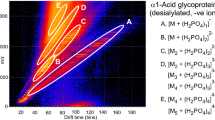
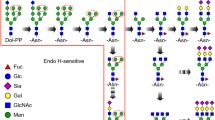
Abstract
The structure of N-linked glycans is determined by a complex, anabolic, intracellular pathway but the exact role of individual glycans is not always clear. Characterization of carbohydrates attached to glycoproteins is essential to aid understanding of this complex area of biology. Specific mass spectral detection of glycopeptides from protein digests may be achieved by on-line HPLC-MS, with selected ion monitoring (SIM) for diagnostic product ions generated by cone voltage fragmentation, or by precursor ion scanning for terminal saccharide product ions, which can yield the same information more rapidly. When glycosylation is heterogeneous, however, these approaches can result in spectra that are complex and poorly resolved. We have developed methodology, based around precursor ion scanning for ions of high m/z, that allows site specific detection and structural characterization of glycans at high sensitivity and resolution. These methods have been developed using the standard glycoprotein, fetuin, and subsequently applied to the analysis of the N-linked glycans attached to the scrapie-associated prion protein, PrPSc. These glycans are highly heterogeneous and over 30 structures have been identified and characterized site specifically. Product ion spectra have been obtained on many glycopeptides confirming structure assignments. The glycans are highly fucosylated and carry Lewis X or sialyl Lewis X epitopes and the structures are in-line with previous results. [Abbreviations: Hex—Hexose, C6H12O6 carbohydrates, including mannnose and galactose; HexNAc—N-acetylhexosamine, C8H15NO6 carbohydrates, including N-acetylglucosamine and N-acetylgalactosamine; GlcNAc—N-acetylglucosamine; GalNAc—N-acetylgalactosamine; Fuc—Fucose; NeuAC—N-acetylneuraminic acid or sialic acid; TSE—Transmissible Spongiform Encephalopathy.]
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Helenius, A.; Aebi, M. Intracellular Functions of N-Linked Glycans. Science 2001, 291, 2364–2369.
Dwek, R. A. Glycobiology: Toward Understanding the Function of Sugars. Chem. Rev. 1996, 96, 683–720.
Rudd, P. M.; Elliott, T.; Cresswell, P.; Wilson, I. A.; Dwek, R. A. Glycosylation and the Immune System. Science 2001, 291, 2370–2376.
Parekh, R. B.; Dwek, R. A.; Sutton, B. J.; Fernandes, D. L.; Leung, A.; Stanworth, D.; Rademacher, T. W. Association of Rhuematoid Arthritis and Primary Osteoarthritis with Changes in the Glycosylation Pattern of Total Serum IgG. Nature 1985, 316, 452–457.
Parekh, R.; Isenberg, D.; Rook, G.; Roitt, A. Comparative Analysis of Disease-Associated Changes in the Galactosylation of Serum IgG. J. Autoimmun. 1989, 2, 101–114.
Turner, G. Haptoglobin—A Potential Reporter Molecule for Glycosylation Changes in Disease. Adv. Exp. Med. Biol. 1995, 376, 231–238.
Maguire, T.; Breen, K. A Decrease in Neural Sialyltransferase Activity in Alzheimers Disease. Dementia 1995, 6, 185–90.
Maguire, T.; Thakore, J.; Dinan, T. G.; Hopwood, S.; Breen, K. C. Plasma Sialyltransferase Levels in Psychiatric Disorders as a Possible Indicator of HPA Axis Function. Biol. Psychiat. 1997, 41, 1131–1136.
Mackiewicz, A.; Dewey, M. J.; Berger, F. G.; Baumann, H. Acute Phase Mediated Change in Glycosylation of Rat Alpha 1-Acid Glycoprotein in Transgenic Mice. Glycobiol. 1991, 1, 265–269.
Medzihradszky, K. F.; Maltby, D. A.; Hall, S. C.; Settineri, C. A.; Burlingame, A. L. Characterization of Protein N-Glycosylation by Reversed-Phase Microbore Liquid Chromatography/Electrospray Mass Spectrometry, Complementary Mobile Phases, and Sequential Exoglycosidase Digestion. Am. Soc. Mass Spectrom. 1994, 5, 350–358.
Wilm, M.; Neubauer, G.; Mann, M. Parent Ion Scans of Unsepparated Peptide Mixtures. Anal. Chem. 1996, 68, 527–533.
Huddleston, M. J.; Bean, M. F.; Carr, S. A. Collisional Fragmentation of Glycopeptides by Electrospray Ionization LC MS and LC MS MS—Methods for Selective Detection of Glycopeptides in Protein Digests. Anal. Chem. 1993, 65, 877–884.
Gerwig, G.; Vliegenthart, J. Analysis of Glycoprotein-Derived Glycopeptides. EXS 2000, 88, 159–186.
Stimson, E.; Hope, J.; Chong, A.; Burlingame, A.L. Site-specific characterization of the N-linked glycans of murine prion protein by high-performance liquid chromatography/electrospray mass spectrometry and exoglycosidase digestions. Biochem. 1999, 38, 4885–4895.
