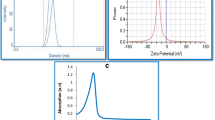
Abstract
Gold nanoparticles functionalized with folic acid toward the internalization into cancer cells have received considerable attention recently. Folic acid is recognized by folate receptors, which are overexpressed in several cancer cells; it is limited in normal cells. In this work, pterin-6-carboxylic acid is proposed as an agonist of folic acid since the pterin-6-carboxylic acid structure has a pterin moiety, the same as folic acid that is recognized by the folate receptor. Here a simple photochemical synthesis of gold nanoparticles functionalized with pterin-6-carboxylic acid is studied. These conjugates were used to cause photothermal damage of HeLa cells irradiating with a diode laser of 808 nm. Pterin-6-carboxylic acid-conjugated gold nanoparticles caused the death of the cell after near-infrared irradiation, dose-dependently. These results indicate a possible internalization of AuNPs via folate receptor-mediated endocytosis due to the recognition or interaction between the folate receptors of HeLa cells and pterin, P6CA.
Graphical abstract












Similar content being viewed by others
References
Huang, W. C., & Chen, Y. C. (2008). Photochemical synthesis of polygonal gold nanoparticles. Journal of Nanoparticle Research, 10(4), 697–702. https://doi.org/10.1007/s11051-007-9293-8
Dong, S., Tang, C., Zhou, H., & Zhao, H. (2004). Photochemical synthesis of gold nanoparticles by the sunlight radiation using a seeding approach. Gold Bulletin, 37(3–4), 187–195. https://doi.org/10.1007/BF03215212
Ahmed, M., & Narain, R. (2010). Rapid synthesis of gold nanorods using a one-step photochemical strategy. Langmuir, 26(23), 18392–18399. https://doi.org/10.1021/la103339g
Rodríguez-Torres, M. P., Díaz-Torres, L. A., & Romero-Servin, S. (2014). Heparin assisted photochemical synthesis of gold nanoparticles and their performance as SERS substrates. International Journal of Molecular Sciences, 15(10), 19239–19252. https://doi.org/10.3390/ijms151019239
Lee, K., Lee, H., Bae, K. H., & Park, T. G. (2010). Heparin immobilized gold nanoparticles for targeted detection and apoptotic death of metastatic cancer cells. Biomaterials, 31(25), 6530–6536. https://doi.org/10.1016/j.biomaterials.2010.04.046
Sega, E. I., & Low, P. S. (2008). Tumor detection using folate receptor-targeted imaging agents. Cancer and Metastasis Reviews, 27(4), 655–664. https://doi.org/10.1007/s10555-008-9155-6
Duan, L., & Zhao, Y. (2018). Selective binding of folic acid and derivatives by imprinted nanoparticle receptors in water. Bioconjugate Chemistry, 29(4), 1438–1445. https://doi.org/10.1021/acs.bioconjchem.8b00121
Boca-Farcau, S., Potara, M., Simon, T., Juhem, A., Baldeck, P., & Astilean, S. (2014). Folic acid-conjugated, SERS-labeled silver nanotriangles for multimodal detection and targeted photothermal treatment on human ovarian cancer cells. Molecular Pharmaceutics, 11(2), 391–399. https://doi.org/10.1021/mp400300m Epub 2013 Dec 12 PMID: 24304361.
