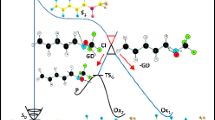
Abstract
The photochemistry of aryl chlorides having a X-SiMe3 group (X = O, NR, S, SiMe2) tethered to the aromatic ring has been investigated in detail, with the aim to generate valuable ϭ,π-heterodiradicals. Two competitive pathways arising from the excited triplet state of the aromatics have been observed, namely heterolysis of the aryl–chlorine bond and homolysis of the X–silicon bond. The former path is found in chlorinated phenols and anilines, whereas the latter is exclusive in the case of silylated thiophenols and aryl silanes. A combined experimental/computational approach was pursued to explain such a photochemical behavior.
Graphical abstract











Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Fensterbank, L., & Ollivier, C. (Eds.). (2021). Free radicals: Fundamentals and applications in organic synthesis. Georg Thieme Verlag KG.
Chatgilialoglu, C., & Studer, A. (Eds.). (2014). Encyclopedia of radicals in chemistry. Biology and Materials Wiley. For seminal books in the field.
Abe, M. (2013). Diradicals. Chemical Review, 113, 7011–7088. https://doi.org/10.1021/cr400056a
(2012). IUPAC Compendium of Chemical Terminology, release 2.3.2 (p. 168). International Union of Pure and Applied Chemistry (IUPAC).
Stuyver, T., Chen, B., Zeng, T., Geerlings, P., De Proft, F., & Hoffmann, R. (2019). Do diradicals behave like radicals? Chemical Reviews, 119, 11291–11351. https://doi.org/10.1021/acs.chemrev.9b00260
Albini, A., & Fagnoni, M. (2013). Photochemically-generated intermediates in synthesis (pp. 131–167). Wiley.
Ravelli, D., Protti, S., & Fagnoni, M. (2016). Carbon-carbon bond forming reactions via photogenerated intermediates. Chemical Reviews, 116, 9850–9913. https://doi.org/10.1021/acs.chemrev.5b00662
Kärkäs, M. D., Porco, J. A., Jr., & Stephenson, C. R. J. (2016). Photochemical approaches to complex chemotypes: Applications in natural product synthesis. Chemical Reviews, 116, 9683–9747. https://doi.org/10.1021/acs.chemrev.5b00760
Itagaki, N., & Iwabuchi, Y. (2007). Enantio- and diastereocontrolled synthesis of (+)-juvabione employing organocatalytic desymmetrization and photoinduced fragmentation. Chemical Communications, 11, 1175–1176. https://doi.org/10.1039/B616641E
Wessig, P. (2004). Regioselective photochemical synthesis of carbo- and heterocyclic compounds: The Norrish/Yang reaction. In F. Lenci & W. Horspool (Eds.), CRC handbook of organic photochemistry and photobiology (2nd ed., pp. 571–5720). CRC Press.
Wagner, P. J. (2004). Yang photocyclization: Coupling of biradicals formed by intramolecular hydrogen abstraction of ketones. In F. Lenci & W. Horspool (Eds.), CRC handbook of organic photochemistry and photobiology (2nd ed., pp. 58-1-58–70). CRC Press.
Wagner, P. J. (2005). Abstraction of γ-hydrogens by excited carbonyls. In A. G. Griesbeck & J. Mattay (Eds.), Molecular and supramolecular photochemistry. Synthetic organic photochemistry (Vol. 12, pp. 11–40). Marcel Dekker.
Wessig, P., & Mühling, O. (2005). Abstraction of (γ±n)-hydrogens by excited carbonyls. In A. G. Griesbeck & J. Mattay (Eds.), Molecular and supramolecular photochemistry. Synthetic organic photochemistry (Vol. 12, pp. 41–88). Marcel Dekker.
Garcia-Garibay, M. A., & Campos, L. M. (2004). Photochemical decarbonylation of ketones: Recent advances and reactions in crystalline solids. In F. Lenci & W. Horspool (Eds.), CRC handbook of organic photochemistry and photobiology (2nd ed., pp. 48-1-48–41). CRC Press.
