Abstract
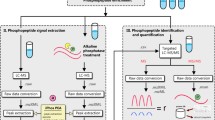
Recently, we reported a fast on-line alkaline micro-liquid chromatography/electrospray-atmospheric pressure ionization/collision-induced dissociation/mass spectrometric approach for sensitive phosphopeptide screening of a tryptic digested protein and subsequent characterization of the identified phosphopeptide. Based on this study, we now applied an improved method for the identification of phosphorylation sites in insulin receptor substrate 1, an important mediator in insulin signal transduction which was phosphorylated in vitro by protein kinase C-ζ. The approach consists of an on-line alkaline negative-ion micro-liquid chromatography/electrospray-atmospheric pressure ionization/collision-induced dissociation/mass spectrometric hybrid scan experiment using a triple-quadrupole mass spectrometer with fractionation and subsequent off-line nanoES-MS (ion trap) analysis of the phosphopeptide-containing fractions. During the liquid chromatography (LC)/ES-MS experiment, the phosphopeptides of the enzymatic digest mixture of the studied insulin receptor substrate 1 fragment were detected under high skimmer potential (API-CID) using phosphorylation-specific m/z 79 marker ions as well as the intact m/z-values of the peptides which were recorded under low skimmer potential. Subsequently, the targeted fractions were analyzed by off-line nanoES-MS/MS and MS3. Using this approach, serine 318 was clearly identified as a major in vitro protein kinase C-ζ phosphorylation site in the insulin receptor substrate −1 fragment. Together, our results indicate that the applied strategy is useful for unequivocal and fast analysis of phosphorylation sites in low abundant signaling proteins.
Article PDF
Similar content being viewed by others
References
Zierath, J. R.; Wallberg-Henriksson, H. From Receptor to Effector: Insulin Signal Transduction in Skeletal Muscle From Type II Diabetic Patients. Ann. N.Y. Acad. Sci. 2002, 967, 120–134.
Liu, Y. F.; Paz, K.; Hershkowitz, A.; Alt, A.; Tenenbaum, T.; Sampson, S.; Ohba, M.; Kuroki, T.; LeRoith, D.; Zick, Y. Insulin Stimulates PKC-zeta-Mediated Phosphorylation of Insulin Receptor Substrate-1 (IRS-1): A Self-Attenuated Mechanism to Negatively Regulate IRS Proteins Function. J. Biol. Chem. 2001, 276, 14459–14465.
Ravichandran, L. V.; Esposito, D. L.; Chen, J.; Quon, M. J. PKC-zeta Phosphorylates IRS-1 and Impairs Its Ability to Activate PI 3-Kinase in Response to Insulin. J. Biol. Chem. 2001, 276, 3543–3549.
Quadroni, M.; James, P. Phosphopeptide Analysis. In Proteomics in Functional Genomics: Protein Structure Analysis; Jolles, P.; Jörnvall, H., Eds.; Birkhäuser-Verlag: Basel, Boston, Berlin, 2000.
Larsen, M. R.; Sorensen, G. L.; Fey, S. J.; Larsen, P. M.; Roepstorff, P. Phospho-Proteomics: Evaluation of the Use of Enzymatic De-Phosphorylation and Differential Mass Spectrometric Peptide Mass Mapping for Site Specific Phosphorylation Assignment in Proteins Separated by Gel Electrophoresis. Proteomics 2001, 1, 223–238.
Aebersold, R.; Goodlett, D. R. Mass Spectrometry in Proteomics. Chem. Rev. 2001, 101, 269–295.
Mann, M.; Ong, S. E.; Gronborg, M.; Steen, H.; Jensen, O. N.; Pandey, A. Analysis of Protein Phosphorylation Using Mass Spectrometry: Deciphering the Phosphoproteome. Trends Biotechnol 2002, 20, 261–268.
Huddleston, M. J.; Annan, R. S.; Bean, M. F.; Carr, S. A. Selective Detection of Phosphopeptides in Complex Mixtures by Electrospray Liquid Chromatography/Mass Spectrometry. J. Am. Soc. Mass Spectrom. 1993, 4, 710–717.
Ding, J.; Burkhart, W.; Kassel, D. B. Identification of Phosphorylated Peptides from Complex Mixtures Using Negative Ion Orifice-Potential Stepping and Capillary Liquid Chromatography/Electrospray Ionization Mass Spectrometry. Rapid Commun. Mass Spectrom. 1994, 8, 94–98.
