Abstract
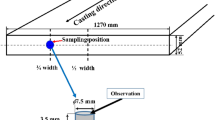
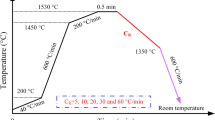
The radial growth of δ-phase in Fe-0.15%C-0.8%Mn steel during solidification was in-situ observed under a high-temperature confocal scanning laser microscope (HTCSLM). The correlation between radial growth rate of cylindrical solid and time in melt was investigated, and the expression was deduced. The results indicate that the radius of cellular δ-phase rapidly enlarges at the beginning, and then the enlargement amplitude gradually declines. The variation of radial growth rate vs time is the same for each cellular δ-phase, and the radial growth rate of δ-phase rapidly declines to about 1.5 μm/s within 10 s at a cooling rate of 2. 7 K/min, after that the growth rate slowly falls. The experimental data of the radial growth rate of cellular δ-phase are consistent with the calculation results for Fe-0.15%C-0.8%Mn steel.
Similar content being viewed by others
References
Beatty K M, Jackson K A. Orientation Dependence of the Distribution Coefficient Obtained From a Spin-1 Ising Model [J]. Journal of Crystal Growth, 1997, 174(1/2/3/4): 28.
Tiller W A, Jackson K A, Rutter J W, et al. The Redistribution of Solute Atoms During the Solidification of Metals [J]. Acta Metall, 1953, 1(4): 428.
Mullins W W, Sekerka R F. Stability of a Panar Interface During Solidification of a Dilute Alloy [J]. J Appl Phys, 1964, 35(2): 444.
Langr J S, Muller-Krumbhaar H. Theory of Dendritic Growth-II. Instabilities in the Limit of Vanishing Surface Tension [J]. Acta Metall, 1978, 26(11): 1689.
Kurz W, Fisher D J. Dendrite Growth at the Limit of Stability: Tip Radius and Spacing [J]. Acta Metall, 1981, 29(1): 11.
Trivedi R. Theory of Dendritic Growth During the Directional Solidification of Binary Alloys [J]. Journal of Crystal Growth, 1980, 49(2): 219.
CHANG Guo-wei, JIN Guang-can, CHEN Shu-ying, et al. Study on Lateral Growth Rate of Peritectic Reaction Products [J]. Acta Metallurgica Sinica, 2011, 47(3): 380 (in Chinese).
Shibata H, Arai Y, Suzuki M, et al. Kinetics of Peritectic Reaction and Transformation in Fe-C Alloys [J]. Metallurgical and Materials Transactions, 2000, 31B(5): 981.
Mcdonald N J, Sridhar S. Peritectic Reaction and Solidification in Iron-Nickel Alloys [J]. Metallurgical and Materials Transactions, 2003, 34A(9): 1931.
Coriell S R, Paker R L. Stability of the Shape of a Solid Cylinder Growing in Diffusion Field [J]. J Appl Phys, 1965, 36(2): 632.
Hardy S D, Coriell S R. Morphological Stability of Cylindrical Ice Crystals [J]. Journal of Crystal Growth, 1969, 5(5): 329.
Trivedi R, Somboonsuk K. Pattern Formation During the Directional Solidification of Binary Systems [J]. Acta Metallurgica, 1985, 33(6): 1061.
Zhang D, Terasaki H, Komizo Y. In Situ Observation of the Formation of Intragranular Acicular Ferrite at Non-metallic Inclusions in C-Mn Steel [J]. Acta Materialia, 2010, 58(4): 1369.
Zhang X F, Terasaki H, Komizo Y, et al. In Situ Observation of High Temperature Creep Behavior During Annealing of Steel [J]. Metallurgical and Materials Transactions, 2012, 43A(13): 5067.
Song C J, Guo Y Y, Li K F, et al. In Situ Observation of Phase Transformation and Structure Evolution of a 12 pet Cr Ferritic Stainless Steel [J]. Metallurgical and Materials Transactions, 2012, 43B(5): 1127.
Zhang D, Shintaku Y, Suzuki S, et al. In Situ Observation of Phase Transformation in Low-Carbon, Boron-Treated Steels [J]. Metallurgical and Materials Transactions, 2012, 43A(2): 447.
Kurz W, Fisher D J. Fundamentals of Solidification [M]. Switzerland: Trans Tech Publications, 1998.
Phelan D, Reid M, Dippenaar R. Kinetics of the Peritectic Phase Transformation: In-Situ Measurements and Phase Field Modeling [J]. Metallurgical and Materials Transactions, 2006, 37A(3): 985.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation Item: Item Sponsored by National Natural Science Foundation of China (50874060); Program of Excellent Talents of Liaoning Province in University of China (LR201019)
Rights and permissions
About this article
Cite this article
Chen, Sy., Chang, Gw., Yue, Xd. et al. Correlation Between Radial Growth Rate of Cylindrical Solid and Time in Melt. J. Iron Steel Res. Int. 20, 1–6 (2013). https://doi.org/10.1016/S1006-706X(13)60188-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1006-706X(13)60188-0