Natarajan, A., Ng, D., Yang, Z., & Garcia-Garibay, M. A. (2007). Parallel syntheses of (+)-and (-)-α-cuparenone by radical combination in crystalline solids. Angewandte Chemie International Edition, 46, 6485–6487. https://doi.org/10.1002/anie.200700679
Dotson, J. J., Perez-Estrada, S., & Garcia-Garibay, M. A. (2018). Taming radical pairs in nanocrystalline ketones: Photochemical synthesis of compounds with vicinal stereogenic all-carbon quaternary centers. Journal of the American Chemical Society, 140, 8359–8371. https://doi.org/10.1021/jacs.8b03988
Adam, W., Oppenlander, T., & Zang, G. (1985). Photochemistry of the azoalkanes 2,3-diazabicyclo[2.2.l]hept-2-ene and spiro[cyclopropane-1,70-[2,3]diazabicyclo[2.2.1] hept-2-ene]: On the questions of one-bond vs. two-bond cleavage during the denitrogenation, cyclization vs. rearrangement of the 1,3-diradicals, and double inversion. The Journal of Organic Chemistry, 50, 3303–3312. https://doi.org/10.1021/jo00218a012
Adam, W., Nau, W. M., & Sendelbach, J. (1994). Temperature dependence of the α versus β bond cleavage in the direct and triplet-sensitized photolysis of azoalkanes of the 2,3-diazabicyclo[2.2.1]hept-2-ene type. Journal of the American Chemical Society, 116, 7049–7054. https://doi.org/10.1021/ja00095a006
Adam, W., Garcıa, H., Martì, V., Moorthy, J. N., Peters, K., & Peters, E.-M. (2000). Photochemical denitrogenation of norbornene-annelated 2,3-diazabicyclo[2.1.1]hept-2-ene-type azoalkanes: Crystal-lattice versus zeolite-interior effects. Journal of the American Chemical Society, 122, 3536–3537. https://doi.org/10.1021/ja994114q
Wender, P. A., Kee, J.-M., & Warrington, J. M. (2008). Practical synthesis of prostratin, DPP, and their analogs, adjuvant leads against latent HIV. Science, 320, 649–652. https://www.science.org/doi/10.1126/science.1154690
Ao, M. S., & Burgess, E. M. (1971). Benzothiazete 1, l-dioxides. Journal of the American Chemical Society, 93, 5298–5299. https://doi.org/10.1021/ja00749a078
Magauer, T., Martin, H. J., & Mulzer, J. (2009). Total synthesis of the antibiotic kendomycin by macrocyclization using photo-Fries rearrangement and ring-closing metathesis. Angewandte Chemie International Edition, 48, 6032–6036. https://doi.org/10.1002/anie.200900522
Bhunia, A., Yetra, S. R., & Biju, A. T. (2012). Recent advances in transition-metal-free carbon–carbon and carbon–heteroatom bond-forming reactions using arynes. Chemical Society Reviews, 41, 3140–3152. https://www.pubs.rsc.org/en/content/articlehtml/2012/cs/c2cs15310f
Wentrup, C. (2010). The benzyne story. Australian Journal of Chemistry, 63, 979–986. https://doi.org/10.1071/CH10179
Bracken, C., Batsanov, A. S., & Baumann, M. (2021). Development of a continuous photochemical benzyne-forming process. SynOpen, 5, 29–35. https://doi.org/10.1055/s-0040-1706016
Raviola, C., Protti, S., Ravelli, D., & Fagnoni, M. (2016). (Hetero)aromatics from dienynes, enediynes and enyne-allenes. Chemical Society Reviews, 45, 4364–4390. https://doi.org/10.1039/C6CS00128A
Alabugin, I. V., Yang, W.-Y. & Pal, R. (2012) In A. Griesbeck, M. Oelgemoeller, & F. Ghetti. (Eds.), CRC handbook of organic photochemistry and photobiology (3rd ed., pp. 549−592). CRC Press.
