Abstract
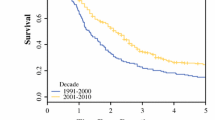
This large-volume, single-institution review examines factors influencing long-term survival after resection in patients with adenocarcinoma of the head, neck, uncinate process, body, or tail of the pancreas. Between January 1984 and July 1999 inclusive, 616 patients with adenocarcinoma of the pancreas underwent surgical resection. A retrospective analysis of a prospectively collected database was performed. Both univariate and multivariate models were used to determine the factors influencing survival. Of the 616 patients, 526 (85%) underwent pancreaticoduodenectomy for adenocarcinoma of the head, neck, or uncinate process of the pancreas, 52 (9%) underwent distal pancreatectomy for adenocarcinoma of the body or tail, and 38 (6%) underwent total pancreatectomy for adenocarcinoma extensively involving the gland. The mean age of the patients was 64.3 years, with 54% being male and 91% being white. The overall perioperative mortality rate was 2.3%, whereas the incidence of postoperative complications was 30%. The median postoperative length of stay was 11 days. The mean tumor diameter was 3.2 cm, with 72 % of patients having positive lymph nodes, 30% having positive resection margins, and 36% having poorly differentiated tumors. Patients undergoing distal pancreatectomy for left-sided lesions had larger tumors (4.7 vs. 3.1 cm, P <0.0001), but fewer node-positive resections (59% vs. 73%, P = 0.03) and fewer poorly differentiated tumors (29% vs. 36%, P<0.001), as compared to those undergoing pancreaticoduodenectomy for right-sided lesions. The overall survival of the entire cohort was 63% at 1 year and 17% at 5 years, with a median survival of 17 months. For right-sided lesions the 1- and 5-year survival rates were 64% and 17%, respectively, compared to 50% and 15% for left-sided lesions. Factors shown to have favorable independent prognostic significance by multivariate analysis were negative resection margins (hazard ratio [HR] = 0.64, confidence interval [CI] = 0.50 to 0.82, P = 0.0004), tumor diameter less than 3 cm (HR = 0.72, CI = 0.57 to 0.90, P = 0.004), estimated blood loss less than 750 ml (HR = 0.75, CI = 0.58 to 0.96, P = 0.02), well/moderate tumor differentiation (HR = 0.71, CI = 0.56 to 0.90, P = 0.005), and postoperative chemoradiation (HR = 0.50, CI = 0.39 to 0.64, P } < 0.0001). Tumor location in head, neck, or uncinate process approached significance in the final multivariate model (HR = 0.60, CI = 0.35 to 1.0, P = 0.06). Pancreatic resection remains the only hope for long-term survival in patients with adenocarcinoma of the pancreas. Completeness of resection and tumor characteristics including tumor size and degree of differentiation are important independent prognostic indicators. Adjuvant chemoradiation is a strong predictor of outcome and likely decreases the independent significance of tumor location and nodal status.
Similar content being viewed by others
References
Greenlee RT, Murray T, Bolden S, et al.. Cancer statistics, 2000. CA Cancer J Clin 2O00;50:7–33.
Brennan MF, Moccia RD, Klimstra D. Management of ade- nocarcinoma of the body and tail of the pancreas. Ann Surg 1996;223:506–512.
Crile G Jr. The advantages of bypass over radical pancreati- coduodenectomy in the treatment of pancreatic carcinoma. Surg Gynecol Obstet 1970;130:1049–1053.
Shapiro TM. Adenocarcinoma of the pancreas. A statistical analysis of bypass vs. Whipple resection in good risk patients. Ann Surg 1975;182:715–721.
Crist DL, Sitzmann JV, Cameron JL. Improved hospital mor- bidity, mortality and survival after the Whipple procedure. Ann Surg 1987;206:358–365.
Geer RJ, Brennan MF. Prognostic indicators for survival af- ter resection of pancreatic adenocarcinoma. Am J Surg 1993; 217:43–49.
Trede M, Schwall G, Saeger H-D. Survival after pancreatico- duodenectomy: 118 consecutive resections without a mortal- ity. Ann Surg 1987;206:358–365.
Cameron JL, Pitt HA, Yeo CJ, et al.. One hundred and forty five consecutive pancrearicoduodenectomies without mortal- ity. Ann Surg 1993;217:430–438.
Yeo CJ, Cameron JL, Sohn TA, et al.. Six hundred fifty con- secutive pancrearicoduodenectomies in the 1990s: Pathology, complications, outcomes. Ann Surg 1997;226:248–260.
