Abstract
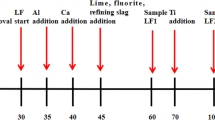
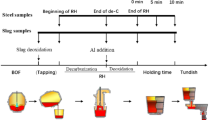
Titanium is the impurity in some special steel grades. The existence of titanium decreases the grain size, depresses the yield strength, and results in the low quality of these steels in various properties. Thus, titanium should be removed to the minimum. Based on the industrial production of ultra low carbon Al-Si killed steel, the physical-chemical behavior of titanium was investigated in vacuum degassing refining (RH) process with and without desulfurization. The influences of titanium content in hot metal, ladle slag composition, and ladle slag quantity, etc, on titanium content of refined liquid steel were discussed. The results showed that the partition ratio of titanium between ladle slag and liquid steel is in inverse proportion to the 4/3 square of aluminum content. The maximum partition ratio of titanium between top slag and liquid steel can be obtained by adjusting an optimum slag composition including contents of FeOx and A12O3 and the slag basicity, and the suitable range of them should be controlled higher than 6%, less than 20%, and within 1. 5 to 3. 0, respectively. Moreover, desulfurization refining by RH decreases the partition ratio of titanium between ladle slag and liquid steel significantly. To ensure the titanium content stably less than 15 × 10−6 in a 300 t ladle, the titanium content in hot metal must be less than 500 × 10−6 and the thickness of basic oxygen furnace (BOF) slag carrying over must be less than 50 mm.
Similar content being viewed by others
Abbreviations
- yx:
-
Activity coefficient of x in slag relative to pure solid
- fx:
-
Activity coefficient of x in infinitely diluted liquid alloy based on mass percentage concentration
- ax:
-
Activity of x in infinitely diluted liquid alloy based on mass percentage concentration
- axy:
-
Activity of xy in slag relative to pure solid
- xy:
-
Mole fraction of y in slag relative to pure solid
- K:
-
Equilibrium constant
- ΔG:
-
Change of standard Gibbs free energy
- nx:
-
Total mole number of oxides x in 100 g slag
- Mx:
-
Molecular mass of x
References
Ohta M, Morita K. Interaction Between Silicon and Titanium in Molten Steel [J]. ISIJ Int, 2003, 43(2): 256.
Jung S M, Fruehan R J. Thermodynamics of Titanium Oxide in Ladle Slags [J]. ISIJ Int, 2001, 41(12): 1447.
LIU Yue, WU Wei, LIU Liu, et al. Thermodynamics Behavior of Titanium for BOF Smelting Bearing Steel [J]. Journal of Iron and Steel Research, International, 2006, 13(6): 74.
Okamura K. Method of Steelmaking With Low Titanium Content: Japan, 2003-73726A [P]. 2003-03-12 (in Japanese).
Miyamoto K, Kanno H, Masao Y. Method of Steelmaking for Steels With Very Low Titanium Content: Japan, 2004-307942A [P]. 2004-11-04 (in Japanese).
Ohta H, Suito H. Activities of SiO2 and A12O3 and Activity Coefficients of FetO and MnO in CaO-SiOz-Al203-MgO Slags [J]. Met Mat Trans, 1998, 29B: 119.
Fruehan R J. Over View of Steelmaking Processes and Their Development [C] // The Making, Shaping and Treating of Steel. Pittsburgh: AISE Steel Foundation, 1998: 1.
Pak J J, Yoo J T, Jeong Y S, et al. Thermodynamics of Titanium and Nitrogen in Fe-Si Melt [J]. ISIJ Int, 2005, 45(1): 23.
Kishi M, Inoue R, Suito H. Thermodynamics of Oxygen and Nitrogen in Liquid Fe-20 Mass% Cr Alloy Equilibrated With Titania-Based Slags [J]. ISIJ Int, 1994, 34(11): 859.
Sigworth G K, Elliott J F. The Thermodynamics of Liquid Dilute Iron Alloys [J]. Met Sci, 1974(8): 298.
The Japan Society for the Promotion of Science, The 19th Com mittee on Steelmaking. Steelmaking Data Sourcebook [M]. New York: Gordon at Breach Science Publishers, 1998.
Oojima K, Masahiro S. Smelting Method of Steels With Very Low Titanium Content: Japan, H05086413 [P]. 1993-04-06 (in Japanese).
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation Item:Item Sponsored by National Natural Science Foundation of China (51104109)
Rights and permissions
About this article
Cite this article
Zhang, F., Li, Gq. Control of Ultra Low Titanium in Ultra Low Carbon Al-Si Killed Steel. J. Iron Steel Res. Int. 20, 20–25 (2013). https://doi.org/10.1016/S1006-706X(13)60077-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1006-706X(13)60077-1