Rudd, P. M.; Endo, T.; Colominas, C.; Groth, D.; Wheeler, S. F.; Harvey, D. J.; Wormwald, M. R.; Serban, H.; Prusiner, S. B.; Kobata, A.; Dwek, R. A. Glycosylation Differences Between the Normal and Pathogenic Prion Isoforms. PNAS U.S.A. 1999, 96, 13044–13049.
Hope, J.; Multhaup, G.; Reekie, L. J.; Kimberlin, R. H.; Beyreuther, K. Molecular Pathology of Scrapie-Associated Fibril Protein (PrP) in Mouse Brain Affected by the ME7 Strain of Scrapie. Eur. J. Biochem. 1988, 172, 271–277.
Annan, R. S.; Carr, S. A. The Essential Role of Mass Spectrometry in Characterizing Protein Structure: Mapping Posttranslational Modifications. J. Protein Chem. 1997, 16, 391–402.
Domon, B.; Costello, C.E. A Sytematic Nomenclature for Carbohydrate Fragmentations in FAB/MS Spectra of Glycocojugates. Glycoconj. 1988, 5, 397–409.
Green, E. D.; Adelt, G.; Baenziger, J. U.; Wilson, S.; Van Halbeek, H. The Asparagine Linked Oligosaccharides on Bovine Fetuin. J. Biol. Chem. 1988, 263, 18253–18268.
Rice, K. G.; Rao, N. B. N.; Lee, Y. C. Large Scale Preparation and Characterization of N-Linked Glycopeptides from Bovine Fetuin. Anal. Biochem. 1990, 184, 249–258.
Stahl, N.; Baldwin, M. A.; Hecker, R.; Pan, K. M.; Burlingame, A. L.; Prusiner, S. B. Glycosylinositol Phospholipid Anchors of the Scrapie and Cellular Prion Proteins Contain Sialic-Acid. Biochem. 1992, 31, 5043–5053.
Turk, E.; Teplow, D. B.; Hood, L. E.; Prusiner, S. B. Purification and Properties of the Cellular and Scrapie Hamster Prion Proteins. Eur. J. Biochem. 1988, 176, 21–30.
Gill, A. C.; Ritchie, M. A.; Hunt, L. G.; Steane, S. E.; Davis, K. G.; Bocking, S. P.; Rhie, A. G. O.; Bennett, A. D.; Hope, J. Post-Translational Hydroxylation at the N-Terminus of the Prion Protein Reveals the Presence of PPII Structure in Vivo. EMBO J. 2000, 19, 5324–5331.
Lehmann, S.; Harris, D. A. Blockade of Glycosylation Promotes Acquisition of Scrapie-Like Properties by the Prion Protein in Cultured Cells. J. Biol. Chem. (1997, 272, 21479–21487)
Hope, J.; Wood, S.; Birkett, C.; Chong, A.; Bruce, M.; Cairns, D.; Goldmann, W.; Hunter, M.; Bostock, C. Molecular Analysis of Ovine Prion Protein Identifies Similarties Between BSE and an Experimental Isolate of Natural Scrapie, CH1641. J. Gen. Virol. 1999, 80, 1–4.
Priola, S. A.; Lawson, V. A. Glycosylation Influences Cross-Species Formation of Protease-Resistant Prion Protein. EMBO 2001, 20, 3392–6699.
Somerville, R. A. Host and Transmissible Spongiform Encephalopathy Agent Strain Control Glycosylation of PrP. J. Gen. Virol. 1999, 80, 1865–1872.
Stahl, N.; Baldwin, M. A.; Teplow, D.; Hood, L.; Beavis, R.; Chait, B.; Gibson, B. W.; Burlingame, A. L.; Prusiner, S. B. Prion Diseases of Humans and Animals. Ellis Horwood Ltd: Chichester, UK, 1992; pp. 361–379.
Chen, Y. J.; Wing, D. R.; Guile, G. R.; Dwek, R. A.; Harvey, D. J.; Zamze, S. Nuetral N-Glycans in Adult Rat Brain Tissue. Eur. J. Biochem. 1997, 251, 691–703.
Zamze, S.; Harvey, D. J.; Chen, Y. J.; Guile, G. R.; Dwek, R. A.; Wing, D. R. Sialylated N-Glycans in Adult Rat Brain Tissue—a Widespread Distribution of Disialylated Antennae in Complex and Hybrid Structures. Eur. J. Biochem. 1998, 258, 243–270.
Harvey, D. J. Electrospray Mass Spectrometry and Fragmentation of N-linked Carbohydrates Derivatized at the Reducing Terminus. J. Am. Soc. Mass Spectrom. 2000, 11, 900–915.
Spina, E.; Cozzolino, R.; Ryan, E.; Garozzo, D. Sequencing of Oligosaccharides by Collision Induced Dissociation Matrix Assisted Laser Desorption/Ionization Mass Spectrometry. J. Mass. Spectrom. 2000, 35, 1042–1048.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ritchie, M.A., Gill, A.C., Deery, M.J. et al. Precursor ion scanning for detection and structural characterization of heterogeneous glycopeptide mixtures. J. Am. Soc. Spectrom. 13, 1065–1077 (2002). https://doi.org/10.1016/S1044-0305(02)00421-X
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/S1044-0305(02)00421-X