Grissom, J. W., Gunawardena, G. U., Klingberg, D., & Huang, D. (1996). The chemistry of enediynes, enyne allenes and related compounds. Tetrahedron, 52, 6453–6518. https://doi.org/10.1016/0040-4020(96)00016-6
Galm, U., Hager, M. H., Van Lanen, S. G., Ju, J., Thorson, J. S., & Shen, B. (2005). Antitumor antibiotics: Bleomycin, enediynes, and mitomycin. Chemical Reviews, 105, 739–758. https://doi.org/10.1021/cr030117g
Smith, A. L., & Nicolaou, K. C. (1996). The enediyne antibiotics. Journal of Medicinal Chemistry, 39, 2103–2117. https://doi.org/10.1021/jm9600398
Nicolau, K. C., & Smith, A. L. (1992). Molecular design, chemical synthesis, and biological action of enediynes. Accout of Chemical Research, 25, 497–503. https://doi.org/10.1021/ar00023a003
Nagata, R., Yamanaka, H., Okazaki, E., & Saito, I. (1989). Biradical formation from acyclic conjugated eneyne-allene system related to neocarzinostatin and esperamicin-calichemicin. Tetrahedron Letters, 30, 4995–4998. https://doi.org/10.1016/S0040-4039(01)80564-5
Myers, A. G., Kuo, E. Y., & Finney, N. S. (1989). Thermal generation of α,3-dehydrotoluene from (Z)-1,2,4-heptatrien-6-yne. Journal of the American Chemical Society, 111, 8057–8059. https://doi.org/10.1021/ja00202a079
Dichiarante, V., Protti, S., & Fagnoni, M. (2017). Phenyl cation: A versatile intermediate. Journal of Photochemistry & Photobiology A: Chemistry, 339, 103–113. https://doi.org/10.1016/j.jphotochem.2017.02.007
Lazzaroni, S., Ravelli, D., Protti, S., Fagnoni, M., & Albini, A. (2017). Photochemical synthesis: Using light to build C-C bonds under mild conditions. Synthèse photochimique: Utiliser la lumière pour construire des liaisons Carbon-Carbon dans des conditions douces. Comptes Rendue Chimie, 20, 261–271. https://doi.org/10.1016/j.crci.2015.11.024
Protti, S., Ravelli, D., Mannucci, B., Albini, A., & Fagnoni, M. (2012). α, n-didehydrotoluenes by photoactivation of (chlorobenzyl)trimethylsilanes. An alternative to enyne-allenes cyclization. Angewandte Chemie International Edition, 51, 8577–8580. https://doi.org/10.1002/anie.201202794
Ravelli, D., Protti, S., Fagnoni, M., & Albini, A. (2013). From phenyl chlorides to α, n-didehydrotoluenes (α, n-DHTs) via phenyl cations. A CPCM-CASMP2 investigation. The Journal of Organic Chemistry, 78, 3814–3820. https://doi.org/10.1021/jo400269s
Protti, S., Ravelli, D., Fagnoni, M., & Albini, A. (2013). Smooth photogeneration of α, n-didehydrotoluenes (DHTs). Pure & Applied Chemistry, 85, 1479–1486. https://doi.org/10.1351/PAC-CON-12-10-03/html
Pedroli, C., Ravelli, D., Protti, S., Albini, A., & Fagnoni, M. (2017). Singlet vs triplet reactivity of photogenerated α, n-didehydrotoluenes. The Journal of Organic Chemistry, 82, 6592–6603. https://doi.org/10.1021/acs.joc.7b00610
Raviola, C., Ravelli, D., Protti, S., & Fagnoni, M. (2014). Methoxy-substituted α, n-didehydrotoluenes. Photochemical generation and polar vs diradical reactivity. Journal of the American Chemical Society, 136, 13874–13881. https://doi.org/10.1021/ja507735u
Ravelli, D., Protti, S., & Fagnoni, M. (2015). Photogenerated α, n-didehydrotoluenes from chlorophenylacetic acids at physiological pH. The Journal of Organic Chemistry, 80, 852–858. https://doi.org/10.1021/jo502318v
