015). https://doi.org/10.1016/j.bjp.2015.07.011
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2015.07.011