Abstract
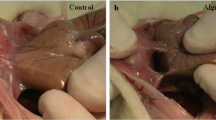
Intra-abdominal adhesions form in more than 90% of patients undergoing major abdominal surgery and can lead to significant complications. Application of a bioresorbable gel consisting of chemically modified hyaluronic acid (HA) and carboxymethylcellulose (CMC) has shown promise as a means of preventing intra-abdominal adhesions, but there have been concerns that the presence of the gel might interfere with the integrity and healing of bowel anastomoses. We tested the effects of HA/CMC gel on adhesion formation and anastomotic healing in 60 New Zealand white rabbits after transection and complete (100%) or incomplete (90%) anastomosis of the ileum. Half of the animals underwent application of HA/CMC gel and half served as control subjects. Animals were killed at 4, 7, or 14 days after surgery. Anastomotic adhesions were scored in a blinded fashion. Integrity of the anastomosis was tested by measuring bursting pressure at the anastomotic site and in an adjacent section of intact bowel. With complete anastomosis, HA/CMC gel significantly reduced adhesion formation at 7 and 14 days after surgery (P<0.05), but gel application did not inhibit adhesion formation when the anastomosis was incomplete. Anastomosed segments of bowel burst at a lower pressure than intact bowel 4 days after surgery, but bursting pressures were normal at 7 and 14 days. Burst pressures of anastomoses receiving an application of HA/CMC gel were nearly identical to control anastomoses at all three time points. HA/CMC gel did not interfere with the normal healing process of bowel anastomoses. Furthermore, HA/CMC gel decreased adhesion formation after complete anastomoses, yet it did not affect adhesion formation in the presence of anastomotic disruption.
Similar content being viewed by others
References
Becker JM, Dayton MT, Fazio VW, et al. Sodium hyaluronate-based bioresorbable membrane (HAL-F) in the prevention of postoperative abdominal adhesions: A prospective, randomized, double-blinded multicenter study. J Am Coll Surg 1996;183:297–307.
The Operative Laparoscopy Study Group. Postoperative adhesion development after operative laparoscopy: Evaluation at early second-look procedures. Fertil Steril 1991;55:700–704.
Monk BJ, Berman ML, Montz FJ. Adhesions after extensive gynecological surgery: Clinical significance, etiology, and prevention. Am J Obstet Gynecol 1994;170:1396–1403.
Stewart RM, Page CP, Brender J, et al. The incidence and risk of early postoperative small bowel obstruction: A cohort study. Am J Surg 1987;154:643–647.
di Zerega GS. Contemporary adhesion prevention. Fertil Steril 1994;61:219–235.
Diamond M. The Seprafilm Adhesion Group. Seprafilm (HAL-F) reduces postoperative adhesions: Initial results of a multicenter gynecologic clinical study. Presented at the Fourth Congress of the European Society for Gynecological Endoscopy. Brussels: December, 1995.
Burgess LS, Rose RL, Colt MJ, et al. The evaluation of an injectable bioresorbable gel on adhesion reduction in two animal models. Presented at the Third International Congress on Pelvic Surgery and Adhesion Prevention. San Diego: March, 1996.
Holzman S, Connolly RJ, Schwaitzberg SD. The effects of hyaluronic solution on healing of bowel anastomoses. J Invest Surg 1994;7:431–437.
Peritoneal adhesiolysis. National Inpatient Profile 1993: Baltimore, HICA Inc., 1994;427:635–655.
Hertzler AE. The Peritoneum. St. Louis: CV Mosby, 1919, pp 20–22.
Raftery AT. Regeneration of parietal and visceral peritoneum: An electron microscopical study. J Anat 1973;115:375–392.
Raftery AT. Regeneration of peritoneum: A fibrinolytic study. J Anat 1979;129:659–664.
Thompson JN, Whawell SA. Pathogenesis and prevention of adhesion formation, Br J Surg 1995;82:3–5.
Whawell SA, Thompson JN. Cytokine induced release of plasminogen activator inhibitor-1 by mesothelial cells. Eur J Surg 1995;161:315–317.