Crespi, S., Protti, S., Ravelli, D., Merli, D., & Fagnoni, M. (2017). Sugar assisted photogeneration of didehydrotoluenes from chlorobenzylphosphonic acids. The Journal of Organic Chemistry, 82, 12162–12172. https://doi.org/10.1021/acs.joc.7b01963
Crespi, S., Ravelli, D., Protti, S., Albini, A., & Fagnoni, M. (2014). Competing pathways in the photogeneration of didehydrotoluenes from (trimethylsilylmethyl)aryl sulfonates and phosphates. Chemistry A European Journal, 20, 17572–17578. https://doi.org/10.1002/chem.201404787
Nakatani, K., Isoe, S., Maekawa, S., & Saito, I. (1994). Photoinduced DNA cleavage by designed molecules with conjugated Ene-Yne-ketene functionalities. Tetrahedron Letters, 35, 605–608. https://doi.org/10.1016/S0040-4039(00)75850-3
Sullivan, R. W., Coghlan, V. M., Munk, S. A., Reed, M. W., & Moore, H. W. (1994). DNA cleavage by 4-alkynyl-3-methoxy-4-hydroxycyclobutenones. The Journal of Organic Chemistry, 59, 2276–2278. https://doi.org/10.1021/jo00088a002
Xia, H., & Moore, H. W. (1992). Rearrangements of 4-alkynylcyclobutenones. Annelated spiroepoxycyclohexadienones and quinones from 4-(1,5-alkadiynyl)-4-methoxy- or -hydroxycyclobutenones. The Journal of Organic Chemistry, 57, 3765–3766. https://doi.org/10.1021/jo00040a008
Wang, K. K. (1996). Cascade radical cyclizations via biradicals generated from enediynes, enyne-allenes, and enyne-ketenes. Chemical Reviews, 96, 207–222. https://doi.org/10.1021/cr950030y
Musch, P. W., Remenyi, C., Helten, H., & Engels, B. (2002). On the regioselectivity of the cyclization of enyne-ketenes: A computational investigation and comparison with the Myers-Saito and Schmittel reaction. Journal of the American Chemical Society, 124, 1823–1828. https://doi.org/10.1021/ja017532f
Li, H., Yang, H., Petersen, J. L., & Wang, K. K. (2004). Biradicals/zwitterions from thermolysis of enyne-isocyanates. application to the synthesis of 2(1H)-pyridones, benzofuro[3,2-c]pyridin-1(2H)-ones, 2,5-dihydro-1H-pyrido[4,3-b]indol-1-ones, and related compounds. The Journal of Organic Chemistry, 69, 4500–4508. https://doi.org/10.1021/jo049716t
Zhang, Y., Irshaidat, T., Wang, H., Waynant, K. V., Wang, H., & Herndon, J. W. (2008). Coupling of Fischer carbene complexes with conjugated enediynes featuring radical traps: Novel structure and reactivity features of chromium complexed arene diradical species. Journal of Organometallic Chemistry, 693, 3337–3345. https://doi.org/10.1016/j.jorganchem.2008.08.003
Gonçalves, T. P., Mohamed, M., Whitby, R. J., Sneddon, H. F., & Harrowven, D. C. (2015). Exploring diradical chemistry: A carbon-centered radical may act as either an anion or electrophile through an orbital isomer. Angewandte Chemie International Edition, 54, 4531–4534. https://doi.org/10.1002/anie.201411334
Baba, G., Tea, C. G., Touré, S. A., Lesvier, M., & Denis, J.-M. (2002). Tandem eneyne-phosphaallene/Myers type cyclization via base-induced isomerisation of enediynephosphine. Journal of Organometallic Chemistry, 643–644, 342–349. https://doi.org/10.1016/S0022-328X(01)01392-4
