Abstract
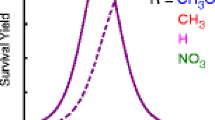
Metastable ion decompositions, collision-activated dissociation (CAD), and neutralization-reionization mass spectrometry are utilized to study the unimolecular chemistry of distonic ion ·CH2CH2CH−OH (2+·) and its enol-keto tautomers CH3CH=CHOH−· (1 +·) and CH3CH2CH=O +· (3+·). The major fragmentation of metastable 1+·–3+· is H· loss to yield the propanoyl cation, CH3CH2C≡O+. This reaction remains dominant upon collisional activation, although now some isomeric CH2=CH-CH+ OH is coproduced from all three precursors. The CAD and neutralization-reionization (+NR+) spectra of keto ion 3 +· are substantially different from those of tautomers 2+· and 1+·. Hence, 3+· without sufficient energy for decomposition (i. e. , “stable” 3+·) does not isomerize to the ther-modynamically more stable ions 2+· or 1+·, and the 1,4-H rearrangement H-CH2CH2CH=O+·(3 +·) → CH2CH2CH+ O-H (2 +·) must require an appreciable critical energy. Although the fragment ion abundances in the + NR + (and CAD) spectra of 1 +· and 2 +· are similar, the relative and absolute intensities of the survivor ions (recovered C3H6O+· ions in the +NR+ spectra) are markedly distinct and independent of the internal energy of 1 +· and 2 +·. Furthermore, 1 +· and 2 +· show different MI spectra. Based on these data, distonic ion 2 +· does not spontaneously rearrange to enol ion 1 +· (which is the most stable C3H6O+· of CCCO connectivity) and, therefore, is separated from it by an appreciable barrier. In contrast, the molecular ions of cyclopropanol (4 +·) and allyl alcohol (5 +·) isomerize readily to 2 +·, via ring opening and 1,2-H− shift, respectively. The sample found to generate the purest 2 +· is α-hydroxy-γ-butyrolactone. Several other precursors that would yield 2 +· by a least-motion reaction cogenerate detectable quantities of enol ion 1 +·, or the enol ion of acetone (CH2=C(CH3)OH+·, 6 +·), or methyl vinyl ether ion (CH3OCH=CH +·2 , 7 +·). Ion 6 +· is coproduced from samples that contain the —CH2—CH(OH)—CH2— substructure, whereas 7 +· is coproduced from compounds with methoxy substituents. Compared to CAD, metastable ion characteristics combined with neutralization-reionization allow for a superior differentiation of the ions studied.
Article PDF
Similar content being viewed by others
References
McLafferty, F. W.; Stauffer, D. B. Wiley/NBS Registry of Mass Spectral Data; Wiley: New York, 1989
Bouma, W. J.; MacLeod, J. K.; Radom, L. J. Am. Chem. Soc. 1980, 102, 2246–2252.
Hoppilliard, Y.; Bouchoux, G.; Jaudon, P. Nouv. J. Chim. 1982, 6, 43–52.
Bouchoux, G.; Hoppilliard, Y. Int. J. Mass Spectrom. Ion Processes 1983/1984, 55, 47–53.
Bouchoux, G.; Tortajada, J. Rapid Commun. Mass Spectrom. 1987, 1, 86–87.
Hudson, C. E.; McAdoo, D. J. Tetrahedron 1990, 46, 331–334.
Van de Sande, C. C.; McLafferty, F. W. J. Am. Chem. Soc. 1975, 97, 4617–4620.
McAdoo, D. J.; Witiak, D. K. J. Chem. Soc., Perkin Trans. 1981, 2, 770–773.
Hudson, C. E.; McAdoo, D. J. Org. Mass Spectrom. 1982, 17, 366–368.
Hudson, C. E.; McAdoo, D. J. Org. Mass Spectrom. 1984, 19, 1–6.
Turecek, F.; Hanus, V.; Gäumann, T. I J. Mass Spectrom. Ion Processes 1986, 69, 217–231.
McAdoo, D. J.; Hudson, C. E.; Traeger, J. C. Org. Mass Spectrom. 1988, 23, 760–764.
