Abstract
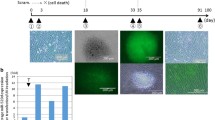
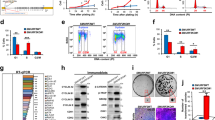
As a member of the SMAD family, SMAD4 plays a crucial role in several cellular biological processes. However, its function in UVB radiation-induced keratinocyte damage is not yet clarified. Our study aims to provide mechanistic insight for the development of future UVB protective therapies and therapeutics involving SMAD4. HaCaT cells were treated with UVB, and the dose dependence and time dependence of UVB were measured. The cell function of UVB-treated HaCaT cells and the activity of epithelial-mesenchymal transition (EMT) after overexpression or silencing of SMAD4 was observed by flow cytometry, quantitative reverse transcription PCR (qRT-PCR) and Western Blots (WB). We found that a significant decrease in SMAD4 was observed in HaCaT cells induced by UVB. Our data confirm SMAD4 as a direct downstream target of miR-664. The down-regulation of SMAD4 preserved the viability of the UVB-treated HaCaT cells by inhibiting autophagy or apoptosis. Furthermore, the silencing of SMAD4 activated the EMT process in UVB-treated HaCaT cells. Down-regulation of SMAD4 plays a protective role in UVB-treated HaCaT cells via the activation of EMT.
Graphical abstract








Similar content being viewed by others
Data availability
Data will be made available on request.
References:
Alkhasawneh, A., Duckworth, L. V., George, T. J., Desai, N. V., Sommerfeld, A. J., Lu, X., & Toro, T. Z. (2016). Clinical, morphologic, and immunophenotypic characteristics of ampullary carcinomas with an emphasis on SMAD4 expression. J Gastrointest Oncol, 7(6), 974–981. https://doi.org/10.21037/jgo.2016.06.14
Amani, H., Shahbazi, M. A., D’Amico, C., Fontana, F., Abbaszadeh, S., & Santos, H. A. (2021). Microneedles for painless transdermal immunotherapeutic applications. Journal of Controlled Release, 330, 185–217. https://doi.org/10.1016/j.jconrel.2020.12.019
Chaudhary, S. C., Waseem, M., Rana, M., Xu, H., Kopelovich, L., Elmets, C. A., & Athar, M. (2017). Naproxen inhibits UVB-induced basal cell and squamous cell carcinoma development in Ptch1(+/-) /SKH-1 hairless mice. Photochemistry and Photobiology, 93(4), 1016–1024. https://doi.org/10.1111/php.12758
Clayton, D. A., Doda, J. N., & Friedberg, E. C. (1974). The absence of a pyrimidine dimer repair mechanism in mammalian mitochondria. Proc Natl Acad Sci U S A, 71(7), 2777–2781. https://doi.org/10.1073/pnas.71.7.2777
Connolly, E. C., Freimuth, J., & Akhurst, R. J. (2012). Complexities of TGF-beta targeted cancer therapy. International Journal of Biological Sciences, 8(7), 964–978. https://doi.org/10.7150/ijbs.4564
Cullinane, C., & Bohr, V. A. (1998). DNA interstrand cross-links induced by psoralen are not repaired in mammalian mitochondria. Cancer Research, 58(7), 1400–1404.
David, C. J., Huang, Y. H., Chen, M., Su, J., Zou, Y., Bardeesy, N., Iacobuzio-Donahue, C. A., & Massagué, J. (2016). TGF-β Tumor Suppression through a Lethal EMT. Cell, 164(5), 1015–1030. https://doi.org/10.1016/j.cell.2016.01.009
Deng, H., Wan, M., Li, H., Chen, Q., Li, R., Liang, B., & Zhu, H. (2021). Curcumin protection against ultraviolet-induced photo-damage in Hacat cells by regulating nuclear factor erythroid 2-related factor 2. Bioengineered, 12(2), 9993–10006. https://doi.org/10.1080/21655979.2021.1994720
Derynck, R., Akhurst, R. J., & Balmain, A. (2001). TGF-beta signaling in tumor suppression and cancer progression. Nature Genetics, 29(2), 117–129. https://doi.org/10.1038/ng1001-117
Diffey, B. L. (2002). Sources and measurement of ultraviolet radiation. Methods, 28(1), 4–13. https://doi.org/10.1016/s1046-2023(02)00204-9
Drigeard Desgarnier, M. C., & Rochette, P. J. (2018). Enhancement of UVB-induced DNA damage repair after a chronic low-dose UVB pre-stimulation. DNA Repair, 63, 56–62. https://doi.org/10.1016/j.dnarep.2018.01.008