Li, H., Petersen, J. L., & Wang, K. K. (2003). Cascade cyclizations via N,4-didehydro-2-(phenylamino)pyridine biradicals/zwitterions generated from enyne-carbodiimides. The Journal of Organic Chemistry, 68, 5512–5518. https://doi.org/10.1021/jo020760n
Shi, C., Zhang, Q., & Wang, K. K. (1999). Biradicals from thermolysis of N-[2-(1-alkynyl)phenyl]-N¢-phenylcarbodiimides and their subsequent transformations to 6H-Indolo[2,3-b]quinolines. The Journal of Organic Chemistry, 64, 925–932. https://doi.org/10.1021/jo981845k
Zhang, Q., Shi, C., Zhang, H.-R., & Wang, K. K. (2000). Synthesis of 6H-indolo[2,3-b][1,6]naphthyridines and related compounds as the 5-aza analogues of ellipticine alkaloids. The Journal of Organic Chemistry, 65, 7977–7983. https://doi.org/10.1021/jo000978e
Lu, X., Petersen, J. L., & Wang, K. K. (2002). Synthesis of novel heteroaromatics structurally related to ellipticine alkaloids via thermolysis of pyridannulated enyne-carbodiimides. The Journal of Organic Chemistry, 67, 5412–5415. https://doi.org/10.1021/jo0202031
Schmittel, M., Steffen, J.-P., Engels, B., Lennartz, C., & Hanrath, M. (1998). Two novel thermal biradical cyclizations in theory and experiment: new synthetic routes to 6h-indolo[2,3-b]quinolines and 2-aminoquinolines from enyne-carbodiimides. Angewandte Chemie International Edition, 37, 2371–2373. https://doi.org/10.1002/(SICI)1521-3773(19980918)37:17%3c2371::AID-ANIE2371%3e3.0.CO;2-N
Grabner, G., Richard, C., & Köhler, G. (1994). Formation and reactivity of 4-oxocyclohexa-2,5-dienylidene in the photolysis of 4-chlorophenol in aqueous solution at ambient temperature. Journal of the American Chemical Society, 116, 11470–11480. https://doi.org/10.1021/ja00104a029
Durand, A. P., Brown, R. G., Worrall, D., & Wilkinson, F. (1998). Study of the aqueous photochemistry of 4-fluorophenol, 4-bromophenol and 4-iodophenol by steady state and nanosecond laser flash photolysis. Journal of Chemical Society Perkin Transaction, 2, 365–370. https://doi.org/10.1039/A705287A
Bonnichon, F., Grabner, G., Guyot, G., & Richard, C. (1999). Photochemistry of substituted 4-halogenophenols: Effect of a CN substituent. Journal of Chemical Society Perkin Transaction, 2, 1203–1210. https://doi.org/10.1039/A900141G
Protti, S., Fagnoni, M., Mella, M., & Albini, A. (2004). Aryl cations from aromatic halides. photogeneration and reactivity of 4-hydroxy(methoxy)phenyl cation. The Journal of Organic Chemistry, 69, 3465–3473. https://doi.org/10.1021/jo049770%2B
Sander, W., Müller, W., & Sustmann, R. (1988). 4-Oxo-2,5-cyclohexadienylidene—A carbene with a stable triplet and metastable singlet state? Angewandte Chemie International Edition, 27, 572–574. https://doi.org/10.1002/anie.198805721
Sander, W., Kötting, C., & Hübert, R. (2000). Super-electrophilic carbenes and the concept of Philicity. Journal of Physical Organic Chemistry, 13, 561–568. https://doi.org/10.1002/1099-1395(200010)13:10%3c561::AID-POC239%3e3.0.CO;2-U
Sander, W., Hubert, R., Kraka, E., Grafenstein, J., & Cremer, D. (2000). 4-Oxo-2,3,5,6-tetrafluorocyclohexa-2,5-dienylidene—A highly electrophilic triplet carbene. Chemistry A European Journal, 6, 4567–4579. https://doi.org/10.1002/1521-3765(20001215)6:24%3c4567::AID-CHEM4567%3e3.0.CO;2-A