Hunter, A. P.; Games, D. E. Chromatographic and Mass Spectrometric Methods for the Identification of Phosphorylation Sites in Phosphoproteins. Rapid Commun. Mass Spectrom. 1994, 8, 559–570.
Jedrzejewski, P. T.; Lehmann, W. D. Detection of Modified Peptides in Enzymatic Digests by Capillary Liquid Chromatography/Electrospray Mass Spectrometry and a Programmable Skimmer CID Acquisition Routine. Anal. Chem. 1997, 69, 294–301.
Annan, R. S.; Huddleston, M. J.; Verma, R.; Deshaies, R. J.; Carr, S. A. A Multidimensional Electrospray MS-Based Approach to Phosphopeptide Mapping. Anal. Chem. 2001, 73, 393–404.
Beck, A.; Deeg, M.; Möschel, K.; Schmidt, E. K.; Schleicher, E. D.; Voelter, W.; Lehmann, R. Alkaline Liquid Chromatography Electrospray-Ionization Skimmer Collision-Induced Dissociation Mass Spectrometry for Phosphopeptide Screening. Rapid. Commun. Mass Spectrom. 2001, 15, 2324–2333.
Zappacosta, F.; Huddleston, M. J.; Karcher, R. L.; Gelfand, V. I.; Carr, S. A.; Annan, R. S. Improved Sensitivity for Phosphopeptide Mapping Using Capillary Column HPLC and Microionspray Mass Spectrometry: Comparative Phosphorylation Site Mapping from Gel-Derived Proteins. Anal. Chem. 2002, 74, 3221–3231.
Hunter, A. P.; Games, D. E. Chromatographic and Mass Spectrometric Methods for the Identification of Phosphorylation Sites in Phosphoproteins. Rapid. Commun. Mass Spectrom. 1994, 8, 559–570.
Carr, S. A.; Huddleston, M. J.; Annan, R. S. Selective Detection and Sequencing of Phosphopeptides at the Femtomole Level by Mass Spectrometry. Anal. Biochem. 1996, 239, 180–192.
Wilm, M.; Neubauer, G.; Mann, M. Parent Ion Scans of Unseparated Peptide Mixtures. Anal. Chem. 1996, 68, 527–533.
Neubauer, G.; Mann, M. Mapping of Phosphorylation Sites of Gel-Isolated Proteins by Nanoelectrospray Tandem Mass Spectrometry: Potentials and Limitations. Anal. Chem. 1999, 71, 235–242.
Stensballe, A.; Jensen, O. N.; Olsen, J. V.; Haselmann, K. F.; Zubarev, R. A. Electron Capture Dissociation of Singly and Multiply Phosphorylated Peptides. Rapid Commun. Mass Spectrom. 2000, 14, 1793–1800.
Shi, S. D. H.; Hemling, M. E.; Carr, S. A.; Horn, D. M.; Lindh, I.; McLafferty, F. W. Phosphopeptide/Phosphoprotein Mapping by Electron Capture Dissociation Mass Spectrometry. Anal. Chem. 2001, 73, 19–22.
Mansoori, B. A.; Volmer, D. A.; Boyd, R. K. “Wrong-Way-Round” Electrospray Ionization of Amino Acids. Rapid Commun. Mass Spectrom. 1997, 11, 1120–1130.
DeGnore, J. P.; Qin, J. Fragmentation of Phosphopeptides in an Ion Trap Mass Spectrometer. J. Am. Soc. Mass Spectrom. 1998, 9, 1175–1188.
Tholey, A.; Reed, J.; Lehmann, W. D. Electrospray Tandem Mass Spectrometric Studies of Phosphopeptides and Phosphopeptide Analogues. J. Mass Spectrom. 1999, 34, 117–123.
Reid, G. E.; Simpson, R. J.; O’Hari, R. A. Leaving Group and Gas Phase Neighboring Group Effects in the Side Chain Losses from Protonated Serine and Its Derivatives. J. Am. Soc. Mass Spectrom. 2000, 11, 1047–1060.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online March 14, 2003
Rights and permissions
About this article
Cite this article
Beck, A., Moeschel, K., Deeg, M. et al. Identification of an in vitro insulin receptor substrate-1 phosphorylation site by negative-ion μLC/ES-API-CID-MS hybrid scan technique. J Am Soc Mass Spectrom 14, 401–405 (2003). https://doi.org/10.1016/S1044-0305(03)00122-3
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S1044-0305(03)00122-3