Abstract
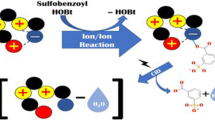
The behavior of para-hydroxy-benzyl and hydroxy-phenylethyl fatty acid esters and methoxy derivatives toward the NH3/NH2 − system was investigated. Under these negative ion chemical ionization (NICI) conditions, proton abstraction takes place mainly at the more acidic site (i.e., phenol); however, this reaction is not entirely regioselective. Using NICI-ND3 conditions, both isomeric phenoxide and enolate molecular species are produced in competition from these phenol esters. Their respective low-energy collision-activated dissociation spectra are studied, and they strongly differ, showing that these molecular species are not convertible to a common structure. Analysis of specific fragmentations of the OD-enolate parent species labeled by ND3 in the gas phase, indicates that by charge-promoted cleavage, isomerization into an ion-dipole intermediate takes place prior to dissociation. This complex, containing a ketene moiety, isomerizes into different isomeric forms via two consecutive proton transfers: the first, which is very exothermic, is irreversible in contrast to the second, less exothermic reaction, which occurs via a reversible process. It is evidenced by the loss of labeling at phenol or enolizable sites in the fragment ions. Such a stepwise process does not take place from the phenoxide parent ion, which preferentially yields a very stable carboxylate ion. A thermochemical approach, using estimated acidity values, yields a rationalization of the observed reactivities of the various substrates studied.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
McAdoo, D. J. Muss Spectrom. Rev. 1988, 7, 367; (b) Longevialle, P.; Boiler, R. J. Chem. Soc., Chem. Com mutt. 1980, 823; (c) Bowen, R. D.; Williams, D. H. J. Chem. Soc. Chem. Commun. 1980, 823; (d) Morton, T. H. Tetrahedron 1982, 38, 3195.
Bowie, J. H. In Mass Spectrometry; Rose, M. E., Ed. (Spetialist Periodical Reports). The Royal Society of Chemistry; London, 1989; Vol. 10, p. 145; (b) Bowie, J. H. Mass Spectrom. Rev. 1990, 9, 349.
Hayes, R. N.; Sheldon, J. C.; Bowie, J. H.; Lewis, D. E. J. Chem. Soc., Chem. Commun. 1984, 1431.
Budzikiewicz, H. Mass Spectrom. Rev. 1990, 9, 349.
Donnelli, A.; Chowdhury, S. K.; Harrison, A. G. Org. Mass Spectrom. 1989, 24, 89.
Prome, J. C.; Aurelle, H.; Prome, D.; Savignae, A. Org. Mass. Spectrom. 1987, 26, 6.
Cole, R. B.; Tabet, J. C.; Salles, C.; Jallageas, J. C.; Crouzet, J. Rapid Commun. Mass Spectrom. 1989, 3, 59.
Eckesley, M.; Bowie, J. H.; Hayes, R. N. Int. J. Mass Spectrom. Ion Processes 1989, 93, 199.
Waugh, R. J.; Bowie, J. H.; Hayes, R. N. Org. Mass Spectrom. 1991, 26, 250.
Fournier, F.; Salles, C.; Tabet, J. C.; Debrauwer, L.; Rao, D.; Paris, A.; Bories, G. Anal. Chim. Acta 1990, 241, 265.
Fournier, F.; Tabet, J. C.; Debrauwer, L.; Rao, D.; Paris, A.; Bories, G. Rapid Commun. Mass Spectrom. 1991, 6, 44.
Hayes, R. N.; Bowie, J. H. J. Chem. Soc., Perkin Trans II 1986, 1827; (b) Stringer, M. B.; Bowie, J. H.; Eichinger, P. C. H.; Currie, G. J. J. Chem. Soc., Perkin Trans II 1987, 385.
Harrison, A. G.; Young, R. B. Int. J. Mass Spectrom. Ion Processes 1989, 94, 321.
Beloeil, J. C.; Bertranne, M.; Stahl, D.; Tabet, J. C. J. Am. Chem. Soc. 1983, 105, 1355; (b) Hunt, D. F.; Sethi, S. K.; Shabanowitz, J. Environ. Health Perspect. 1980, 36, 33.
Lias, S. G.; Bartmess, J. E.; Liebman, J. L.; Holmes, J. L.; Levin, R. D.; Mallard, W. G. J. Phys. Chem. Ref. Data 1988, 17, Suppl. 1.
Boand, G.; Houriet, R.; Gaumann, T. J. Am. Chem. Soc. 1983, 205, 2203.
Harrison, A. G. In Fundamentals of Cas Phase Ion Chemistry; Jenning, K., Ed.; NATO ASI Series, Series C., Vol. 347, p. 289, 1991; (b) Majumdar, K. T.; Clairet, F.; Tabet, J. C.; Cooks, R. G. J. Am. Chem. Soc. 1992, 114, 2897.
Sürig, T.; Grützmacher, H. F. Org. Mass Spectrom. 1989, 24, 851.
Raftery, H. J.; Bowie, J. H.; Sheldon, J. C. J. Chem. Soc., Perkin Trans II 1988, 363.
Graul, S. T.; Schnute, M. E.; Squires, R. R. Int. Mass Spectrom. Ion Processes 1990, 96, 1981.
Eichinger, P. C. H.; Hayes, R. N. N.; Bowie, J. H. J. Am. Chem. Soc. 1991, 113, 1949.
Chowdhury, S. K.; Harrison, A. G. Org. Mass Spectrom. 1989, 24, 123.
Grutzmacher, H. F.; Mazur, A. Org. Mass Spectrom. 1988, 23, 223.
Chowdhury, S. K.; Harrison, A. G. Int. J. Mass Spectrom. Ion Processes 1989, 92, 135.
Melton-Nussbaum, S. H.; Ponticarvo, L.; Schatz, F; Hochberg R. B. J. Biol. Chem. 1982, 70, 5678.
Vetter, W.; Meister, W.; Oesterholf, G. Org. Mass Spectrom. 1988, 23, 566.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fournier, F., Remaud, B., Blasco, T. et al. Ion-dipole complex formation from deprotonated phenol fatty acid esters evidenced by using gas-phase labeling combined with tandem mass spectrometry. J Am Soc Mass Spectrom 4, 343–351 (1993). https://doi.org/10.1016/1044-0305(93)85057-5
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/1044-0305(93)85057-5