Brennan, J., Cadogan, J. I. G., & Sharp, J. T. (1976). Novel Reactions of diazonium salts: direct conversion of o- and p-chlorobenzenediazonium acetates into hydroxybenzenediazonium salts, of o-acetoxybenzenediazonium acetate into the carbene, 2-oxocyc1ohexa-3,5-dienylidene, and of o-thioacetoxybenzenediazonium acetate into 1,2,3-benzothiadiazole. Journal of Chemical Society Chemical Communications. https://doi.org/10.1039/C39760000850
Brennan, J., Cadogan, J. I. G., & Sharp, J. T. (1977). Acylarylnitrosamines. Part 13 promotion of ionic reactions of o- and p-chioro-, o- and p-acetoxy -, and o-acetylthio-N-nitrosoacetanilides. Formation of hydroxybenzenediazonium salts, the carbene 2-oxocyclohexa-3,5-dienylidene, and 1,2,3- benzothiadiazole. Journal of Chemical Society Perkin Transaction, 1, 1844–1848. https://doi.org/10.1039/P19770001844
Burdzinski, G., Kubicki, J., Sliwa, M., Rėhault, J., Zhang, Y., Vyas, S., Luk, H. L., Hadad, C. M., & Platz, M. S. (2013). Mechanistic aspects of ketene formation deduced from femtosecond photolysis of diazocyclohexadienone, o-phenylene thioxocarbonate, and 2-chlorophenol. The Journal of Organic Chemistry, 78, 2026–2032. https://doi.org/10.1021/jo302023a
Canevari, V., Fagnoni, M., Bortolus, P., & Albini, A. (2011). Environmental implications of the surfactants effect on the photochemistry of (substituted) 4-chlorophenols in water. Chemsuschem, 4, 98–103. https://doi.org/10.1002/cssc.201000277
Othmen, K., Boule, P., & Richard, C. (1999). Mechanism of 3-halogenoaniline photolysis in methanol. New Journal of Chemistry, 23, 857–861. https://doi.org/10.1039/A902609F
Othmen, K., Boule, P., Szczepanik, B., Rotkiewicz, K., & Grabner, G. (2000). Photochemistry of 4-chloroaniline in solution. Formation and kinetic properties of a new carbene, 4-iminocyclohexa-2,5-dienylidene. Journal of Physical Chemistry A, 104, 9525–9534. https://doi.org/10.1021/jp0010381
Chan, W. S., Leung, K. H., Ong, S. Y., & Phillips, D. L. (2002). Transient resonance raman investigation of 4-iminocyclohexa-2,5-dienylidene and 4-oxocyclohexa-2,5-dienylidene carbenes in aqueous solution. Journal of Physical Chemistry A, 106, 6254–6261. https://doi.org/10.1021/jp014254y
Fagnoni, M., Mella, M., & Albini, A. (1999). Smooth synthesis of aryl- and alkylanilines by photoheterolysis of haloanilines in the presence of aromatics and alkenes. Organic Letters, 1, 1299–1301. https://doi.org/10.1021/ol990982g
Guizzardi, B., Mella, M., Fagnoni, M., Freccero, M., & Albini, A. (2001). Generation and reactivity of the 4-aminophenyl cation by photolysis of 4-chloroaniline. The Journal of Organic Chemistry, 66, 6353–6363. https://doi.org/10.1021/jo0104680
Weber, W. P. (1983). Silicon reagents for organic synthesis. Springer-Verlag.
Lazzaroni, S., Dondi, D., Fagnoni, M., & Albini, A. (2007). Photochemical arylation reactions by 4-chlorothioanisole. European Journal of Organic Chemistry. https://doi.org/10.1002/ejoc.200700290
Frisch, M. J., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., Scalmani, G., Barone, V., Petersson, G. A., Nakatsuji, H., Li, X., Caricato, M., Marenich, A. V., Bloino, J., Janesko, B. G., Gomperts, R., Mennucci, B., Hratchian, H. P., Ortiz, J. V., … Fox, D. J. (2016). Gaussian 16, Revision B.01. Gaussian Inc.