Gordon TA, Burleyson GP, Tielsch JM, et al.. The effects of regionalization on cost and outcome for one general high-risk surgical procedure. Ann Surg 1995;221:43–49.
Sosa JA, Bowman HM, Bass EB, et al.. Importance of hospital volume in the overall management of pancreatic cancer. Ann Surg 1998;228:429–438.
Lieberman MD, Kilburn H, Lindsey, et al.. Relation of peri- operative deaths to hospital volume among patients undergo- ing pancreatic resection for malignancy. Ann Surg 1995; 222:638–645.
Coquard R, Ayzae L, Romestaing P, et al.. Adenocarcinoma of the body and tail of the pancreas: Is there room for adjuvant radiotherapy? Tumori 1997;83:740–742.
Dalton RR, Sarr MG, van Heerden JA, et al.. Carcinoma of the body and tail of the pancreas: Is curative resection justi- fied? Surgery 1992; 111:489–494.
Willett CG, Lewandrowski K, Warshaw AL, et al.. Resection margins in carcinoma of the head of the pancreas: Implica- tions for radiation therapy. Ann Surg 1993;217:144–148.
Allison DC, Bose KK, Hruban RH, et al.. Pancreatic cancer cell DNA content correlates with long-term survival after pancreaticoduodenectomy. Ann Surg 1991;214:648–656.
Yeo CJ, Cameron JL, Lillemoe KD, et al.. Pancreaticoduo- denectomy for cancer of the head of the pancreas: 201 pa- tients. Ann Surg 1995;221:721–733.
Seymour AB, Hruban RH, Redston M, et al.. Allelotype of pancreatic carcinoma. Cancer Res 1994;54:2761–2764.
Redston MS, Caldas C, Seymour AB, et al.. p53 mutations in pancreatic carcinoma and evidence of common involvement of homocopolymer tracts in DNA microdeletions. Cancer Res 1994;54:3O25–3O33.
Yeo CJ, Abrams RA, Grochow LB, et al.. Pancreaticoduo- denectomy for pancreatic adenocarcinoma: Postoperative ad- juvant chemoradiation improves survival. A prospective, sin- gle-institution experience. Ann Surg 1997;225:621–636.
Yeo CJ, Sohn TA, Cameron JL, et al.. Periampullary adeno- carcinoma: Analysis of 5-year survivors. Ann Surg 1998;227:821–831.
Delcore R, Rodriquez FJ, Forster J, et al.. Significance of lymph node metastases in patients with pancreatic cancer un- dergoing curative resection. Am J Surg 1996;172:463–469.
Conlon KC, Klimstra DS, Brennan MF. Long-term survival after curative resection of pancreatic ductal adenocarcinoma. Clinicopathologic analysis of 5-year survivors. Ann Surg 1996; 223:273–279.
Satake K, Niskiwaki H, Yokomatsu H, et al.. Surgical curabil- ity and prognosis for standard versus extended resections for T1 carcinoma of the pancreas. Surg Gynecol Obstet 1992;175:259–265.
Pedrazzoli S, DiCarlo V, Dionigi R, et al.. Standard versus ex- tended lymphadenectomy associated with pancreaticoduo- denectomy in the surgical treatment of adenocarcinoma of the head of the pancreas. A multicenter, prospective, randomized study. Ann Surg 1998;228:508–517.
Yeo CJ, Cameron JL, Sohn TA, et al.. Pancreaticoduodenec- tomy with or without extended retroperitoneal lymphadenec- tomy for periampullary adenocarcinoma. Comparison of mor- bidity, mortality, and short-term outcome. Ann Surg 1999; 229:613–623.
Yeo CJ, Cameron JL, Maher MM, et al.. A prospective ran- domized trial of pancreaticogastrostomy versus pancreatico- jejunostomy after pancreaucoduodenectomy. Ann Surg 1995; 222:580–592.
Yeo CJ, Barry MK, Sauter PK, et al.. Erythromycin acceler- ates gastric emptying following pancreaticoduodenectomy: A prospective, randomized, placebo-controlled trial. Ann Surg 1993;218:229–238.
Kaiser MH, Ellenberg SS. Pancreatic cancer: Adjuvant com- bined radiation and chemotherapy following curative resec-tion. Arch Surg 1985;120:899–903.
Gastrointestinal Tumor Study Group. Further evidence of ef- fective adjuvant combined radiation and chemotherapy fol- lowing curative resection of pancreatic cancer. Cancer 1987; 59:2006–2010.
