Abstract
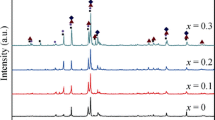
As the alloy with the most suitable Ni/(La+Mg) ratio has higher capacity and good cycle stability, the effects of Ni/(La+Mg) ratios on the electrochemical performances of the La0.80Mg0.20Nix (x = 3.5 to 5.0) alloys have been investigated to find the most suitable Ni/(La+Mg) ratio. The results of XRD and SEM observations show that the phase composition of the alloys varies with different Ni/(La+Mg) ratios. When Ni/(La+Mg) is not more than 4.25, all the alloys contain LaNi5 and (La, Mg)2Ni7 phases, in addition, the LaMg and (La, Mg)Ni3 phases exist in the x = 3.5 and 3.75 alloys, respectively. The LaMg2Ni9 phase exists in the x = 4.25 alloy. There are the LaNi5 and LaMg2Ni9 phases in the x = 4.5, 4.75, and 5.0 alloys. The phase abundance and cell volume change with different Ni content. When the Ni/(La+Mg) ratio is not more than 4.25, the alloys possess excellent activation capability, however, the activation capabilities of the alloys decrease with a further increase in the Ni/(La+Mg) ratio. With increasing the Ni/(La+Mg) ratio, the maximum discharge capacities, the medium voltages, and the cycle stabilities of the alloys first increase and then decrease. When the Ni/(La+Mg) ratio is 3.75, the corresponding alloy has the maximum discharge capacity among all the alloys. However, the cycle stability of the Ni/(La+Mg) = 4.0 alloy is better than that of the others.
Similar content being viewed by others
References
CHEN J, Takeshita H T, Tanaka H, et al. Hydriding Properties of LaNi3 and CaNi3 and Their Substitutes With PuNi3-Type Structure [J]. J Alloys Compd, 2000, 302: 304.
PAN H G, LIU Y F, GAO M X, et al. An Investigation on the Structural and Electrochemical Properties of La0.7Mg0.3 (Ni0.85Co0.15)x(x=3.15−3.80) Hydrogen Storage Electrode Alloys [J]. J Alloys Compd, 2003, 351, 228.
Akiba E, Hayakawa H, Kohno T. Crystal Structures of Novel La-Mg-Ni Hydrogen Absorbing Alloys [J]. J Alloys Compd, 2006, 408–412: 280.
Yasuoka S, Magari Y, Murata T, et al. Development of High-Capacity Nickel-Metal Hydride Batteries Using Superlattice Hydrogen-Absorbing Alloys [J]. J Power Sources, 2006, 156: 662.
Kohno T, Yoshida H, Kawashima F, et al. Hydrogen Storage Properties of New Ternary System Alloys: La2MgNi9, La5Mg2Ni23, La3MgNi14 [J]. J Alloys Comp, 2000, 311: L5.
TANG R, LIU L Q, LIU Y N, et al. Study on the Microstructure and the Electrochemical Properties of Ml0.7Mg0.2Ni2.8Co0.6 Hydrogen Storage Alloys [J]. Int J Hydrogen Energy, 2003, 28: 815.
CHANG F H. A New X-Ray Diffraction Method for Quantitative Multicomponent Analysis [J]. Advances in X-Ray Analysis, 1974, 17: 106.
Kadir K, Sakai T, Uehara T, et al. Structural Investigation and Hydrogen Capacity of LaMg2Ni9 and (La0.65Ca0.35)(Mg1.32Ca0.58)Ni9 of the AB2C9 Type Structure [J]. J Alloys Comp, 2000, 302: 112.
LIU Y F, PAN H G, YUE Y J, et al. Cycling Durability and Degradation Behavior of La-Mg-Ni-Co-Type Metal Hydride Electrodes [J]. J Alloys Compd, 2005, 395: 291.
Willems J J G. Metal Hydride Electrodes Stability of LaNi5-Related Compounds [J]. Philips J Res, 1984, 39(Supplement): 1.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation Item: Item Sponsored by National Natural Science Foundation of China (50642033) ; Natural Science Foundation of Inner Mongolia of China (200711020703) ; Science and Technology Planned Project of Inner Mongolia of China (20050205)
Rights and permissions
About this article
Cite this article
Dong, Xp., Yang, Ly., Zhang, Yh. et al. Effect of Ni/(La+Mg) ratio on structure and electrochemical performance of La-Mg-Ni alloy system. J. Iron Steel Res. Int. 16, 83–88 (2009). https://doi.org/10.1016/S1006-706X(09)60049-2
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1016/S1006-706X(09)60049-2