Abstract
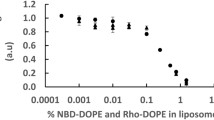
FRET has been massively used to see if biomolecules were bounded or not by labelling both biomolecules by one dye of a FRET pair. This should give a digital answer to the question (fluorescence of the acceptor: high FRET efficency: molecules associated, fluorescence of the donor: low FRET efficency: molecules dissociated). This has been used, inter alia, at the single-molecule scale in containers, such as liposomes. One perspective of the field is to reduce the container’s size to study the effect of confinement on binding. The problem is that if the two dyes are encapsulated inside a small liposome, they could have a significant probability to be close one from the other one (and thus to undergo a high FRET efficiency event without binding). This is why we suggest here a theoretical model which gives mean FRET efficiency as a function of liposome radius (the model applies to any spherical container) and Förster radius to help the experimentalist to choose their experimental set-up. Besides, the influence of side effect on mean FRET efficiency has been studied as well. We show here that if this “background FRET” is most of the time non-quantitative, it can remain significant and which makes data analysis trickier. We could show as well that if this background FRET obviously increases when liposome radius decreases, this variation was lower than the one which could be expected because of side effect. We show as well the FRET efficiency function distribution which let the experimentalist know the probability to get one FRET efficiency value.




Similar content being viewed by others
References
Li, Z., Hayman, R. B., & Walt, D. R. (2008). Detection of single-molecule DNA hybridization using enzymatic amplification in an array of femtoliter-sized reaction vessels. Journal of the American Chemical Society, 130, 12622–12623.
Zsila, F., Bikadi, Z., Malik, D., Hari, P., Pechan, I., Berces, A., & Hazai, E. (2011). Evaluation of drug–human serum albumin binding interactions with support vector machine aided online automated docking. Bioinformatics, 27, 1806–1813.
Vinogradov, S. V. (2006). Colloidal microgels in drug delivery applications. Current Pharmaceutical Design, 12, 4703–4712.
Paschke, R., Paetz, C., Mueller, T., Schmoll, H.-J., Mueller, H., Sorkau, E., & Sinn, E. (2003). Biomolecules linked to transition metal complexes-new chances for chemotherapy. Current Medicinal Chemistry, 10, 2033–2044.
Ji, T. H., & Benson, A. (1968). Association of lipids and proteins in chloroplast lamellar membrane. Biochimica et Biophysica Acta (BBA)-Biomembranes, 150, 686–693.
Carrero, G., McDonald, D., Crawford, E., de Vries, G., & Hendzel, M. J. (2003). Using FRAP and mathematical modeling to determine the in vivo kinetics of nuclear proteins. Methods, 29, 14–28.
Nosek, L., Semchonok, D., Boekema, E. J., Ilík, P., & Kouřil, R. (2017). Structural variability of plant photosystem II megacomplexes in thylakoid membranes. The Plant Journal, 89, 104–111.
Amunts, A., Drory, O., & Nelson, N. (2007). The structure of a plant photosystem I supercomplex at 3.4 Å resolution. Nature, 447, 58–63.
Acín-Pérez, R., Fernández-Silva, P., Peleato, M. L., Pérez-Martos, A., & Enriquez, J. A. (2008). Respiratory active mitochondrial supercomplexes. Molecular cell, 32, 529–539.
Schägger, H., & Pfeiffer, K. (2000). Supercomplexes in the respiratory chains of yeast and mammalian mitochondria. The EMBO journal, 19, 1777–1783.
Butler, T. Z., Pavlenok, M., Derrington, I. M., Niederweis, M., & Gundlach, J. H. (2008). Single-molecule DNA detection with an engineered MspA protein nanopore. Proceedings of the National Academy of Sciences, 105, 20647–20652.
Junesch, J., Emilsson, G., Xiong, K., Kumar, S., Sannomiya, T., Pace, H., et al. (2015). Location-specific nanoplasmonic sensing of biomolecular binding to lipid membranes with negative curvature. Nanoscale, 7, 15080–15085.
Seidel, M., & Gauglitz, G. (2003). Miniaturization and parallelization of fluorescence immunoassays in nanotiter plates. TrAC Trends in Analytical Chemistry, 22, 385–394.
