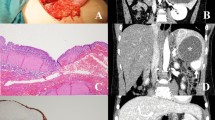
Abstract
Splenic hemangioma is a rare disorder but remains the most common benign neoplasm of the spleen. It often has a latent clinical picture; however, spontaneous rapture has been reported to occur in as many as 25% of this patient population.1 Treatment most often consists of splenectomy. This report reviews an 8-year experience with splenic hemangioma at Mayo Clinic. Thirty-two patients were identified with SH during the 8-year study period. The average age was 63 years (range 23 to 94 years) with 17 women and 15 men. Six patients presented with symptoms potentially related to the SH. The remainder (80%) were asymptomatic, and the SH was discovered incidentally during evaluation for other disorders. A mass or palpable spleen was appreciated in only four patients (12.5%). SHs ranged in size from 0.3 to 7 cm maximum diameter. A diagnosis of SH was made in 11 patients based on the findings of a splenic mass on computed tomography or ultrasound. Each of these SHs was ≤4 cm. Three of the 11 patients had multiple SHs. All 11 patients were managed successfully with observation. All but one of the patients remains asymptomatic, and no complications have developed during follow-up (range 0.6 to 7 years, mean 2.9 years). The diagnosis of splenic hemangioma was made at the time of surgery in the remaining 21 patients (65%). Splenectomy was performed for suspicion of primary or secondary splenic pathology. There were no instances of spontaneous rupture of the SH. Small splenic lesions, which meet the radiologie criteria for hemangiomas, may be safely observed.
Similar content being viewed by others
References
Hodge GB Jr. Angioma cavernosum of the spleen. Med Rec 1895;48:418.
Husni EA. The clinical course of splenic hemangioma with emphasis of spontaneous rupture. Arch Surg 1961;83:681–688.
Edoute Y, Ben-Haim SA, Ben-Arie Y, Fishman A, Barzilai D. Difficult preoperative diagnosis of a patient with sclerosing splenic hemangioma. Am J Gastroenterol 1989;84:817–819.
Goerge C, Schwerk W, Goerg K. Splenic lesions: Sono- graphic patterns, follow-up, differential diagnosis. Eur J Ra- diol 1991;13:59–66.
Kaplan J, Mcintosh GS. Spontaneous rupture of a splenic, vas- cular malformation. J R Coll Surg Edinb 1987;32:346–347.
Arber DA, Strickler JG, Chen YY, Weiss LM. Splenic vascu- lar tumors: A histologie, immunophenotypic, and virologie study. AmJ Surg Pathol 1997;21:827–83S.
Tang JS, Wu CC, Liu TJ, Wu TC, Yang MD, Hwang CR. Spontaneous rupture of splenic hemangioma: A case report. ChinMedJ 1993;51:241–243.
Ros PR, Moser RP Jr, Cadhman AH, Murari PJ, Olmsted WWL. Hemangioma of the spleen: Radiologie-pathologie correlation in ten cases. Radiology 1987;162:73–77.
Niizawa M, Ishida H, Morikawa P, Naganuma H, Masamune O. Color Doppler sonography in a case of splenic heman- gioma: Value of compressing the tumor. AJR Am J Roentgenol 1991;157:965–966.
Disler DG, Chew FS. Splenic hemangioma. AJR Am J Roentgenol 1991;157:44.
Pietrabissa A, Giulianotti P, Campatelli A, Di Candio G, Fa-rina F, Signori S, Mosca F. Management and follow-up of 78 giant hemangiomas of the liver. Br J Surg 1996;83:915–918.
Bostick WL. Primary splenic neoplasms. Division of Pathology, University of California Medical School, February 12,1945.
Wick MR, Scheithaur BW, Smith SL, Beart RW Jr. Primary nonlymphoreticular malignant neoplasms of the spleen. Am J Surg Pathol 1982;6:229–242.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Willcox, T.M., Speer, R.W., Schlinkert, R.T. et al. Hemangioma of the spleen: Presentation, diagnosis, and management. J Gastrointest Surg 4, 611–613 (2000). https://doi.org/10.1016/S1091-255X(00)80110-9
Issue Date:
DOI: https://doi.org/10.1016/S1091-255X(00)80110-9