Abstract
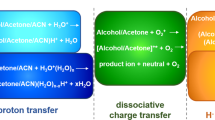
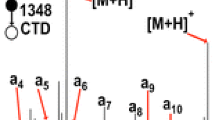
The unimolecular dissociation of (CH3)2C+OC2H5 ions (I) and their deuterated analogs, generated by ion-molecule reactions (IMR) in acetone-ethyl iodide mixtures was studied by tandem mass Spectrometry methods. Two significant processes that yielded I ions were identified. The Fourier transform ion cyclotron resonance study showed that the reaction between ionized ethyl iodide and neutral acetone was the principal source of I. This process involved the formation of the stable mixed ionized dimer, [C2H5I·O=C(CH3)2]+· (II), which dissociated by the loss of an I atom. Other important fragmentation pathways of II were the formation of C2H5I+·, (CH3)2CO+·; and (CH3)2COI+ and the loss of CH3CHI·. The major dissociation of I was the loss of C2H4. The activation energy for this reaction was determined by metastable ion appearance energy measurements to be ∼55 kJ mol−1 above the thermochemical minimum. The analysis of the metastable and collision-induced dissociation of D-labeled I showed an unusual time-energy effect on the degree of H/D mixing, with the highest selectivity for the ethene loss [β-H(D)-atom shift] being observed for ions with the lowest internal energies. Collisional excitation could not produce significant H/D mixing among dissociating ions. The results were rationalized by the existence of two species— the classical (2-ethoxypropyl) and nonclassical (proton-bound acetone-ethene pair) isomers of I. The classical structure was originally formed by IMR or from II. The energy barrier for the classical to nonclassical isomerization lay well above the thermochemical threshold for C2H4 loss, providing only limited H-atom mixing in nonclassical ions that were always formed in their dissociative state. The effect of the proton affinity of the carbonyl compound on the H/D mixing in RR′C+OC2H5 ions was studied. It was shown that the selectivity for the ethene loss (β-H-atom shift) generally increased with the increase of the proton affinity of RR′CO. Neutralization-reionization mass spectrometry was applied to a study of (CH3)2C+OR ions, where R = H, I, C2H5. The observation of a recovery signal for the ion I was attributed to the formation of the 2-ethoxypropyl radical. Neutral counterparts of (CH3)2COI+ ions were also generated, being the first example of IO-substituted alkyl radicals.
Similar content being viewed by others
References
Sirois, M.; George, M.; Holmes, J. L. Org. Mass Spectrom. 1994, 29, 11;
Wesdemiotis, C.; Fura, A.; McLafferty, F. W. J. Am. Soc. Mass Spectrom. 1991, 2, 459;
Harrison, A. G. Org. Mass Spectrom. 1987, 29, 637;
Jarrold, M. F.; Kirchner, J.; Liu, S.; Bowers, M. T. J. Phys. Chem. 1986, 90, 78.
Zagorevskii, D. V.; Sirois, M.; Cao, J. R.; George, M.; Holmes, J. L.; Ross, C. W., III. J. Mass Spectrom. 1995, 31, 55.
Bowen, R. D.; Derrick, P. J. J. Chem. Soc., Perkin Trans. 1992, 2, 1033.
Mead, T. J.; Williams, D. H. J. Chem. Soc., Perkin Trans. 1972, 2, 876.
Audier, H. E.; Monteiro, C.; Robin, D. Org. Mass Spectrom. 1989, 24, 146.
Sirois, M. Ph. D. Thesis University of Ottawa, Ontario, Canada, 1993.
Bouchoux, G.; Hoppilliard, Y. J. J. Am. Chem. Soc. 1990, 112, 9110;
Swanton, D. J.; Marsden, D. C. J.; Radom, L. Org. Mass Spectrom. 1991, 26, 227.
Bowen, R. D.; Stapleton, B. J.; Williams, D. H. J. Chem. Soc. Chem. Commun. 1978, 24;
Bowen, R. D.; Williams, D. H. J. Am. Chem. Soc. 1980, 302, 2752;
Bowen, R. D.; Williams, D. H. Int. J. Am. Chem. Soc. 1978, 100, 7454;
Bowen, R. D.; Williams, D. H. Int. J. Mass Spectrom. Ion Phys. 1979, 29, 47.
Traeger, J. C.; Mommers, A. A. Org. Mass Spectrom. 1987, 22, 592.
Holmes, J. L.; Osborne, A. D. Int. J. Mass Spectrom. Ion Phys. 1977, 23, 189;
Burgers, P. C.; Holmes, J. L. Org. Mass Spectrom. 1982, 17, 123.
Marshall, A. G.; Wang, T.-C. L.; Ricca, T. L. J. Am. Chem. Soc. 1985, 107, 7893.
Zagorevskii, D. V.; Palii, S. P.; Holmes, J. L. J. Am. Soc. Mass Spectrom. 1994, 5, 814.
Zagorevskii, D. V.; Holmes, J. L. Org. Mass Spectrom. 1994, 29, 594.
Lossing, F. P. J. Am. Chem. Soc. 1977, 99, 7526.
Pottie, R. F.; Hamill, W. H. J. Phys. Chem. 1959, 63, 877.
Lias, S. G.; Bartmess, J. E.; Liebman, J. F.; Holmes, J. L.; Levin, R. D.; Mallard, W. G. J. Phys. Ref. Data 1988, 17, Suppl. 1.
Holmes, J. L.; Lossing, F. P. J. Am. Chem. Soc. 1988, 110, 7344.
Livant, P.; Illies, A. J. Am. Chem. Soc. 1991, 113, 1510.
Burgers, P. C.; Holmes, J. L. Int. J. Mass Spectrom. Ion Processes 1984, 58, 15.
Harnish, D.; Holmes, J. L. J. Am. Chem. Soc. 1991, 113, 9729.
Wagner, W.; Heimbach, H.; Levsen, K. Int. J. Mass Spectrom. Ion Phys. 1980, 36, 125.
Burgers, P. C.; Holmes, J. L.; Szulejko, J. E.; Mommers, A. A.; Terlouw, J. K. Org. Mass Spectrom. 1983, 18, 254.
Larson, J. W.; McMahon, T. B. J. Am. Chem. Soc. 1982, 204, 6255.
Bowen, R. D.; Williams, D. H.; Hvistendahl, G.; Kalman J. R. Org. Mass Spectrom. 1978, 23, 721;
McAdoo, D. J.; Akhmed, M. S.; Hudson, C. E.; Giam, C. S. Int. J. Mass Spectrom. Ion Processes 1990, 100, 579.
Hvistendahl, G. J. Am. Chem. Soc. 1975, 97, 3097;
Holmes, J. L.; Rye, R. T. B.; Terlouw, J. K. Org. Mass Spectrom. 14 1979, 606.
Hehre, W. J.; Radom, L.; Schleyer, P. v. R.; Pople, J. A. Ab Initio Molecular Orbital Theory; Wiley: New York, 1986, p 384 and references therein.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zagorevskii, D.V., Holmes, J.L. & Ross, C.W. A tandem mass spectrometry and Fourier transform-ion cyclotron resonance study of ionized ethylated acetone and deuterated analogs. J Am Soc Mass Spectrom 8, 327–336 (1997). https://doi.org/10.1016/S1044-0305(96)00280-2
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S1044-0305(96)00280-2