Goerigk, L., Hansen, A., Bauer, C., Ehrlich, S., Najibi, A., & Grimme, S. (2017). A look at the density functional theory zoo with the advanced GMTKN55 database for general main group thermochemistry, kinetics and noncovalent interactions. Physical Chemistry Chemical Physics, 19, 32184–32215. https://doi.org/10.1039/C7CP04913G
Steinmetz, M., Hansen, A., Ehrlich, S., Risthaus, T., & Grimme, S. (2014). Accurate thermochemistry for large molecules with modern density functionals. Topics in Current Chemistry, 365, 1–23. https://doi.org/10.1007/128_2014_543
Song, H., Freixas, V. M., Fernandez-Alberti, S., White, A. J., Zhang, Y., Mukamel, S., Govind, N., & Tretiak, S. (2021). An ab initio multiple cloning method for non-adiabatic excited-state molecular dynamics in NWChem. Journal of Chemical Theory and Computation, 17, 3629–3643. https://doi.org/10.1021/acs.jctc.1c00131
Barone, V., Ceselin, G., Fusè, M., & Tasinato, N. (2020). Accuracy meets interpretability for computational spectroscopy by means of hybrid and double-hybrid functionals. Frontiers in Chemistry, 8, 584203. https://doi.org/10.3389/fchem.2020.584203
Zanders, D., Bačic, G., Leckie, D., Odegbesan, O., Rawson, J., Masuda, J. D., Devi, A., & Barry, S. T. (2020). A rare low-spin CoIV Bis(b-silyldiamide) with high thermal stability: steric enforcement of a doublet configuration. Angewandte Chemie International Edition, 59, 14138–14142. https://doi.org/10.1002/anie.202001518
Lazzaroni, S., Dondi, D., Fagnoni, M., & Albini, A. (2010). Selectivity in the reaction of triplet phenyl cations. The Journal of Organic Chemistry, 75, 315–323. https://doi.org/10.1021/jo9017974
Lazzaroni, S., Dondi, D., Fagnoni, M., & Albini, A. (2008). Geometry and energy of substituted phenyl cations. The Journal of Organic Chemistry, 73, 206–211. https://doi.org/10.1021/jo7020218
Baranac-Stojanović, M. (2020). Substituent effect on triplet state aromaticity of benzene. The Journal of Organic Chemistry, 85, 4289–4297. https://doi.org/10.1021/acs.joc.9b03472
Zhang, B., Zhang, J., Yang, D.-D.H., & Yang, N.-C.C. (1996). Photoamidation of N-acetyl-2-chlorotyrosine methyl ester and 3-chlorophenol. The Journal of Organic Chemistry, 61, 3236–3237. https://doi.org/10.1021/jo960037v
Dichiarante, V., Dondi, D., Protti, S., Fagnoni, M., & Albini, A. (2007). A meta effect in organic photochemistry? The case of SN1 reactions in methoxyphenyl derivatives. Journal of the American Chemical Society, 129, 5605–5611. https://doi.org/10.1021/ja068647s
Qrareya, H., Raviola, C., Protti, S., Fagnoni, M., & Albini, A. (2013). Transition-metal-free arylations via photogenerated triplet 4-alkyl-and 4-trimethylsilyl-phenyl cations. The Journal of Organic Chemistry, 78, 6016–6024. https://doi.org/10.1021/jo4007046
Park, S. K., & Kim, H. A. (2011). Photoreactions of (2-acetoxyphenyl)pentamethyldisilane. Bulletin of the Korean Chemical Society, 32, 3155–3157. https://doi.org/10.5012/bkcs.2011.32.8.3155
Ishikawa, M., Fuchikami, T., Sugaya, T., & Kumada, M. (1975). Photolysis of organopolysilanes. Novel addition reaction of aryl substituted disilanes to olefins. Journal of the American Chemical Society, 97, 5923–5924. https://doi.org/10.1021/ja00853a052 For selective examples.
Ishikawa, M., Fuchikami, T., & Kumada, M. (1976). Photochemically generated silicon-carbon double-bonded intermediates: III. The reaction of p-tolylpentamethyldisilane with methanol and methanol-d1. Journal of Organometallic Chemistry, 118, 155–160. https://doi.org/10.1016/S0022-328X(00)92150-8(Forselectiveexamples)
Kira, M., Miyazawa, T., Sugiyama, H., Yamaguchi, M., & Sakurai, H. (1993). σ, π* orthogonal intramolecular charge-transfer (OICT) excited states and photoreaction mechanism of trifluoromethyl-substituted phenyldisilanes. Journal of the American Chemical Society, 115, 3116–3124. https://doi.org/10.1021/ja00061a010 For selective examples.
