Abstract
Objective
To assess the predominant subtype of calcium channel present in neurons of the paracervical ganglia (PG) of the female rat and the ability of neuroactive peptides to modulate total calcium current.
Methods
Whole-cell patch clamp techniques were used on isolated PG neurons to assess calcium current modulation in the presence and absence of selective calcium channel subtype inhibitors and neuropeptides. Digital imaging analysis of cells was used to determine neuronal cell size distributions within the ganglia.
Results
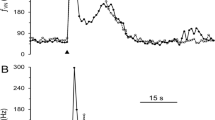
Average PG cell diameter was 28.1 μm. Most PG neurons (64%) had an N-type calcium current that contributed 41% of the total calcium current. The low voltage-activated or T-type current was not present. Very few neurons (22%) were sensitive to the P-type calcium channel blocker ω-agatoxin IVA, and in only 10% of neurons was the calcium current sensitive to the L-type channel blocker nimodipine. Neuropeptide Y (NPY) inhibited the calcium current by 41% in 79% of the neurons, but vasoactive intestinal peptide (VIP) had no effect. Calcitonin gene-related peptide (CGRP) both increased and decreased the calcium current in separate cell populations.
Conclusions
Calcium currents in female rat PG neurons are carried primarily through N-type calcium channels with a small contribution from L- and P-type channels. An unidentified calcium channel type is also present. Modulation of the calcium current by NPY is demonstrated, and support for the presence of a local, CGRP-mediated circuit is presented.
Similar content being viewed by others
References
Traurig HH, Papka RE. Autonomic efferent and visceral sensory innervation of the female reproductive system: Special reference to the functional roles of nerves in reproductive organs. In: Maggio CA, ed. Nervous control of the urogenital system. Chur, Switzerland: Harwood Academic Publishers, 1993:103–41.
Papka RE, Traurig HH. Autonomic efferent and visceral sensory innervation of the female reproductive system: Special reference to neurochemical markers in nerves and ganglionic connections. In: Maggio CA, ed. Nervous control of the urogenital system. Chur, Switzerland: Harwood Academic Publishers, 1993:423–66.
Tabatabai M, Booth AM, de Groat WC. Morphological and electrophysiological properties of pelvic ganglion cells in the rat. Brain Res 1986;382:61–70.
Morris JL, Gibbins IL. Neuronal colocalization of peptides, catecholamines, and catecholamine-synthesizing enzymes in guinea pig paracervical ganglia. J Neurosci 1987;7:3117–30.
Papka RE, Traurig HH, Klenn P. Paracervical ganglia of the female rat: Histochemistry and immunohistochemistry of neurons, SIF cells, and nerve terminals. Am J Anat 1987;179:245–57.
Inyama CO, Hacker GW, Gu J, Dahl D, Bloom SR, Polak JM. Cytochemical relationships in the paracervical ganglion (Frankenhauser) of rat studies by imnmno-histochemistry. Neurosci Lett 1985;55:311–6.
Gu J, Polak JM, Lazarides M, et al. Decrease of vasoactive intestinal peptide (VIP) in the penises from impotent men. Lancet 1984;ii:315–7.
Alm P, Alumets J, Hakanson R, et al. Origin and distribution of VIP (vasoactive intestinal pep tide)-nerves in the genitourinary tract. Cell Tissue Res 1980;205:337–47.
Lynch EM, Wharton J, Bryant MG, Bloom SR, Polak JM, Elder MG. The differential distribution of vasoactive intestinal polypeptide in the normal human genital tract. Histochemistry 1980;67:169–77.
Helm G, Ottesen B, Fahrenkrug J, et al. Vasoactive intestinal polypeptide (VIP) in the human female reproductive tract. Biol Reprod 1981;25:227–34.
Fahrenkrug J. Transmitter role of vasoactive intestinal peptide. Pharmacol Toxicol 1993;72:354–63.
Gibson SJ, Polak JM, Bloom SR, et al. Calcitonin gene-related peptide immunoreactivity in the spinal cord of man and eight other species. J Neurosci 1984;4:3101–11.
Papka RE. Some nerve endings in the rat pelvic paracervical autonomic ganglia and varicosities in the uterus contain calcitonin gene-related peptide and originate from dorsal root ganglia. Neuroscience 1990;39:459–70.
Inyama CO, Wharton J, Su HC, Polak JM. CGRP-immuno-reactive nerves in the genitalia of the female rat originate from dorsal root ganglia T11-L3 and L6-S1: A combined immunohis-tochemical and retrograde tracing study. Neurosci Lett 1986;69:13–8.
Katz B, Miledi R. The effect of calcium on acetycholine release from motor nerve terminals. Proc R Soc Lond [Biol] 1965;161:496–503.
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Archiv 1981;391:85–100.
Forscher P, Oxford GS. Modulation of calcium channel by norepinephrine in internally dialyzed avian sensory neurons. J Gen Physiol 1985;85:743–63.
Cohen CJ, McCarthy RT. Nimodipine block of calcium channels in rat anterior pituitary cells. J Physiol 1987;387:195–225.
Olivera BM, Miljanich GP, Ramachandran J, Adams ME. Calcium channel diversity and neurotransmitter release: The w-conotoxins and w-agatoxins. Ann Rev Biochem 1994;63:823–67.
Plummer MR, Logothetis DE, Hess P. Elementary properties and pharmacological sensitivities of calcium channels in mammalian peripheral neurons. Neuron 1989;2:1453–63.
Zhang JF, Randall DA, Ellinor PT, Home WA, Sather WA, Tanabe T, Schwarz TL, Tsien TW. Distinctive pharmacology and kinetics of cloned neuronal Ca2+ channels and their possible counterparts in mammalian CNS neurons. Neuropharmacology 1993;32:1075–88.
