Abstract
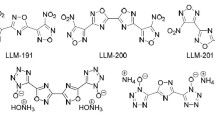
The mass spectrometric behavior of lithiated derivatives of 2,5-disubstituted-1,3,4-oxadiazoles has confirmed the skeletal rearrangement presented earlier for protonated derivatives. In the case of [M + H]+ ions the loss of isocyanic acid was observed and for [M + Li]+ ions the loss of lithium isocyanate occurred. On the other hand, benzoyl ions [RCO]+ were formed from [M + H]+ ions, but not from [M + Li]+ ions. Formation of benzoyl ions was in agreement with the differences between bond orders calculated for [M+H]+ ions and neutral molecules. From [M + Li]+ ions the [RCNLi]+ fragment ions were formed, but the formation of [RCNH]+ fragment ions from [M + H]+ ions was not observed. This result can be explained on the basis of theoretically calculated stabilities of these fragment ions, since the calculated heats of formation of [RCNLi]+ ions were found to be substantially lower than those of the respective [RCNH]+ ions.
Article PDF
Similar content being viewed by others
References
Asam, M. R.; Glish, G. L. Tandem Mass Spectrometry of Alkali Cationized Polysaccharides in a Quadrupole Ion Trap. J. Am. Soc. Mass Spectrom. 1997, 8, 987–995.
Madhusudanan, K. P.; Bajpai, L. K.; Bhaduri, A. P. Effect on Lithium Cationization on Oxygen Atom Transfer from the Nitro Group to the Double Bond During Collision-Induced Decomposition in Certain Substituted β-Nitrostyrenes. Rapid Commun. Mass Spectrom. 1997, 11, 1263–1265.
Da Silva, E. L. M.; Roblot, F.; Hocquemiller, R.; Serani, L.; Laprévote, O. Structure Elucidation of Annoheptocins, Two New Heptahydroxylated C37 Acetogenins by High Energy Collision-Induced Dissociation Tandem Mass Spectrometry. Rapid Commun. Mass Spectrom. 1998, 12, 1936–1944.
Madhusudanan, K. P.; Katti, S. B. Elimination of Neutral Lithium Atom from Lithiated Nucleobases Upon Collision-Induced Decomposition. J. Mass Spectrom. 1998, 33, 20–24.
Hsu, F. F.; Turk, J. Distinction Among Isomeric Unsaturated Fatty Acids as Lithiated Adducts by Electrospray Ionization Mass Spectrometry Using Low Energy Collisionally Activated Dissociation on a Triple Stage Quadrupole Instrument. J. Am. Soc. Mass Spectrom. 1999, 10, 600–612.
Ngoka, L. C. M.; Gross, M. L. Multistep Collisionally Activated Decomposition in an Ion Trap for the Determination of the Amino Acid Sequence and Gas-Phase Ion Chemistry of Lithium-Coordinated Valinomycin. Int. J. Mass Spectrom. 2000, 194, 247–259.
Hsu, F. F.; Bohrer, A.; Turk, J. Formation of Lithiated Adducts of Glyceroposphoholine Lipids Facilitates Their Identification by Electrospray Ionization Tandem Mass Spectrometry. J. Am. Soc. Mass Spectrom. 1998, 9, 516–526.
Wang, W.; Liu, Z.; Ma, L.; Hao, C.; Liu, S.; Voinov, V. G.; Kalinovskaya, N. I. Electrospray Ionization Multiple-Stage Tandem Mass Spectrometric Analysis of Diglycosyldiacylglycerol Glycolipids from the Bacteria. Bacillus pumilus. Rapid Commun. Mass Spectrom. 1999, 12, 1189–1196.
Hsu, F. F.; Turk, J. Structural Determination of Glycosphingolipids as Lithiated Adducts by Electrospray Ionization Mass Spectrometry Using Low Energy Collision-Activated Dissociation on a Triple Stage Quadrupole Instrument. J. Am. Soc. Mass Spectrom. 2001, 12, 61–79.
Madhusudanan, K. P.; Mathad, V. T. Shefali; Raj, K.; Bhaduri, A. P. Characterization of Iridoids by Fast Atom Bombardment Mass Spectrometry Followed by Collision-Induced Dissociation of [M+Li]+ ions. J. Mass Spectrom. 2000, 35, 321–329.
Madhusudanan, K. P.; Raj, K.; Bhaduri, A. P. Effect of Metal Cationization on the Low Energy Collision-Induced Dissociation of Loganin, epi-Loganin, and Ketologanin Studied by Electrospray Ionization Tandem Mass Spectrometry. J. Mass Spectrom. 2000, 35, 901–911.
