Abstract
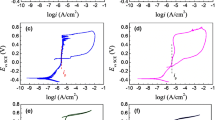
The effect of solution pH, Cl− concentration and temperature on the electrochemical corrosion behavior of PH13-8Mo steel in acidic solution was investigated by using the electrochemical tests, scanning electron microscopy and X-ray photoelectron spectroscopy. The PH13-8Mo martensitic precipitation hardened stainless steel is in the passivity state when the pH value is above 3.0, below which the anodic polarization curves of the steel are actively dissolved. The corrosion current density gradually decreases with increasing the solution pH and decreasing Cl− concentration and solution temperature. Pits are initiated on the sample surface in the presence of the Cl− and gradually developed into uniform corrosion with increasing the Cl− concentrations. Moreover, the corrosion is more serious with an increase in solution temperature.
Similar content being viewed by others
References
S. P. Brühl, R. Charadia, S. Simison, D. G. Lamas, A. Cabo, Surf. Coat. Technol. 204 (2010) 3280–3286.
H. Zhang, Y. L. Zhao, Z. D. Jiang, Mater. Lett. 59 (2005) 3370–3374.
D. Nakhaie, M. H. Moayed, Corros. Sci. 80 (2014) 290–298.
J. Mittra, G. K. Dey, D. Sen, A. K. Patra, S. Mazumder, P. K. De, Scripta Mater. 51 (2004) 349–353.
Z. Guo, W. Sha, D. Vaumousse, Acta Mater. 51 (2003) 101–116.
H. Leitner, R. Schnitzer, M. Schober, S. Zinner, Acta Mater. 59 (2011) 5012–5022.
M. Jong, F. Schmalz, J. W. Rensman, N. V. Luzginova, O. Wouters, J. B. J. Hegeman, J. G. Vander, J. Nucl. Mater. 417 (2011) 145–148.
Y. B. Hu, C. F. Dong, M. Sun, K. Xiao, P. Zhong, X. G. Li, Corros. Sci. 53 (2011) 4159–4165.
P. Ghods, O. B. Isgor, G. A. Mcrae, G. P. Gu, Corros. Sci. 52 (2010) 1649–1659.
D. Cicolin, M. Trueba, S. P. Trasatti, Electrochim. Acta 124 (2014) 27–35.
B. Zaid, D. Saidi, A. Benzaid, S. Hsdji, Corros. Sci. 50 (2008) 1841–1847.
D. Thirumalaikumarasamy, K. Shanmugam, V. Balasubramanian, J. Magnesium Alloy. 2 (2014) 325–334.
C. Cao, M. M. S. Cheung, Constr. Build. Mater. 51 (2014) 75–81.
V. S. Raja, B. S. Padekar, Corros. Sci. 75 (2013) 176–183.
S. M. A. El-Haleem, S. A. El-Wanees, Mater. Chem. Phys. 128 (2011) 418–426.
H. S. Klapper, J. Goellner, A. Burkert, A. Heyn, Corros. Sci. 75 (2013) 239–247.
Y. Wang, G. Cheng, W. Wu, Y. Li, X. Li, Appl. Surf. Sci. 349 (2015) 746–756.
S. Liu, H. Sun, L. Sun, H. Fan, Corros. Sci. 65 (2012) 520–527.
M. C. Zhao, M. Liu, G. L. Song, A. Atrens, Corros. Sci. 50 (2008) 3168–3178.
D. N. Zou, R. Liu. J. Li, W. Zhang, D. Wang, Y. Han, J. Iron Steel Res. Int. 21 (2014) 630–636.
M. J. Carmezim, A. M. Simoes, M. F. Montemor, M. D. C. Belo, Corros. Sci. 47 (2005) 581–591.
J. Kang, J. Li, K. Y. Zhao, X. Bai, Q. L. Yong, J. Su. J. Iron Steel Res. Int. 22 (2015) 1156–1163.
A. A. A. Azim, S. H. Sanad, Electrochim. Acta 17 (1972) 1699–1704.
I. Nicic, D. D. Macdonald, J. Nucl. Mater. 379 (2008) 54–58.
A. Fattah-Alhosseini, M. A. Golozar, A. Saatchi, K. Raeissi, Corros. Sci. 52 (2010) 205–209.
S. Yang, D. D. Macdonald, Electrochim. Acta 52 (2007) 1871–1879.
J. Kang, Y. Yang, X. Jiang, H. Shao, Corros. Sci. 50 (2008) 3576–3580.
P. Zhong, K. Xiao, C. Dong, J. Zhong, M. Sun, X. Li, Microstructure, Properties and Corrosion Behavior of Ultra-high Strength Steel, Science Press, Beijing, 2014 (in Chinese).
M. Bojinov, G. Fabricius, P. Kinnunen, T. Laintinen, K. Makela, T. Saario, G. Sundholm, Electrochim. Acta 45 (2000) 2791–2802.
A. Pardo, M. C. Merino, A. E. Coy, F. Viejo, R. Arrabal, E. Matykina, Corro. Sci. 50 (2008) 780–794.
M. Keddam, O. R. Mattos, H. Takenouti, Electrochim. Acta 31 (1986) 1159–1165.
I. Annergren, M. Keddam, H. Takenouti, D. Thierry, Electrochim. Acta 38 (1993) 763–771.
X. C. Han, J. Li, K. Y. Zhao, W. Zhang, J. Su, J. Iron Steel Res. Int. 20 (2013) No. 5, 74–79.
R. M. Fernández-Domene, E. Blasco-Tamarit, D. M. García-García, J. Garcia-Anton, Electrochim. Acta 95 (2013) 1–11.
C. V. Vidal, A. I. Muñoz, Electrochim. Acta 55 (2011) 8445–8452.
P. Kwakhong, A. Artnaseaw, C. Kruehong, J. Iron Steel Res. Int. 22 (2015) 746–751.
L. Zhang, X. G. Li, C. W. Du, J. Iron Steel Res. Int. 16 (2009) No. 6, 52–57.
A. Fattah-Alhosseini, F. Soltani, F. Shirsalimi, B. Ezadi, N. Attarzadeh, Corros. Sci. 53 (2011) 3186–3192.
L. W. Tsay, H. L. Lu, C. Chen, Corros. Sci. 50 (2008) 2506–2511.
A. Di Paola, Electrochim. Acta 34 (1989) 203–210.
H. H. Mao, X. Qi, J. Cao, L. C. An, Y. T. Yang, J. Iron Steel Res. Int. 24 (2017) 561–566.
J. Huang, X. Wu, E. H. Han, Corros. Sci. 52 (2010) 3444–3452.
J. Huang, X. Wu, E. H. Han, Corros. Sci. 51 (2009) 2976–2982.
Y. F. Cheng, F. R. Steward, Corros. Sci. 46 (2004) 2405–2420.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, Xy., Fan, Ch., Wu, Ql. et al. Effect of solution pH, Cl− concentration and temperature on electrochemical behavior of PH13-8Mo steel in acidic environments. J. Iron Steel Res. Int. 24, 1238–1247 (2017). https://doi.org/10.1016/S1006-706X(18)30023-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1006-706X(18)30023-2