Abstract
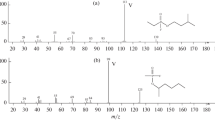
Long-chain aldehydes, encountered as insect pheromones, were converted into Schiff bases with 3-aminopyridine, 3-(aminomethyl)pyridine, or 2-aminopyrimidine to provide derivatives suitable for revealing the alkyl chain structure by mass spectrometry. The two pyridine-containing derivatives were satisfactory in initiating a radical-induced cleavage. of the chain to give a series of fragment ions, the masses and relative abundance of which revealed the chain structure. The derivatives were applied to aldehydes having straight, branched (iso and anteiso), and unsaturated (delta-7, delta-9, delta-ll, and delta-13) structures; these all gave the fragmentation patterns that have been seen earlier for similar pyridine-containing derivatives of fatty acids (picolinyl esters) and alcohols (nicotinates).Of the two derivatives, those from 3-aminopyridine gave slightly simpler spectra. Derivatives formed from 2-aminopyrimidine were less satisfactory in revealing chain structure.
Article PDF
Similar content being viewed by others
References
Brand, J. M.; Young, J. C.; Silverstein, R. M. Fortschr, Chem. Org. Natural. 1979, 37, 1–190.
Aldrich, J. R.; Lusby. W. R.; Kochansky, P. Experientia 1986, 42, 583–585.
Harvey. D. J. Biomed. Mass Spectrom. 1982, 9, 33–38.
Harvey, D. J. Biomed. Mass Spectrom. 1984 11, 187–192.
Harvey. D. J. Biomed. Mass Spectrom. 1984, 11, 340–347.
Vetter, W.; Meister, W. Org. Mass Spectrom. 1981, 16, 118- 122.
Harvey, D. J.; Tiffany, J. M. Biomed. Mass Spectrom. 1984, 11, 353- 359.
Harvey, D. J. Biomed. Environ. Mass Spectrom. in press.
Christie, W. W.; Brechany, E. Y.; Holman, R. T. Lipids 1987, 22, 224–228.
Harvey, D. J. Biomed. Environ. Mass Spectrom. 1989, 18, 719–723.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Harvey, D.J. Pyridine-Containing schiff base derivatives for the struxtural determination of long-chain aldehydes by gas chromatography combined with mass spectrometry. J Am Soc Mass Spectrom 2, 245–249 (1991). https://doi.org/10.1016/1044-0305(91)80051-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1016/1044-0305(91)80051-8