Hue, N.; Montagnac, A.; Pais, M.; Serani, L.; Laprévote, O. Structural Elucidation of Eighteen Cerebrosides from Holothuria coronopertusa in a Complex Mixture by High Energy Collision-Induced Dissociation of [M+Li]+ ions. Eur. J. Mass Spectrom. 2001, 7, 409–417.
Cui, M.; Song, F.; Liu, Z.; Liu, S. Metal Ion Adducts in the Structural Analysis of Ginsenosides by Electrospray Ionization with Multi-Stage Mass Spectrometry. Rapid Commun. Mass Spectrom. 2001, 15, 586–595.
Chen, R.; Li, L. Lithium and Transition Metal Ions Enable Low Energy Collision-Induced Dissociation of Polyglycols in Electrospray Ionization Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2001, 12, 832–839.
Hsu, F. F.; Turk, J.; Stewart, M. E.; Downing, D. T. Structural Studies on Ceramides as Lithiated Adducts by Low Energy Collision-Activated Dissociation Tandem Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2002, 13, 680–695.
Seldes, A. M. D.’; Accorso, N.; Souto, M. F.; Alho, M. M.; Arabehety, C. G. Gas-Phase Conversion of Tetrazoles to Oxadiazoles: Isolation and Acetylation of the N-Acylated Intermediate. J. Mass Spectrom. 2001, 36, 1069–1073.
Frański, R.; Schroeder, G.; Rybachenko, V.; Szwajka, O. P. Loss of Isocyanic Acid from the Internal Oxadiazole Ring of Protonated Molecules of Some 2,5-Diaryl-1,3,4-Oxadiazoles. Rapid Commun. Mass Spectrom. 2002, 16, 390–395.
Qiang, X.; Zhang, R. Synthesis and Insecticidal Activities of Novel 2,5-Disubstituted-1,3,4-Oxadiazoles. J. Chem. Technol. Biotechnol. 1996, 67, 124–130.
Rai, K. M. L.; Linganna, N. Synthesis and Evaluation of Antimitotic Activity of Alkylated 2-Amino-1,3,4-Oxadiazoles. II. Farmaco 2000, 55, 389–392.
Liu, Z.; Yang, G.; Qin, X. Synthesis and Biological Activities of Novel Diheterocyclic Compounds Containing 1,2,4,-Triazolo[1,5-α]Pyrimidine and 1,3,4-Oxadiazole. J. Chem. Technol. Biotechnol. 2001, 76, 1154–1158.
Shi, W.; Qian, X.; Zhang, R.; Song, G. Synthesis and Quantitative Structure-Activity Relationships of New 2,5-Disubstituted-1,3,4-Oxadiazoles. J. Agri. Food Chem. 2001, 49, 124–130.
Chen, H.; Li, Z.; Han, Y. Synthesis and Fungicidal Activity Against Rhizoctonia solani of 2-Alkyl (Alkylthio)-5-Pyrazolyl-1,3,4-Oxadiazoles (Thiadiazoles). J. Agri. Food Chem. 2000, 48, 5312–5315.
Belen’kii, L. I.; Luiksaar, S. I.; Poddubnyi, I. S.; Krayushkin, M. M. Novel Syntheses of Symmetrical 2,5-Diaryl-1,3,4-Oxadiazoles and 1,4-Phenylenebis-1,3,4-Oxadiazoles. Izv. Akad. Nauk Ser. Khim. 1998, 11, 2309–2315.
Janietz, S.; Anlaouf, S. A New Class of Organosoluble Rigid-Rod Fully Aromatic Poly(1,3,4-Oxadiazole)s and Their Solid State Properties. 1. Synthesis. Macromol. Chem. Phys. 2002, 203, 427–432.
Grekov, A. P.; Szwajka, O. P. Synthesis of Some 2,5-Diphenyl-1,3,4-Oxadizoles. Zh. Obshch. Khim 1960, 30, 3082–3086 (in Russian).
Szwajka, O. P.; Mnacakanova, T. P.; Golovatyi, V. G.; Schevchuk, L. G. Investigation of Oxadiazole Derivatives. Chem. Heter. Comp 1970, 2, 300–303 (in Russian).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published online February 27, 2003
Rights and permissions
About this article
Cite this article
Frański, R., Eitner, K., Schroeder, G. et al. Mass spectrometric study of some protonated and lithiated 2,5-disubstituted-1,3,4-oxadiazoles. J Am Soc Mass Spectrom 14, 289–294 (2003). https://doi.org/10.1016/S1044-0305(03)00005-9
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S1044-0305(03)00005-9