Bean BP. Neurotransmitter inhibition of neuronal calcium currents by changes in channel voltage dependence. Nature 1989;340:153–56.
Zhu Y, Zboran EL, Ikeda SR. Phenotype-specific expression of T-type calcium channels in neurons of the major pelvic ganglion of the adult male rat. J Physiol 1995;489:363–75.
Hillyard DR, Monje VD, Mintz IM, et al. A new conus peptide ligand for mammalian presynaptic Ca2+ channels. Neuron 1992;9:69–77.
Sather WA, Tanabe T, Zhang JF, Mori Y, Adams ME, Tsien RW. Distinctive biophysical and pharmacological properties of class A (B1) calcium channel a, subunits. Neuron 1993;11:292–303. ’
Randall AD, Wendland B, Schweizer F, Miljanich G, Adams ME, Tsein RW. Five pharmacologically distinct high voltage-activated Ca2+ channels in cerebellar granule cells. Soc Neurosci Abstr 1993;19:1478.
Mintz IM, Venema VJ, Swiderek KM, Lee TD, Bean BP, Adams ME. P-type calcium channels blocked by the spider toxic w-Aga-IVA. Nature 1992;355:827–29.
Mintz IM, Adams ME, Bean BP. P-type calcium channels in rat central and peripheral neurons. Neuron 1992;9:85–95.
Soong TW, Stea A, Hodson CD, Dubel SJ, Vincent SR, Snutch TP. Structure and functional expression of a member of the low voltage-activated calcium channel family. Science 1993;260:1133–6.
Stjernquist M, Owman C. Interaction of noradrenaline, NPY, and VIP with the neurogenic cholinergic response of the rat uterine cervix in vitro. Acta Physiol Scand 1987;131:553–62.
Wiley JW, Gross RA, Lu Y, MacDonald RL. Neuropeptide Y reduces calcium current and inhibits acetylcholine release in nodose neurons via a pertussis toxin-sensitive mechanism. J Neurophysiol 1990;63:1499–507.
Ewald DA, Sternweiss PC, Miller RJ. Guanine nucleotidebinding protein Go-induced coupling of neuropeptide Y receptors to Ca2+ channels in sensory neurons. Proc Natl Acad Sci 1988;85:3633–7.
Hirning LD, Fox AP, Miller RJ. Inhibition of calcium currents in cultured myenteric neurons bv neuropeptide Y. Brain Res 1990;532:120–30.
Ryu PD, Gerber G, Murase K, Randic M. Calcitonin gene-related peptide enhances calcium current of rat dorsal root ganglion neurons and spinal excitatory synaptic transmission. Neurosci Lett 1988;89:305–12.
Burden HW, Leonard M, Smith CP, Lawrence I, Jr. The sensory innervation of the ovary: A horseradish peroxidase study in the rat. Anatom Rec 1983;207:623–7.
Ono K, Delay M, Nakajima T, Insawa H, Giles W. Calcitonin gene-related peptide regulates calcium current in heart muscle. Nature 1989;340:721–4.
Ono K, Giles W. Electrophysiological effects of calcitonin gene-related peptide in bull-frog and guinea-pig atrial myocytes. J Physiol 1991;436:195–217.
Barsony J, Marx SJ. Dual effects of calcitonin and calcitonin gene-related peptide on intracellular cyclic 3′,5′-monophosphate in a human breast cancer cell line. Endocrinology 1988;122:1218–23.
Hokfelt T, Johansson O, Ljungdahl A, LundbergJM, Schultzberg M. Peptidergic neurons. Nature 1980;284:515–21.
Lundberg JM. Enkephalin, substance P, VIP, somatostatin, gastrin/CCK and neurotensin in peripheral neurons. Acta Physiol Scand Suppl 1979;496:14.
Kawatani M, Rutigliano M, deGroat WC. Depolarization and muscarinic excitation induced in a sympathetic ganglion by vasoactive intestinal polypeptide. Science 1985;229:879–81.
Isihara T, Shigemoto R, Mori K, Takahashi K, Nagata S. Functional expression and tissue distribution of a novel receptor for vasoactive intestinal polypeptide. Neuron 1992;8:811–9.
Hoffman F, Biel M, Flockerzi V. Molecular basis for Ca2+ channel diversity. Annu Rev Neurosci 1994;17:399–418.
Hess P. Calcium channels in vertebrate cells. Annu Rev Neurosci 1990:13:337–56.
Kerr LM, Yoshikami D. A venom peptide with a novel presynaptic blocking action. Nature 1984;308:282–4.
Tsien RW, Ellinor PT, Home WA. Molecular diversity of voltage-dependent Ca2+ channels. Trends Pharmacol Sci 1991;12:349–54.
Author information
Authors and Affiliations
Additional information
This work was supported by a grant from the National Institute of Neurological Disorders and Stroke, National Institutes of Health NS28894 (DLL). The authors thank E. Zboran for excellent technical assistance, Dr. R. E. Papka for mitial discussion, and Drs. P. McDonough, L. Plouffe, Jr, and K. Hansen for their critical review of the manuscript.
Rights and permissions
About this article
Cite this article
Cohen, D.P., Ikeda, S.R. & Lewis, D.L. Neuropeptide Y and Calcitonin Gene-Related Peptide Modulate Voltage-Gated Ca2+ Channels in Mature Female Rat Paracervical Ganglion Neurons. Reprod. Sci. 3, 342–349 (1996). https://doi.org/10.1016/S1071-5576(96)00041-X
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1071-5576(96)00041-X