Siwowska, K., Schmid, R. M., Cohrs, S., Schibli, R., & Müller, C. (2017). Folate receptor-positive gynecological cancer cells: In vitro and in vivo characterization. Pharmaceuticals, 10(3), 72. https://doi.org/10.3390/ph10030072
Wang, L., Wu, C., Qiao, L., Yu, W., Guo, Q., Zhao, M., Yang, G., Zhao, H., & Lou, J. (2017). Clinical significance of folate receptor-positive circulating tumor cells detected by ligand-targeted polymerase chain reaction in lung cancer. Journal of Cancer, 8(1), 104–110. https://doi.org/10.7150/jca.16856
Chen, Y., Cao, W., Zhou, J., Pidhatika, B., Xiong, B., Huang, L., Tian, Q., Shu, Y., Wen, W., Hsing, I., & Wu, H. (2015). Poly(l -lysine)- graft -folic acid-coupled poly(2-methyl-2-oxazoline) (PLL- g -PMOXA- c -FA): A bioactive copolymer for specific targeting to folate receptor-positive cancer cells. ACS Applied Materials and Interfaces, 7(4), 2919–2930. https://doi.org/10.1021/am508399w
Liu, Q., Xu, S., Niu, C., Li, M., He, D., Lu, Z., Ma, L., Na, N., Huang, F., Jiang, H., & Ouyang, J. (2015). Distinguish cancer cells based on targeting turn-on fluorescence imaging by folate functionalized green emitting carbon dots. Biosensors and Bioelectronics, 64, 119–125. https://doi.org/10.1016/j.bios.2014.08.052
Qin, X. C., Guo, Z. Y., Liu, Z. M., Zhang, W., Wan, M. M., & Yang, B. W. (2013). Folic acid-conjugated graphene oxide for cancer targeted chemo-photothermal therapy. Journal of Photochemistry and Photobiology, B: Biology, 120, 156–162. https://doi.org/10.1016/j.jphotobiol.2012.12.005
Xiao, Z., Ji, C., Shi, J., Pridgen, E. M., Frieder, J., Wu, J., & Farokhzad, O. C. (2012). DNA self-assembly of targeted near-infrared-responsive gold nanoparticles for cancer thermo-chemotherapy. Angewandte Chemie, 51(47), 11853–11857. https://doi.org/10.1002/anie.201204018
Bertel, L., Méndez, S. C., & Martínez, F. (2018). Use in vitro of gold nanoparticles functionalized with folic acid as a photothermal agent on treatment of HeLa cells. Journal Mex Chemical Society, 62(1), 1–13. https://doi.org/10.29356/jmcs.v62i1.385
Bellotti, E., Cascone, M. G., Barbani, N., Rossin, D., Rastaldo, R., Giachino, C., & Cristallini, C. (2021). Targeting cancer cells overexpressing folate receptors with new terpolymer-based nanocapsules: Toward a novel targeted DNA delivery system for cancer therapy. Biomedicines, 9(9), 1275. https://doi.org/10.3390/biomedicines9091275
Tsai, S.-W., Liaw, J.-W., Hsu, F.-Y., Chen, Y.-Y., Lyu, M.-J., & Yeh, M.-H. (2008). Surface-modified gold nanoparticles with folic acid as optical probes for cellular imaging. Sensors, 8(10), 6660–6673. https://doi.org/10.3390/s8106660
Li, G., Li, D., Zhang, L., Zhai, J., & Wang, E. (2009). One-step synthesis of folic acid protected gold nanoparticles and their receptor-mediated intracellular uptake. Chemistry A European Journal, 15(38), 9868–9873. https://doi.org/10.1002/chem.200900914
Zhang, Z., Jia, J., Lai, Y., Ma, Y., Weng, J., & Sun, L. (2010). Conjugating folic acid to gold nanoparticles through glutathione for targeting and detecting cancer cells. Bioorganic & Medicinal Chemistry, 18(15), 5528–5534. https://doi.org/10.1016/j.bmc.2010.06.045
Zhang, H., Chen, H. J., Du, X., & Wen, D. (2014). Photothermal conversion characteristics of gold nanoparticle dispersions. Solar Energy, 100, 141–147. https://doi.org/10.1016/j.solener.2013.12.004
Mendes, R., Pedrosa, P., Lima, J. C., Fernandes, A. R., & Baptista, P. V. (2017). Photothermal enhancement of chemotherapy in breast cancer by visible irradiation of Gold Nanoparticles. Scientific Reports, 7(1), 10872. https://doi.org/10.1038/s41598-017-11491-8
Tong, L., Zhao, Y., Huff, T. B., Hansen, M. N., Wei, A., & Cheng, J.-X. (2007). Gold nanorods mediate tumor cell death by compromising membrane integrity. Advanced Materials, 19, 3136–3141. https://doi.org/10.1002/adma.200701974
Chen, C., Ke, J., Edward Zhou, X., Yi, W., Brunzelle, J. S., Li, J., Yong, E. L., Xu, H. E., & Melcher, K. (2013). Structural basis for molecular recognition of folic acid by folate receptors. Nature, 500(7463), 486–489. https://doi.org/10.1038/nature12327
Blach, D., Alves De Souza, C. E., Méndez, S. C., & Martínez, F. O. (2021). Conjugated anisotropic gold nanoparticles through pterin derivatives for a selective plasmonic photothermal therapy: In vitro studies in HeLa and normal human endocervical cells. Gold Bulletin, 54, 9–23. https://doi.org/10.1007/s13404-020-00288-9