Stone K. Adhesions in gynecologic surgery. Curr Opin Obstet Gynecol 1993;5:322–327.
Stangel JJ, Nisbett JD, Settles H. Formation and prevention of postoperative abdominal adhesions. J Reprod Med 1984; 29:143–156
Nishimura K, Shimanuki T, di Zerega GS. Ibuprofen in the prevention of experimentally induced postoperative adhesion. Am J Med 1984;77:102–106.
Jansen RPS. Failure of peritoneal irrigation with heparin during pelvic operations upon young women to reduce adhesions. Surg Gynecol Obstet 1988;166:154–160.
Adhesion Study Group. Reduction of postoperative pelvic adhesions with intraperitoneal 32% dextran 70: A prospective, randomized clinical trial. Fertil Steril 1983;40:612–619.
Rosenberg SM, Board JA. High molecular weight dextran in human infertility surgery. Am J Obstet Gynecol 1984;148:380–385.
Interceed (TC7) Adhesion Barrier Study Group. Prevention of postsurgical adhesions by Interceed (TC7), an absorbable adhesion barrier: A prospective, randomized multicenter clinical study. Fertil Steril 1989;51:933–938.
Interceed (TC7) Adhesion Barrier Study Group 2. Pelvic sidewall adhesion reformation: Microsurgery alone or with Interceed absorbable adhesion barrier. Surg Gynecol Obstet 1993;177:135–139.
Sekiba K. The Obstetrics and Gynecology Adhesion Prevention Committee. Use of Interceed (TC7) absorbable adhesion barrier to reduce postoperative adhesion reformation in infertility and endometriosis surgery. Obstet Gynecol 1992;79:518–522.
The Surgical Membrane Study Group. Prophylaxis of pelvic sidewall adehsions with Gore-Tex Surgical Membrane: A multicenter clinical investigation. Fertil Steril 1991;57:921–923.
McCabe-Fowler JD, Lacy SM, Montz FJ. The inability of Gore-Tex Surgical Membrane to inhibit post-radical pelvic surgery adhesions in the dog model. Gynecol Oncol 1991; 43:141–144.
Skinner KC, Colt MJ, Kirk JF, et al. HAL-F film: A new hyaluronic acid based bioresorbable barrier for the prevention of abdominal adhesion [abstr]. J Invest Surg 1991;4:381.
Comper WD, Laurent TC. Physiologic function of connective tissue polysaccharides. Physiol Rev 1978;58:255–315.
Fredericks CM, Kotry I, Holz G, et al. Adhesion prevention in the rabbit with sodium carboxymethylcellulose solutions. Am J Obstet Gynecol 1986;155:667–670.
Ryan CK, Sax HC. Evaluation of a carboxymethylcellulose sponge for prevention of postoperative adhesions. Am J Surg 1995;169:154–160.
Ganong WF. Review of Medical Physiology, 13th ed. New York: Appleton & Lange, 1987, pp 483–484.
Christensen H, Langfert S, Laurberg S. Bursting strength of experimental colonic anastomosis. Eur Surg Res 1993;25:38–45.
Hesp FL, Hendriks T, Lubbers EJ, de Boer HH. Wound healing in the intestinal wall. Effects of infection on experimental ileal and colonic anastomoses. Dis Colon Rectum 1984; 27:462–467.
Ahrendt GM, Gardner K, Barbul A. Loss of colonic structural collagen impairs healing during intra-abdominal sepsis. Arch Surg 1994;129:1179–1183.
Ryan GB, Gruberty J, Majno G. Postoperative peritoneal adhesions. Am J Pathol 1971;65:117–148.
Diamond MP, Daniell JF, Feste J, et al. Adhesion reformation and de nova reformation after pelvic reproductive surgery. Fertil Steril 1987;47:864–866.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hadaegh, A., Burns, J., Burgess, L. et al. Effects of hyaluronic acid/Carboxymethylcellulose gel on bowel anastomoses in the New Zealand white rabbit. J Gastrointest Surg 1, 569–575 (1997). https://doi.org/10.1016/S1091-255X(97)80074-1
Issue Date:
DOI: https://doi.org/10.1016/S1091-255X(97)80074-1