Fisher, G. J., Datta, S. C., Talwar, H. S., Wang, Z. Q., Varani, J., Kang, S., & Voorhees, J. J. (1996). Molecular basis of sun-induced premature skin ageing and retinoid antagonism. Nature, 379(6563), 335–339. https://doi.org/10.1038/379335a0
Gravitz, L. (2018). Skin. Nature, 563(7732), S83. https://doi.org/10.1038/d41586-018-07428-4
Hay, E. D. (1995). An overview of epithelio-mesenchymal transformation. Acta Anatomica, 154(1), 8–20. https://doi.org/10.1159/000147748
Hemne, P. S., Kunghatkar, R. G., Dhoble, S. J., Moharil, S. V., & Singh, V. (2017). Phosphor for phototherapy: Review on psoriasis. Luminescence, 32(3), 260–270. https://doi.org/10.1002/bio.3266
Hodorogea, A., Calinescu, A., Antohe, M., Balaban, M., Nedelcu, R. I., Turcu, G., Ion, D. A., Badarau, I. A., Popescu, C. M., Popescu, R., Popp, C., Cioplea, M., Nichita, L., Hulea, I., & Brinzea, A. (2019). Epithelial-Mesenchymal transition in skin cancers: a review. Analytical Cellular Pathology, 2019, 3851576. https://doi.org/10.1155/2019/3851576
Ikehata, H., Okuyama, R., Ogawa, E., Nakamura, S., Usami, A., Mori, T., Tanaka, K., Aiba, S., & Ono, T. (2010). Influences of p53 deficiency on the apoptotic response, DNA damage removal and mutagenesis in UVB-exposed mouse skin. Mutagenesis, 25(4), 397–405. https://doi.org/10.1093/mutage/geq019
Kalluri, R., & Weinberg, R. A. (2009). The basics of epithelial-mesenchymal transition. The Journal of Clinical Investigation, 119(6), 1420–1428. https://doi.org/10.1172/JCI39104
Katiyar, S. K., Mantena, S. K., & Meeran, S. M. (2011). Silymarin protects epidermal keratinocytes from ultraviolet radiation-induced apoptosis and DNA damage by nucleotide excision repair mechanism. PLoS ONE, 6(6), e21410. https://doi.org/10.1371/journal.pone.0021410
Krutmann, J., & Berneburg, M. (2021). Sun-damaged skin (photoaging): What is new? Der Hautarzt, 72(1), 2–5. https://doi.org/10.1007/s00105-020-04747-4
Kuhn, A., Wenzel, J., & Weyd, H. (2014). Photosensitivity, apoptosis, and cytokines in the pathogenesis of lupus erythematosus: A critical review. Clin Rev Alle Immunol, 47(2), 148–162. https://doi.org/10.1007/s12016-013-8403-x
Kulms, D., Poppelmann, B., Yarosh, D., Luger, T. A., Krutmann, J., & Schwarz, T. (1999). Nuclear and cell membrane effects contribute independently to the induction of apoptosis in human cells exposed to UVB radiation. Proc Natl Acad Sci U S A, 96(14), 7974–7979. https://doi.org/10.1073/pnas.96.14.7974
Kulms, D., Zeise, E., Poppelmann, B., & Schwarz, T. (2002). DNA damage, death receptor activation and reactive oxygen species contribute to ultraviolet radiation-induced apoptosis in an essential and independent way. Oncogene, 21(38), 5844–5851. https://doi.org/10.1038/sj.onc.1205743
Kyoo-Ri Kwon, M., Alam, B., Park, J.-H., Kim, T.-H., & Lee, S.-H. (2019). Attenuation of UVB-induced photo-aging by polyphenolic-rich Spatholobus Suberectus stem extract via modulation of MAPK/AP-1/MMPs signaling in human keratinocytes. Nutrients, 11(6), 1341. https://doi.org/10.3390/nu11061341
Lee, J. H., Khor, T. O., Shu, L., Su, Z. Y., Fuentes, F., & Kong, A. N. (2013). Dietary phytochemicals and cancer prevention: Nrf2 signaling, epigenetics, and cell death mechanisms in blocking cancer initiation and progression. Pharmacol Therapeut, 137(2), 153–171. https://doi.org/10.1016/j.pharmthera.2012.09.008
Li, H., Gao, A., Jiang, N., Liu, Q., Liang, B., Li, R., Zhang, E., Li, Z., & Zhu, H. (2016). Protective effect of curcumin against acute ultraviolet B irradiation-induced photo-damage. Photochemistry and Photobiology, 92(6), 808–815. https://doi.org/10.1111/php.12628
