Abstract
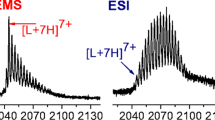
The extent of H/D exchange of the protein calmodulin in solution was monitored by mass spectrometry following electrospray ionization (ESI) of the protein. In the absence of Ca2+, approximately 115 protons are exchanged for deuteriums after 60 min. As the calmodulin is titrated with Ca2+, the extent of exchange decreases significantly (i.e., by 24 protons), indicating Ca2+-induced folding of the protein to a tighter, less solvent-accessible form. The extent of H/D exchange ceases to decrease when the amount of added Ca2+ is sufficient to convert greater than 80% of the calmodulin to a form bound by four calcium ions. Lysozyme, a protein of similar molecular weight, does not show a significant decrease in the extent of H/D exchange as it binds to Ca2+, indicating that the changes in H/D exchange for calmodulin reflect tertiary structural change that occur upon binding with Ca2+.
Similar content being viewed by others
References
Snyder, A. P., Ed. Biochem. Biotechnol. Appl. Electrospray Ionization Mass Spectrom., ACS Symp. Ser. 1996, 619.
Schnier, P. D.; Gross, D. S.; Williams, E. R. J. Am. Soc. Mass Spectrom. 1995, 6, 1086–1097.
Smith, R. D.; Loo, J. A.; Edmonds, C. G.; Barinaga, C. J.; Udseth, H. R. Anal. Chem. 1990, 62, 882–899.
Covey, T. R.; Bonner, R. F.; Shushan, B. I.; Henion, J. Rapid Commun. Mass Spectrom. 1988, 2, 249–256.
Katta, V.; Chait, B. T. Rapid Commun. Mass Spectrom. 1991, 5, 214–217.
LeBlanc, J. C. Y.; Beuchemin, D.; Siu, K. W. M.; Guevremont, R.; Berman, S. S. Org. Mass Spectrom. 1991, 26, 831–839.
Chowdury, S. K.; Cohen, S.; Chait, B. T. J. Am. Chem. Soc. 1990, 112, 9012–9013.
Mirza, U.; Cohen, S.; Chait, B. T. Anal. Chem. 1993, 65, 1–6.
Miranker, A.; Robinson, C. V.; Radford, S. E.; Aplin, R. T.; Dobson, C. M. Science 1993, 262, 896–900.
Robinson, C. V.; Gross, M.; Eyles, S. J.; Ewbank, J. J.; Mayhew, M.; Hartl, F. U.; Dobson, C. M.; Radford, S. E. Nature 1994, 372, 646–651.
Wagner, D. S.; Anderegg, R. J. Anal. Chem. 1994, 66, 706–711.
Wagner, D. S.; Melton, L. G.; Yan, Y. B.; Erickson, B. W.; Anderegg, R. J. Prot. Sci. 1994, 3, 1305–1314.
Katta, V.; Chait, B. T. J. Am. Chem. Soc. 1993, 115, 6317–6321.
Englander, S. W.; Kallenbach, N. R. Q. Rev. Biophys. 1984, 16, 521–655.
Englander, J. J.; Rogero, J. R.; Englander, S. W. Anal. Biochem. 1985, 147, 234–244.
Baldwin, R. L. Trends Biochem. Sci. (Pers. Ed.) 1989, 14, 291–294.
Baldwin, R. L. J. Biomol. NMR 1995, 5, 103–109.
Zhang, Z.; Smith, D. L. Protein Sci. 1993, 2, 522–531.
Liu, Y.; Smith, D. L. J. Am. Soc. Mass Spectrom. 1994, 5, 19–28.
Zhang, Z.; Smith, D. L. Protein Sci. 1996, 5, 1282–1289.
Mills, J. J.; Bailey, B. L.; Johnson, J. D. Biochemistry 1985, 24, 4897–4902.
O’Neill, K. T.; DeGrado, W. F. Proc. Nat. Acad. Sci. USA 1985, 82, 4954–4958.
Creenlee, D. V.; Andreasen, T. J.; Storm, D. R. Biochemistry 1982, 21, 2759–2764.
Barbato, G.; Ikura, M.; Kay, L. E.; Pastor, R. W.; Bax, A. Biochemistry 1992, 31, 5269–5278.