Steemers, F. J., Ferguson, J. A., & Walt, D. R. (2000). Screening unlabeled DNA targets with randomly ordered fiber-optic gene arrays. Nature biotechnology, 18, 91–94.
Ali, M., Nasir, S., Nguyen, Q. H., Sahoo, J. K., Tahir, M. N., Tremel, W., & Ensinger, W. (2011). Metal ion affinity-based biomolecular recognition and conjugation inside synthetic polymer nanopores modified with iron–terpyridine complexes. Journal of the American Chemical Society, 133, 17307–17314.
Cisse, I., Okumus, B., Joo, C., & Ha, T. (2007). Fueling protein–DNA interactions inside porous nanocontainers. Proceedings of the National Academy of Sciences, 104, 12646–12650.
Jares-Erijman, E. A., & Jovin, T. M. (2003). FRET imaging. Nature biotechnology, 21, 1387–1395.
Jofre, A., Case, J., & Hicks, S. (2010). Single molecule observations of DNA hybridization kinetics. Biophysical Journal, 98, 16a.
Ha, T., Enderle, T., Ogletree, D. F., Chemla, D. S., Selvin, P. R., & Weiss, S. (1996). Probing the interaction between two single molecules: fluorescence resonance energy transfer between a single donor and a single acceptor. Proceedings of the National Academy of Sciences, 93, 6264–6268.
Truong, K., & Ikura, M. (2001). The use of FRET imaging microscopy to detect protein–protein interactions and protein conformational changes in vivo. Current Opinion in Structural Biology, 11, 573–578.
Rhoades, E., Gussakovsky, E., & Haran, G. (2003). Watching proteins fold one molecule at a time. Proceedings of the National Academy of Sciences of the United States of America, 100, 3197–3202.
Lee, J. Y., Okumus, B., Kim, D. S., & Ha, T. (2005). Extreme conformational diversity in human telomeric DNA. Proc Natl Acad Sci U S A, 102, 18938–18943.
Benítez, J. J., Keller, A. M., Ochieng, P., Yatsunyk, L. A., Huffman, D. L., Rosenzweig, A. C., & Chen, P. (2008). Probing transient copper chaperone− wilson disease protein interactions at the single-molecule level with nanovesicle trapping. Journal of the American Chemical Society, 130, 2446–2447.
Okumus, B., Arslan, S., Fengler, S. M., Myong, S., & Ha, T. (2009). Single molecule nanocontainers made porous using a bacterial toxin. Journal of the American Chemical Society, 131, 14844–14849.
Mandal, A. K., Ghosh, S., Das, A. K., Mondal, T., & Bhattacharyya, K. (2013). Effect of NaCl on ESPT‐mediated FRET in a CTAC micelle: a femtosecond and FCS study. ChemPhysChem, 14, 788–796.
Gooding, J. J., & Gaus, K. (2016). Single‐molecule sensors: challenges and opportunities for quantitative analysis. Angewandte Chemie International Edition, 55, 11354–11366.
Főrster, T. (1959). 10th Spiers Memorial Lecture. Transfer mechanisms of electronic excitation. Discussions of the Faraday Society, 27, 7–17.
Rolinski, O. J., & Birch, D. J. (2000). Determination of acceptor distribution from fluorescence resonance energy transfer: theory and simulation. The Journal of Chemical Physics, 112, 8923–8933.
Vermette, P., Griesser, H. J., Kambouris, P., & Meagher, L. (2004). Characterization of surface-immobilized layers of intact liposomes. Biomacromolecules, 5, 1496–1502.
Tachiya, M. (1982). Kinetics of quenching of luminescent probes in micellar systems. II. Journal of Chemical Physics, 76, 340–348.
Wolfram Reasearch Inc, https://reference.wolfram.com/language/ref/NIntegrate.html
ATTO-TEC, https://www.atto-tec.com/?cat=c45_R-0--Values--FRET--r-0-values-fret.html
TermoFisher Scientific, https://www.thermofisher.com/fr/fr/home/references/molecular-probes-the-handbook/tables/r0-values-for-some-alexa-fluor-dyes.html.html
Huang, C., Quinn, D., Sadovsky, Y., Suresh, S., & Hsia, K. J. (2017). Formation and size distribution of self-assembled vesicles. Proceedings of the National Academy of Sciences, 114, 2910–2915.
