Abstract
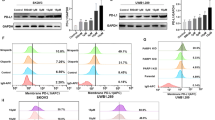
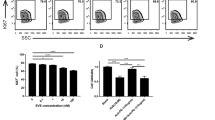
This study explores the synergistic effect of T-CUS245C combined with cyclosporin A, and its possible mechanism. T-CUS245C is an immunotoxin conjugated with tastuzumab and recombinant cucurmosin 245C (CUS245C). Cyclosporin A is a calcium antagonist mainly used as an immune-suppressive agent, which enhances the cytotoxicity of immunotoxins through an unknown mechanism. The sulforhodamine B assay, flow cytometry, DNA fragmentation apoptosis assay, and western blot analysis were used to evaluate the synergistic effects of cyclosporin A combined with T-CUS245C on proliferation inhibition and apoptosis induction in SK-OV-3 ovarian cancer cells with HER-2-overexpressing. Meanwhile, using confocal microscopy and flow cytometry, the effect of cyclosporin A on the intracellular distribution of T-CUS245C was observed and quantified. The results of this study showed that the combination index of cyclosporin A combined with T-CUS245C is less than 0.7, indicating that cyclosporin A significantly enhances the proliferation inhibition and apoptosis induction induced by T-CUS245C in SK-OV-3 cells. The confocal microscopy and flow cytometry results show that cyclosporin A effectively increases the dispersion of T-CUS245C in the cytoplasm by promoting the endolysosomal escape. This study demonstrates that cyclosporin A combined with T-CUS245C has a synergistic anticancer effect, by facilitating the release of T-CUS245C in the intracellular space.
Graphical Abstract





Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Akiyama S, Gottesman MM, Hanover JA, Fitzgerald DJ, Willingham MC, Pastan I (1984) Verapamil enhances the toxicity of conjugates of epidermal growth factor with Pseudomonas exotoxin and antitransferrin receptor with Pseudomonas exotoxin. J Cell Physiol 120:271–279. https://doi.org/10.1002/jcp.1041200303
Andersson Y, Engebraaten O, Fodstad O (2009) Synergistic anti-cancer effects of immunotoxin and cyclosporin in vitro and in vivo. Br J Cancer 101:1307–1315. https://doi.org/10.1038/sj.bjc.6605312
Ballabio A, Bonifacino JS (2020) Lysosomes as dynamic regulators of cell and organismal homeostasis. Nat Rev Mol Cell Biol 21:101–118. https://doi.org/10.1038/s41580-019-0185-4
Cabrera-Pérez MÁ, Pham-The H, Cervera MF, Hernández-Armengol R, Miranda-Pérez de Alejo C, Brito-Ferrer Y (2018) Integrating theoretical and experimental permeability estimations for provisional biopharmaceutical classification: application to the WHO essential medicines. Biopharm Drug Dispos 39:354–368. https://doi.org/10.1002/bdd.2152
Chang HL, Schwettmann B, McArthur HL, Chan IS (2023). Antibody-drug conjugates in breast cancer: overcoming resistance and boosting immune response. J Clin Invest 133. https://doi.org/10.1172/JCI172156
Chou TC (2010) Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res 70:440–446. https://doi.org/10.1158/0008-5472.CAN-09-1947
Cretella D, Saccani F, Quaini F, Frati C, Lagrasta C, Bonelli M, Caffarra C, Cavazzoni A, Fumarola C, Galetti M, La Monica S, Ampollini L, Tiseo M, Ardizzoni A, Petronini PG, Alfieri RR (2014) Trastuzumab emtansine is active on HER-2 overexpressing NSCLC cell lines and overcomes gefitinib resistance. Mol Cancer 13:143. https://doi.org/10.1186/1476-4598-13-143
Fanotto V, Ongaro E, Rihawi K, Avallone A, Silvestris N, Fornaro L, Vasile E, Antonuzzo L, Leone F, Rosati G, Giuliani F, Bordonaro R, Scartozzi M, De Maglio G, Negri FV, Fasola G, Aprile G (2016) HER-2 inhibition in gastric and colorectal cancers: tangible achievements, novel acquisitions and future perspectives. Oncotarget 7:69060–69074. https://doi.org/10.18632/oncotarget.11264
Fu Z, Li S, Han S, Shi C, Zhang Y (2022) Antibody drug conjugate: the “biological missile” for targeted cancer therapy. Sig Transduct Targeted Ther 7:93. https://doi.org/10.1038/s41392-022-00947-7
Fuchs H, Weng A, Gilabert-Oriol R (2016) Augmenting the efficacy of immunotoxins and other targeted protein toxins by endosomal escape enhancers. Toxins 8:200. https://doi.org/10.3390/toxins8070200
Grimm C, Bartel K, Vollmar AM, Biel M (2018) Endolysosomal cation channels and cancer a link with great potential. Pharmaceuticals 11:4. https://doi.org/10.3390/ph11010004