Park, S. K. (2007). Photoreactions of 4,4’-Bis(pentamethyldisilanyl)biphenyl. Bulletin of the Korean Chemical Society, 28, 1045–1048. https://doi.org/10.5012/bkcs.2007.28.6.1045 For selective examples.
Park, S. K., & Kwon, S. T. (2012). A novel photoreaction of (2-hydroxypropoxyphenyl)pentamethyldisilane. Bulletin of the Korean Chemical Society, 33, 3823–3826. https://doi.org/10.5012/bkcs.2012.33.11.3823 For selective examples.
Glendening, E. D., Badenhoop, J. K., Reed, A. E., Carpenter, J. E., Bohmann, J. A., Morales, C. M. P., Karafiloglou, P., Landis, C. R., & Weinhold, F. (2018). NBO 7.0. Theoretical Chemistry Institute, University of Wisconsin.
Hosoya, Y., Kobayashi, I., Mizoguchi, K., & Nakada, M. (2019). Palladium-catalyzed carbothiolation via trapping of the σ-alkyl palladium intermediate with RSTIPS. Organic Letters, 21, 8280–8284. https://doi.org/10.1021/acs.orglett.9b03046
Azipurua, J. M., & Palomo, C. (1985). 1,8-diazabicyclo[5.4.0]undec-7-ene (dbu): An effective base for the introduction of tbutyldimethylsilyl group in organic compounds. Tetrahedron Letters, 26, 475–476. https://doi.org/10.1016/S0040-4039(00)61915-9
Iwao, M. (1990). Directed lithiation of chlorobenzenes. regioselectivity and application to a short synthesis of benzocyclobutenes. The Journal of Organic Chemistry, 55, 3622–3627. https://doi.org/10.1021/jo00298a045
Gabrielli, L., & Mancin, F. (2016). Minimal self-immolative probe for multimodal fluoride detection. The Journal of Organic Chemistry, 81, 10715–10720. https://doi.org/10.1021/acs.joc.6b01787
Qu, W., Hu, B., Babich, J. W., Waterhouse, N., Dooley, M., Ponnala, S., & Urgiles, J. (2020). A general 11C-labeling approach enabled by fluoride-mediated desilylation of organosilanes. Nature Communications, 11, 1736. https://doi.org/10.1038/s41467-020-15556-7
Marzi, E., & Schlosser, M. (2005). The site-selective functionalization of halogen-bearing phenols: An exercise in diversity-oriented organometallic synthesis. Tetrahedron, 61, 3393–3401. https://doi.org/10.1016/j.tet.2004.10.103
Wu, J., Wang, Z., Chen, X.-Y., Wu, Y., Wang, D., Peng, Q., & Wang, P. (2020). Para-selective borylation of monosubstituted benzenes using a transient mediator. Science China Chemistry, 3, 336–340. https://doi.org/10.1007/s11426-019-9652-x
Guggenheim, T. L. (1984). Protection of substituted anilines with 1,1,4,4-tetramethyl-1,4-bis(n, n-dimethylamino)disilethylene. Tetrahedron Letters, 25, 1253–1254. https://doi.org/10.1016/S0040-4039(01)80126-X
Wang, H., Tong, X., Huo, Y., Tang, J., & Xia, C. (2020). Potassium methoxide/disilane-mediated formylation of aryl iodides with DMF at room temperature. Organic Chemistry Frontiers, 7, 4074–4079. https://doi.org/10.1039/D0QO00974A
Funding
L. D. T., F. R. S.P. and M.F. thanks the Universitiamo crowfunding project (University of Pavia) for partial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Additional information
Dedicated to Prof. Angelo Albini in the occasion of his 75th birthday.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Di Terlizzi, L., Roncari, F., Crespi, S. et al. Aryl–Cl vs heteroatom–Si bond cleavage on the route to the photochemical generation of σ,π-heterodiradicals. Photochem Photobiol Sci 21, 667–685 (2022). https://doi.org/10.1007/s43630-021-00119-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-021-00119-6