Li, X. Z., Zhou, C., Zhang, C., Xie, X. X., Zhou, Z. M., Zhou, M. J., Chen, L. H., & Ding, Z. H. (2019). MicroRNA-664 functions as an oncogene in cutaneous squamous cell 1 carcinomas (cSCC) via suppressing interferon regulatory factor 2. Journal of Dermatological Science, 94(3), 330–338. https://doi.org/10.1016/j.jdermsci.2019.05.004
Li, X., Zhang, C., Yuan, Y., Wang, Y., Lu, S., Zhou, Z., Zhen, P., & Zhou, M. (2021). Downregulation of ARMC8 promotes tumorigenesis through activating Wnt/beta-catenin pathway and EMT in cutaneous squamous cell carcinomas. Journal of Dermatological Science, 102(3), 184–192. https://doi.org/10.1016/j.jdermsci.2021.05.002
Liu, A., Yu, C., Qiu, C., Wu, Q., Huang, C., Li, X., She, X., Wan, K., Liu, L., Li, M., Wang, Z., Chen, Y., Hu, F., Song, D., Li, K., Zhao, C., Deng, H., Sun, X., Xu, F., … Wang, G. (2023). PRMT5 methylating SMAD4 activates TGF-β signaling and promotes colorectal cancer metastasis. Oncogene, 42(19), 1572–1584. https://doi.org/10.1038/s41388-023-02674-x
Liu, N., Yu, C., Shi, Y., Jiang, J., & Liu, Y. (2015). SMAD4 expression in breast ductal carcinoma correlates with prognosis. Oncology Letters, 10(3), 1709–1715. https://doi.org/10.3892/ol.2015.3442
Lopes, D. M., & McMahon, S. B. (2016). Ultraviolet radiation on the skin: a painful experience? CNS Neurosci Therapeut, 22(2), 118–126. https://doi.org/10.1111/cns.12444
Lucas, R. M., Norval, M., Neale, R. E., Young, A. R., de Gruijl, F. R., Takizawa, Y., & van der Leun, J. C. (2015). The consequences for human health of stratospheric ozone depletion in association with other environmental factors. Photochemical & Photobiological Sciences, 14(1), 53–87. https://doi.org/10.1039/c4pp90033b
Marais, T. L. D., Kluz, T., Xu, D., Zhang, X., Gesumaria, L., Matsui, M. S., Costa, M., & Sun, H. (2017). Transcription factors and stress response gene alterations in human keratinocytes following solar simulated ultra violet radiation. Science and Reports, 7(1), 13622. https://doi.org/10.1038/s41598-017-13765-7
Massague, J., Blain, S. W., & Lo, R. S. (2000). TGFbeta signaling in growth control, cancer, and heritable disorders. Cell, 103(2), 295–309. https://doi.org/10.1016/s0092-8674(00)00121-5
McCarthy, A. J., & Chetty, R. (2018). Smad4/DPC4. Journal of Clinical Pathology, 71(8), 661–664. https://doi.org/10.1136/jclinpath-2018-205095
Miyaki, M., Iijima, T., Konishi, M., Sakai, K., Ishii, A., Yasuno, M., Hishima, T., Koike, M., Shitara, N., Iwama, T., Utsunomiya, J., Kuroki, T., & Mori, T. (1999). Higher frequency of Smad4 gene mutation in human colorectal cancer with distant metastasis. Oncogene, 18(20), 3098–3103. https://doi.org/10.1038/sj.onc.1202642
Narayanan, D. L., Saladi, R. N., & Fox, J. L. (2010). Ultraviolet radiation and skin cancer. International Journal of Dermatology, 49(9), 978–986. https://doi.org/10.1111/j.1365-4632.2010.04474.x
Richa, & SinhaHäder, R. P. D. P. (2015). Physiological aspects of UV-excitation of DNA. Topics in Current Chemistry, 356, 203–248. https://doi.org/10.1007/128_2014_531
Roland, C. L., Starker, L. F., Kang, Y., Chatterjee, D., Estrella, J., Rashid, A., Katz, M. H., Aloia, T. A., Lee, J. E., Dasari, A., Yao, J. C., & Fleming, J. B. (2017). Loss of DPC4/SMAD4 expression in primary gastrointestinal neuroendocrine tumors is associated with cancer-related death after resection. Surgery, 161(3), 753–759. https://doi.org/10.1016/j.surg.2016.09.002
Schuch, A. P., Moreno, N. C., Schuch, N. J., Menck, C. F. M., & Garcia, C. C. M. (2017). Sunlight damage to cellular DNA: Focus on oxidatively generated lesions. Free Rad Biol Med, 107, 110–124. https://doi.org/10.1016/j.freeradbiomed.2017.01.029
Schwarz, T. (2005). Mechanisms of UV-induced immunosuppression. Keio Journal of Medicine, 54(4), 165–171. https://doi.org/10.2302/kjm.54.165
Sjerobabski Masnec, I., & Poduje, S. (2008). Photoaging. Collegium Antropologicum, 32(Suppl 2), 177–180.