Castillo, J., Bertel, L., Páez-Mozo, E., & Martínez, F. (2013). Photochemical synthesis of the bioconjugate folic acid-gold nanoparticles. Nanomaterials and Nanotechnology, 3, 1–6. https://doi.org/10.5772/57144
Mosmann, T. (1983). Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. Journal of immunological methods, 65(1–2), 55–63. https://doi.org/10.1016/0022-1759(83)90303-4G
Nealon, G. L., Donnio, B., Greget, R., Kappler, J. P., Terazzi, E., & Gallani, J. L. (2012). Magnetism in gold nanoparticles. Nanoscale, 4(17), 5244–5258. https://doi.org/10.1039/c2nr30640a
Eaton, P., Quaresma, P., Soares, C., Neves, C., de Almeida, M. P., Pereira, E., & West, P. (2017). A direct comparison of experimental methods to measure dimensions of synthetic nanoparticles. Ultramicroscopy, 182, 179–190. https://doi.org/10.1016/j.ultramic.2017.07.001
Kowalczyk, B., Lagzi, I., & Grzybowski, B. A. (2011). Nanoseparations: Strategies for size and/or shape-selective purification of nanoparticles. Current Opinion in Colloid and Interface Science, 16(2), 135–148. https://doi.org/10.1016/j.cocis.2011.01.004
Qin, Z., Wang, Y., Randrianalisoa, J., Raeesi, V., Chan, W. C. W., Lipiński, W., & Bischof, J. C. (2016). Quantitative comparison of photothermal heat generation between gold nanospheres and nanorods. Science and Reports, 6, 29836. https://doi.org/10.1038/srep29836
Wangoo, N., Bhasin, K. K., Mehta, S. K., & Suri, C. R. (2008). Synthesis and capping of water-dispersed gold nanoparticles by an amino acid: Bioconjugation and binding studies. Journal of Colloid and Interface Science, 323(2), 247–254. https://doi.org/10.1016/j.jcis.2008.04.043
Kang, K. A., Wang, J., Jasinski, J. B., & Achilefu, S. (2011). Fluorescence manipulation by gold nanoparticles: From complete quenching to extensive enhancement. Journal of Nanobiotechnology, 9, 1–13. https://doi.org/10.1186/1477-3155-9-16
Chhabra, R., Sharma, J., Wang, H., Zou, S., Lin, S., Yan, H., Lindsay, S., & Liu, Y. (2009). Distance-dependent interactions between gold nanoparticles and fluorescent molecules with DNA as tunable spacers. Nanotechnology, 20, 48. https://doi.org/10.1088/0957-4484/20/48/485201
Avila, R., Tamariz, E., Medina-Villalobos, N., Andilla, J., Marsal, M., & Loza-Alvarez, P. (2018). Effects of near infrared focused laser on the fluorescence of labelled cell membrane. Science and Reports, 8(1), 17674. https://doi.org/10.1038/s41598-018-36010-1
Vines, J. B., Yoon, J. H., Ryu, N. E., Lim, D. J., & Park, H. (2019). Gold nanoparticles for photothermal cancer therapy. Frontiers in Chemistry, 7, 1–16. https://doi.org/10.3389/fchem.2019.00167
Chithrani, B. D., Ghazani, A. A., & Chan, W. C. (2006). Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Letters, 6(4), 662–668. https://doi.org/10.1021/nl052396o
Panzarini, E., Mariano, S., Carata, E., Mura, F., Rossi, M., & Dini, L. (2018). Intracellular transport of silver and gold nanoparticles and biological responses: An update. International Journal of Molecular Sciences, 19, 5. https://doi.org/10.3390/ijms19051305
Albanese, A., & Chan, W. (2011). Effect of gold nanoparticle aggregation on cell uptake and toxicity. ACS Nano, 5, 5478–5489. https://doi.org/10.1021/nn2007496
Nakamura, H., & Watano, S. (2018). Direct permeation of nanoparticles across cell membrane: A review. KONA Powder and Particle Journal, 2018(35), 49–65. https://doi.org/10.14356/kona.2018011
Acknowledgements
The authors would like to acknowledge the financial support for this study by the Universidad Industrial de Santander-UIS (Project DIEF 2320) and the teams of Laboratorio de Microscopia for SEM images and Laboratorio de espectroscopia Atómica y Molecular for ATR-FTIR spectra (Parque Tecnológico Guatiguará of Universidad Industrial de Santander, UIS).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Rights and permissions
About this article
Cite this article
Bertel, L., Mendez-Sanchez, S.C. & Martínez-Ortega, F. Laser photo-thermal therapy of epithelial carcinoma using pterin-6-carboxylic acid conjugated gold nanoparticles. Photochem Photobiol Sci 20, 1599–1609 (2021). https://doi.org/10.1007/s43630-021-00122-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-021-00122-x