Kuboniwa, H.; Tjandra, N.; Grzesiek, S.; Ren, H.; Klee, C. B.; Bax, A. Nature Struct. Biol. 1995, 2, 768–776.
Finn, B. E.; Evenäs, J.; Drakenberg, T.; Waltho, J. P.; Thulin, E.; Forsén, S. Nature Struct. Biol. 1995, 2, 777–783.
Zhang, M.; Tanaka, T.; Ikura, M. Nature Struct. Biol. 1995, 2, 758–767.
Nemirovskiy, O.; Ramanathan, R.; Gross, M. L. J. Am. Soc. Mass Spectrom. 1997, 8, 809–812.
Babu, Y. S.; Bugg, C. E.; Cook, W. J. J. Mol. Biol. 1988, 204, 191–204.
Taylor, D. A.; Sack, J. S.; Maune, J. F.; Beckingham, K.; Quiocho, F. A. J. Biol. Chem. 1991, 266, 21375–21380.
Chattopadhyaya, R.; Meador, W. E.; Means, A. R.; Quiocho, F. A. J. Mol. Biol. 1992, 228, 1177–1192.
Heidorn, D. B.; Trewhella, J. Biochemistry 1988, 27, 909–915.
Yoshino, H.; Wakita, M.; Izumi, Y. J. Biol. Chem. 1993, 268, 12123–12128.
Seamon, K. B. Biochemistry 1980, 19, 207–215.
Ikura, M.; Hiraoki, T.; Hikichi, K.; Mikuni, T.; Yazawa, M.; Yagi, K. Biochemistry 1983, 22, 2573–2579.
Ikura, M.; Hiraoki, T.; Hikichi, K.; Minova, O.; Yamaguchi, H.; Yazawa, M.; Yagi, K. Biochemistry 1984, 23, 3124–3128.
Ikura, M.; Minova, O.; Hikichi, K. Biochemistry 1985, 24, 4264–4269.
Ikura, M.; Spera, S.; Barbato, G.; Kay, L. E.; Krinks, M.; Bax, A. Biochemistry 1991, 30, 9216–9228.
Hoffman, R. C.; Klevit, R. E. In Techniques in Protein Chemistry II; Villafranca, J., Ed., Academic: San Diego, 1991; pp 383–391.
Dedman, J. R.; Potter, J. D.; Jackson, R. L.; Johnson, J. D.; Means, A. J. Biol. Chem. 1977, 252, 8415–8422.
Crouch, T. H.; Klee, C. B. Biochemistry 1980, 19, 3692–3698.
Sorensen, B. R.; Shea, M. A. Biophys. J. 1996, 71, 3407–3420.
Yao, Y.; Schöneich, C.; Squier, T. C. Biochemistry 1994, 33, 7797–7810.
Gross, M. L. Tandem mass spectrometry: Multisector magnetic instruments. In Methods in Enzymology, Vol 193, Mass Spectrometry; McCloskey, J. A., Ed.; Academic: San Diego, 1990; pp 131–153.
Englander, J. J.; Calhoun, D. B.; Englander, S. W. Anal. Biochem. 1979, 92, 517–524.
Forsen, S.; Vogel, H. J.; Drakenberg, T. In Calcium and Cell Function; Cheung Ed.; Academic: New York, 1986; Vol. 6, pp 113–157.
Iida, S.; Potter, J. D. J. Biochem. 1986, 99, 1765.
Milos, M.; Schaer, J. J.; Comte, M.; Cox, J. A. Biochemistry 1986, 25, 6279–6287.
Tsuge, H.; Koseki, K.; Miyano, M.; Shimazaki, K.; Chuman, T.; Matsomoto, T.; Noma, M.; Nitta, K.; Sugai, S. Biochim. Biophys. Acta 1991, 1078, 77–84.
Inaka, K.; Kuroki, R.; Kikuchi, M.; Matsushima, M. J. Biol. Chem. 1991, 266, 20666–20671.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nemirovskiy, O., Giblin, D.E. & Gross, M.L. Electrospray ionization mass spectrometry and hydrogen/deuterium exchange for probing the interaction of calmodulin with calcium. J Am Soc Mass Spectrom 10, 711–718 (1999). https://doi.org/10.1016/S1044-0305(99)00036-7
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S1044-0305(99)00036-7