Gu, M.; Turecek, F. Org. Mass Spectrom. 1994, 29, 85–89.
McAdoo, D. J.; Hudson, C. E. J. Mass Spectrom. 1995, 30, 492–496.
Lias, S. G.; Liebman, J. F.; Levin, R. D.; Kafafi, S. A. NIST Standard Reference Database 25, version 2.02, 1994;
Lias, S. G.; Bartmess, J. E.; Liebman, J. F.; Holmes, J. L.; Levin, R. D.; Mallard, W. G.; J. Phys. Chem. Ref. Data 1988, Suppl. 1.
Wesdemiotis, C.; McLafferty, F. W. Tetrahedron Lett. 1981, 22, 3479–3480.
Bouchoux, G. Mass Spectrom. Rev. 1988, 7, 1–39.
Hammerum, S. Mass Spectrom. Rev. 1988, 7, 123–202.
Stirk, K. M.; Kiminkinen, M.; Kenttämaa, H. I. Chem. Rev. 1992, 92, 1649–1665.
Turecek, F.; McLafferty, F. W. J. Am. Chem. Soc. 1984, 106, 2528–2531.
Poke, M. J.; Wesdemiotis, C. J. Am. Soc. Mass Spectrom. 1995, 6, 1030–1036.
Polce, M. J.; Wiedmann, F. A.; Wesdemiotis, C. Proceedings of the 41st ASMS Conference, San Francisco, CA, May 31–June 4, 1993, pp 283a–283b.
Polce, M. J.; Wesdemiotis, C. Proceedings of the 42nd ASMS Conference, Chicago, IL, May 29–June 3, 1994, p 206.
Busch, K. L.; Glish, G. L.; McLuckey, S. A. Mass Spectrometry/Mass Spectrometry; VCH Publishers, Inc.; New York, 1988.
Wesdemiotis, C.; McLafferty, F. W. Chem. Rev. 1987, 87, 485–500.
Terlouw, J. K.; Schwarz, H. Angew. Chem. Int. Ed. Engl. 1987, 26, 805–815.
Holmes, J. L. Mass Spectrom. Rev. 1989, 8, 513–539.
Poke, M. J.; Cordero, M. M.; Wesdemiotis, C.; Bott, P. A. Int. J. Mass Spectrom. Ion Processes 1992, 113, 35–58.
Bouchoux, G.; Alcaraz, C.; Dutuit, O.; Nguyen, M. T. Int. J. Mass Spectrom. Ion Processes 1994, 137, 93–106.
Zeller, L.; Farrell, J. Jr.; Vainiotalo, P.; Kenttämaa, H. I. J. Am. Chem. Soc. 1992, 114, 1205–1214.
Kurland, J. J.; Lutz, R. P. J. Chem. Soc. Chem. Commun. 1968, 1097–1098.
Holmes, J. L.; Burgers, P. C.; Mollah, Y. A. Org. Mass Spectrom. 1982, 27, 127–130.
Bombach, R.; Dannacher, J.; Honegger, E.; Stadelmann, J.-P.; Neier, R. Chem. Phys. 1983, 82, 459–470.
Bouchoux, G.; Flammang, R.; Maquestiau, A. Org. Mass Spectrom. 1985, 20, 154–155.
Borden, W. T., Ed. Diradicals; Wiley: New York, 1982.
Wentrup, C. Reactive Molecules: The Neutral Reactive Intermediate in Organic Chemistry; Wiley-Interscience: New York, 1984; pp 128–161.
Cordero, M. M.; Houser, J. J.; Wesdemiotis, C. Anal. Chem. 1993, 65, 1594–1601.
Danis, P. O.; Feng, R.; McLafferty, F. W. Anal. Chem. 1985, 58, 348–354;
Hop, C. E. C. A.; Holmes, J. L. Org. Mass Spectrom. 1991, 26, 476–480.
Burgers, P. C.; Holmes, J. L.; Mommers, A. A.; Terlouw, J. K. Chem. Phys. Lett. 1983, 102, 1–3.