Carducri MA, Abrams RA, Yeo CJ, et al.. Early evaluation of abdominal/hepatic irradiation and 5-fluorouracil/leucovorin infusion after pancreaticoduodenectomy. Int J Radiat Oncol BiolPhys 1996;35:143–150.
Abrams RA, Grochow LB, Chakravatthy A, et al.. Intensified adjuvant therapy for pancreatic and periampullary adenocar- cinoma: Survival results and observations regarding patterns of failure, radiotherapy dose and CA 19-9 levels. Int J Radiat Oncol Biol Phys 1999;44:1039–1046.
Chakravarthy A, Abrams RA, Yeo CJ, et al. Intensified adju- vant combined modality therapy for resected pancreatic and periampullary adenocarcinoma: Acceptable toxicity with im- proved early results. Int J Radiat Oncol Biol Phys (in press).
Kaplan E, Meier P. Non-parametric estimation from incom- plete observations. J Am Stat Assoc 1958;53:457–481.
Cox DR. Regression models and life tables. J R Stat Soc 1972;34:187–220.
Herbert PC, Well G, Blajchman MA, et al.. A multicenter, ran- domized, controlled clinical tria) of transfusion requirements in critical care. N Engl J Med 1999;340:409–417.
Ncoptolemos JP, Baker P, Beger H, et al.. Progress report. A randomized multicenter European study comparing adjuvant radiotherapy, 6-mo chemotherapy, and combination therapy vs no-adjuvant treatment in resectable pancreatic cancer (ES- PAC-1). IntJ Pancreatol 1997;21:97–104.
Klinkenbijl JH, Jeekel J, Shamoud T, et al.. Adjuvant radio- therapy and 5-fluorouracil after curative resection of cancer of the pancreas and periampullary region: Phase III trial of the EORTC gastrointestinal cooperative group. Ann Surg 1999; 230:776–782.
Sharp KW, Ross CB, Halter SA, et al.. Pancreaticoduodenec- tomy with pyloric preservation for carcinoma of the pancreas. A cautionary note. Surgery 1989;105:645–653.
Roder JD, Stein HJ, Huttl W, et al.. Pylorus-preserving versus standard pancreaticoduodenectomy: An analysis of 110 pancre- atic and periampullary carcinomas. Br J Surg 1992;79:152–155.
Grace PA, Pitt HA, Longmire WP. Pylorus-preserving pancre- aticoduodenectomy: An overview. Br J Surg 1990;77:968–974.
Wenger FA, Jacobi CA, Haubold K, et al.. Gastrointestinal quality of life after duodenopancreatectomyin pancreatic car- cinoma. Preliminary results of a prospective randomized study: Pancreatoduodenectomy or pylorus-preserving pan- creatoduodenectomy. Chirurg 1999;70:1454–1459.
Seiler CA, Wagner M, Sadowski C, et al.. Randomized pro- spective trial of pylorus-preserving vs. classic duodenopancre- atectomy (Whipple procedure): Initial clinical results. J Gas- TRO1NTEST SURG 2000;4:443–452.
Takada T, Yasuda H, Shikita JI, et al.. Postprandial plasma gas- trin and secretin concentrations after pancreatoduodenec- tomy. Ann Surg 1989;210:47–51.
Williamson RCN, Bliouras N, Cooper MJ, et al.. Gastric emp- tying and enterogastric reflux after conservative and conven- tional pancreatoduodenectomy. Surgery 1993;114:82–86.
Caldas C, Hahn SA, Hruban RH, et al.. Detection of K-ras mutations in the stool of patients with pancreatic adenocarci- noma and pancreatic ductal mucinous cell hyperplasia. Cancer Res 1994;54:3568–3573.
Berthelemy P, Bouisson M, Escourrrou J, et al.. Identification of K-ras mutations in pancreatic juice early in the diagnosis of pancreatic cancer. Ann Intern Med 1995;123:188–191.
Wilentz RE, Chung CH, Sturm PDJ, et al.. K-ras mutations in duodenal fluid of patients with pancreas carcinoma. Cancer 1998;82:96–103.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sohn, T.A., Yeo, C.J., Cameron, J.L. et al. Resected adenocarcinoma of the pancreas— 616 patients: Results, outcomes, and prognostic indicators. J Gastrointest Surg 4, 567–579 (2000). https://doi.org/10.1016/S1091-255X(00)80105-5
Issue Date:
DOI: https://doi.org/10.1016/S1091-255X(00)80105-5