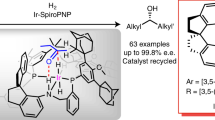
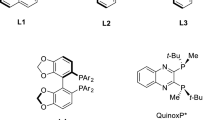
Summary.
A series of novel tridentate ligands with nitrogen and oxygen donor sites was synthesized starting from enantiomerically pure (S)- and (R)-1-(pyridin-2-yl)ethylamine, the preparation and resolution of which was developed. The new optically active ligands were tested as in situ catalysts together with Ru(PPh3)3Cl2 in the enantioselective transfer hydrogenation of acetophenone with isopropanol. The secondary amine ligand (S)-2,4-di-tert-butyl-6-(1-(pyridin-2-yl)ethylamino)methylphenol gave the best results with almost quantitative conversion and 47%ee.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received August 17, 2001. Accepted August 27, 2001
Rights and permissions
About this article
Cite this article
Brunner, H., Niemetz, M. Enantioselective Catalysis CXLI [1]. Tridentate Ligands with 1-(Pyridin-2-yl)ethylamine as Chiral Building Block in the Enantioselective Transfer Hydrogenation of Acetophenone. Monatshefte fuer Chemie 133, 115–126 (2002). https://doi.org/10.1007/s706-002-8241-z
Issue Date:
DOI: https://doi.org/10.1007/s706-002-8241-z