Cooks, R. G.; Beynon, J. H.; Caprioli, R. M.; Lester, G. R. Metastable Ions; Elsevier: Amsterdam, 1973.
Holmes, J. L. Org. Mass Spectrom. 1985, 20, 169–183.
Magrane, J. K.; Cottle, D. L. J. Am. Chem. Soc. 1942, 64, 484–487.
Stahl, C. W.; Cottle, D. L. J. Am. Chem. Soc. 1943, 65, 1782–1783.
Roberts, J. D.; Chambers, V. C. J. Am. Chem. Soc. 1951, 73, 3176–3179.
Ploce, M. J.; Wesdemiotis, C. J. Am. Chem. Soc. 1993, 115, 10849–10856.
Holmes, J. L.; Lossing, F. P. J. Am. Chem. Soc. 1980, 102, 1591–1595.
Levsen, K. Fundamental Aspects of Organic Mass Spectrometry; Verlag Chemie: Weinheim, 1978.
Bouchoux, G.; Hoppilliard, Y.; Flammang, R.; Maquestiau, A.; Meyrant, P. Org. Mass Spectrom. 1983, 18, 340–344.
Burgers, P. C.; Mommers, A. A.; Holmes, J. L. J. Am. Chem. Soc. 1983, 105, 5976–5979.
McLafferty, F. W.; Turecek, F. Interpretation of Mass Spectra, 4th ed.; University Science Books: Mill Valley, CA, 1993.
Kimura, K.; Katsumata, S.; Achiba, Y.; Yamazaki, T.; Iwata, S. Handbook of HeI Photoelectron Spectra of Fundamental Organic Molecules; Halsted Press: New York, 1981.
McAdoo, D. J. Mass Spectrom. Rev. 1988, 7, 363–393.
McAdoo, D. J.; Hudson, C. E.; Traeger, J. C. Int. J. Mass Spectrom. Ion Processes 1987, 79, 183–187.
Lifshitz, C.; Tzidony, E. Int. J. Mass Spectrom. Ion Phys. 1981, 39, 181–195;
Turecek, F.; McLafferty, F. W. J. Am. Chem. Soc. 1984, 106, 2525–2528;
McAdoo, D. J.; Hudson, C. E. Int. J. Mass Spectrom. Ion Processes 1984, 59, 77–83.
Turecek, F.; McLafferty, F. W. J. Am. Chem. Soc. 1984, 106, 2528–2531
Lifshitz, C. Org. Mass Spectrom. 1988, 23, 303–306.
Danis, P. O.; Wesdemiotis, C.; McLafferty, F. W. J. Am. Chem. Soc. 1983, 105, 7454–7456;
Terlouw, J. K.; Kieskamp, W. M.; Holmes, J. L.; Mommers, A. A.; Burgers, P. C. Int. J. Mass Spectrom. Ion Processes 1985, 64, 245–250.
Gilpin, J. A.; McLafferty, F. W. Anal. Chem. 1957, 29, 990–994.
Drewello, T.; Heinrich, N.; Maas, W. P. M.; Nibbering, N. M. M.; Weiske, T.; Schwarz, H. J. Am. Chem. Soc. 1987, 109, 4810–4818.
Goldberg, N.; Schwarz, H. Acc. Chem. Res. 1994, 27, 347–352.
Pedersen, S.; Herek, J. L.; Zewail, A. H. Science 1994, 266, 1359–1364.
Zagorevskii, D. V.; Holmes, J. L. Mass Spectrom. Rev. 1994, 13, 133–154.
Cohen, N.; Benson, S. W. Chem. Rev. 1993, 93, 2419–2438.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Polce, M.J., Wesdemiotis, C. The distonic ion ·CH2CH2CH+OH, keto ion CH3CH2CH=O +·, enol ion CH3CH=CHOH+·, and related C3H6O+· radical cations. Stabilities and isomerization proclivities studied by dissociation and neutralization-reionization. J Am Soc Mass Spectrom 7, 573–589 (1996). https://doi.org/10.1016/1044-0305(96)00053-0
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/1044-0305(96)00053-0