Soehnge, H., Ouhtit, A., & Ananthaswamy, O. N. (1997). Mechanisms of induction of skin cancer by UV radiation. Frontiers in Bioscience, 2, d538–d551. https://doi.org/10.2741/a211
Song, K., Lee, H. S., Jia, L., Chelakkot, C., Rajasekaran, N., & Shin, Y. K. (2022). SMAD4 controls cancer cell metabolism by regulating methylmalonic aciduria cobalamin deficiency (cbl) B Type. Molecular Cell, 45(6), 413–424. https://doi.org/10.14348/molcells.2022.0067
Tachibana, I., Imoto, M., Adjei, P. N., Gores, G. J., Subramaniam, M., Spelsberg, T. C., & Urrutia, R. (1997). Overexpression of the TGFbeta-regulated zinc finger encoding gene, TIEG, induces apoptosis in pancreatic epithelial cells. The Journal of Clinical Investigation, 99(10), 2365–2374. https://doi.org/10.1172/JCI119418
Ullah, I., Liao, Y., Wan, R., Tang, L., & Feng, J. (2018). Alternative Splicing of SMAD4 and Its Function in HaCaT Cells in Response to UVB Irradiation. Journal of Cancer, 9(17), 3177–3186. https://doi.org/10.7150/jca.24756
Wan, R., Feng, J., & Tang, L. (2021). Consequences of mutations and abnormal expression of SMAD4 in tumors and T cells. Oncotarg Ther, 14, 2531–2540. https://doi.org/10.2147/OTT.S297855
Xia, X., Wu, W., Huang, C., Cen, G., Jiang, T., Cao, J., Huang, K., & Qiu, Z. (2015). SMAD4 and its role in pancreatic cancer. Tum Biol, 36(1), 111–119. https://doi.org/10.1007/s13277-014-2883-z
Yang, H., Li, G., Wu, J. J., Wang, L., Uhler, M., & Simeone, D. M. (2013). Protein kinase A modulates transforming growth factor-beta signaling through a direct interaction with Smad4 protein. Journal of Biological Chemistry, 288(12), 8737–8749. https://doi.org/10.1074/jbc.M113.455675
Yoshiki, R., Nakamura, M., & Tokura, Y. (2012). The biological role of UVB-induced cutaneous immunosuppression. Journal of UOEH, 34(1), 77–83. https://doi.org/10.7888/juoeh.34.77
Yoshitomi, Y., Osada, H., Satake, H., Kojima, M., Saito-Takatsuji, H., Ikeda, T., Yoshitake, Y., Ishigaki, Y., Kubo, E., Sasaki, H., & Yonekura, H. (2019). Ultraviolet B-induced Otx2 expression in lens epithelial cells promotes epithelial–mesenchymal transition. Biol Open. https://doi.org/10.1242/bio.035691
Zhang, C., Xie, X., Yuan, Y., Wang, Y., Zhou, M., Li, X., & Zhen, P. (2020). MiR-664 Protects Against UVB Radiation-Induced HaCaT Cell Damage via Downregulating ARMC8. Dose Response, 18(2), 1559325820929234. https://doi.org/10.1177/1559325820929234
Zhao, M., Mishra, L., & Deng, C. X. (2018). The role of TGF-beta/SMAD4 signaling in cancer. International Journal of Biological Sciences, 14(2), 111–123. https://doi.org/10.7150/ijbs.23230
Funding
This work was supported by the National Natural Science Foundation of China (31971167 and 82273582), the Science and Technology Program of Traditional Chinese Medicine of Zhejiang Province(2024ZL1261), the Science and Technology Plan Projects of Liuzhou (2022CAC0229), the Self-Funded Scientific Research Project of Guangxi Health and Family Planning Commission (Z20211580) and College Students' Innovative Entrepreneurial Training Plan Program (202110594024).
Author information
Authors and Affiliations
Contributions
Meijuan Zhou: Conceptualization, Methodology, Writing-original draft. Xiangzhi Li and Yimeng Wang: Conceptualization, Methodology, Writing-original draft and Visualization. Xian Wang, Qingquan He and Cailian Wu: Conceptualization, Methodology, Writing-initial draft. Yi Shen, Yawen Yuan and Shuyi Mao: Methodology, Software, Investigation.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there are no conflicts of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, X., Wang, Y., Wang, X. et al. Downregulation of SMAD4 protects HaCaT cells against UVB-induced damage and oxidative stress through the activation of EMT. Photochem Photobiol Sci (2024). https://doi.org/10.1007/s43630-024-00574-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43630-024-00574-x