Maulucci, G., De Spirito, M., Arcovito, G., Boffi, F., Castellano, A. C., & Briganti, G. (2005). Particle size distribution in DMPC vesicles solutions undergoing different sonication times. Biophysical Journal, 88, 3545–3550.
Auer, A., Strauss, M. T., Schlichthaerle, T., & Jungmann, R. (2017). Fast, background-free DNA-PAINT imaging using FRET-based probes. Nano letters, 17, 6428–6434.
Bienert, R., Zimmermann, B., Rombach-Riegraf, V., & Gräber, P. (2011). Time‐dependent FRET with single enzymes: domain motions and catalysis in H+‐ATP synthases. ChemPhysChem, 12, 510–517.
Seyfert, K., Oosaka, T., Yaginuma, H., Ernst, S., Noji, H., Iino, R., & Börsch, M. (2011). Subunit rotation in a single FoF1-ATP synthase in a living bacterium monitored by FRET. Single Molecule Spectroscopy and Imaging IV, 7905, 79050K.
Mork, A. J., Weidman, M. C., Prins, F., & Tisdale, W. A. (2014). Magnitude of the Förster radius in colloidal quantum dot solids. The Journal of Physical Chemistry C, 118, 13920–13928.
Dexter, D. L. (1953). A theory of sensitized luminescence in solids. The Journal of Chemical Physics, 21, 836–850.
Oliver, R. C., Lipfert, J., Fox, D. A., Lo, R. H., Doniach, S., & Columbus, L. (2013). Dependence of micelle size and shape on detergent alkyl chain length and head group. PLoS ONE, 1, 8.
Yao, X., Choudhury, A. D., Yamanaka, Y. J., Adalsteinsson, V. A., Gierahn, T. M., Williamson, C. A., et al. (2014). Ex vivo expansion of circulating lung tumor cells based on one-step microfluidics-based immunomagnetic isolation. Integrative Biology : Quantitative Biosciences from Nano to Macro, 6, 388–398.
Zambrana-Puyalto, X., Maccaferri, N., Ponzellini, P., Giovannini, G., De Angelis, F., & Garoli, D. (2019). Site-selective functionalization of plasmonic nanopores for enhanced fluorescence emission rate and Förster resonance energy transfer. Nanoscale Advances, 1, 2454–2461.
Gnap, B., Bespalova, I., Yefimova, S., & Sorokin, A. (2011). FRET between cyanine dyes in nanopores of bulk sol-gel silica matrix. Functional Materials, 1, 1.
Gopich, I. V., & Szabo, A. (2012). Theory of the energy transfer efficiency and fluorescence lifetime distribution in single-molecule FRET. Proceedings of the National Academy of Sciences, 109, 7747–7752.
Kratky, O., & Porod, G. (1949). Röntgenuntersuchung gelöster fadenmoleküle. Recueil des Travaux Chimiques des Pays-Bas, 68, 1106–1122.
Borochov, N., Eisenberg, H., & Kam, Z. (1981). Dependence of DNA conformation on the concentration of salt. Biopolymers, 20, 231–235.
Acknowledgements
The authors wish to acknowledge Dr. Fabio Lisi for his kind assistance during this manuscript completion. J.J.G. acknowledges funding from the ARC centre of Excellence in Convergent Bio-Nano Science and Technology (CE140100036), the ARC Laureate Fellowship (FL150100060) program and a National Health and Medical Research Council program grant (1091261). K. G. acknowledges funding from the ARC Centre of Excellence in Advanced Molecular Imaging (CE140100011), the Australian Research Council (LP140100967 and DP130100269) and National Health and Medical Research Council of Australia (1059278 and 1037320).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Rights and permissions
About this article
Cite this article
Longatte, G., Gaus, K. & Gooding, J.J. FRET theoretical predictions concerning freely diffusive dyes inside spherical container: how to choose the best pair?. Photochem Photobiol Sci 20, 275–283 (2021). https://doi.org/10.1007/s43630-021-00016-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-021-00016-y