Hudis CA (2007) Trastuzumab-mechanism of action and use in clinical practice. N Engl J Med 357:39–51. https://doi.org/10.1056/NEJMra043186
Jaffrézou JP, Sikic BI, Laurent G (1994) Cyclosporin A and cyclosporin SDZ PSC 833 enhance anti-CD5 ricin A-chain immunotoxins in human leukemic T cells. Blood 83:482–489
Kim JS, Jun SY, Kim YS (2020) Critical issues in the deevelopment of immunotoxins for anticancer therapy. J Pharm Sci 109:104–115. https://doi.org/10.1016/j.xphs.2019.10.037
Kloudová K, Hromádková H, Partlová S, Brtnický T, Rob L, Bartůňková J, Hensler M, Halaška MJ, Špíšek R, Fialová A (2016) Expression of tumor antigens on primary ovarian cancer cells compared to established ovarian cancer cell lines. Oncotarget 7:46120–46126. https://doi.org/10.18632/oncotarget.10028
Lavaud P, Andre F (2014) Strategies to overcome trastuzumab resistance in HER2-overexpressing breast cancers: focus on new data from clinical trials. BMC Med 12:132. https://doi.org/10.1186/s12916-014-0132-3
Li BT, Michelini F, Misale S, Cocco E, Baldino L, Cai Y, Shifman S, Tu HY, Myers ML, Xu C, Mattar M, Khodos I, Little M, Qeriqi B, Weitsman G, Wilhem CJ, Lalani AS, Diala I, Freedman RA, Lin NU, Solit DB, Berger MF, Barber PR, Ng T, Offin M, Isbell JM, Jones DR, Yu HA, Thyparambil S, Liao WL, Bhalkikar A, Cecchi F, Hyman DM, Lewis JS, Buonocore DJ, Ho AL, Makker V, Reis-Filho JS, Razavi P, Arcila ME, Kris MG, Poirier JT, Shen R, Tsurutani J, Ulaner GA, de Stanchina E, Rosen N, Rudin CM, Scaltriti M (2020) HER2-mediated internalization of cytotoxic agents in ERBB2 amplified or mutant lung cancers. Cancer Discov 10:674–687. https://doi.org/10.1158/2159-8290.CD-20-0215
Nahta R (2012) Molecular mechanisms of trastuzumab-based treatment in HER2-overexpressing breast cancer. ISRN Oncol 2012:428062. https://doi.org/10.5402/2012/428062
Pegram MD, Miles D, Tsui CK, Zong Y (2020) HER2-overexpressing/amplified breast cancer as a testing ground for antibody-drug conjugate drug development in solid tumors. Clin Cancer Res 26:775–786. https://doi.org/10.1158/1078-0432.CCR-18-1976
Pirker R, FitzGerald DJ, Willingham MC, Pastan I (1988) Enhancement of the activity of immunotoxins made with either ricin A chain or Pseudomonas exotoxin in human ovarian and epidermoid carcinoma cell lines. Cancer Res 48:3919–3923
Smith WS, Johnston DA, Wensley HJ, Holmes SE, Flavell SU, Flavell DJ (2020) The role of cholesterol on triterpenoid saponin-induced endolysosomal escape of a saporin-based immunotoxin. Int J Mol Sci 21:8734. https://doi.org/10.3390/ijms21228734
Song ZT, Zhang LW, Fan LQ, Kong JW, Mao JH, Zhao J, Wang FJ (2018) Enhanced anticancer effect of MAP30-S3 by cyclosproin A through endosomal escape. Anticancer Drugs 29:736–747. https://doi.org/10.1097/CAD.0000000000000649
Valabrega G, Montemurro F, Aglietta M (2007) Trastuzumab: mechanism of action, resistance and future perspectives in HER2-overexpressing breast cancer. Ann Oncol 18:977–984. https://doi.org/10.1093/annonc/mdl475
Vargason AM, Anselmo AC, Mitragotri S (2021) The evolution of commercial drug delivery technologies. Nat Biomed Eng 5:951–967. https://doi.org/10.1038/s41551-021-00698-w
von Minckwitz G, Huang CS, Mano MS, Loibl S, Mamounas EP, Untch M, Wolmark N, Rastogi P, Schneeweiss A, Redondo A, Fischer HH, Jacot W, Conlin AK, Arce-Salinas C, Wapnir IL, Jackisch C, DiGiovanna MP, Fasching PA, Crown JP, Wülfing P, Shao Z, Rota Caremoli E, Wu H, Lam LH, Tesarowski D, Smitt M, Douthwaite H, Singel SM, Geyer CE Jr, Katherine Investigators (2019) Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N Engl J Med 380:617–628. https://doi.org/10.1056/NEJMoa1814017
Wensley HJ, Smith WS, Holmes SE, Flavell SU, Flavell DJ (2021) The effect of small molecule pharmacological agents on the triterpenoid saponin induced endolysosomal escape of saporin and a saporin-based immunotoxin in target human lymphoma cells. Biomedicines 9:300. https://doi.org/10.3390/biomedicines9030300
Xiong J, Zhang C, Wu S, Gu X, Cai Y, Xu C, Chen Z, Sun J, Wu X, You X, Huang Z, Xie J (2019) Recombinant cucurmosin-based immunotoxin targeting HER-2 with potent in vitro anti-cancer cytotoxicity. Biochem Biophys Res Commun 513:15–21. https://doi.org/10.1016/j.bbrc.2019.03.090
Zhang C, Cai Y, Dai X, Wu J, Lan Y, Zhang H, Lu M, Liu J, Xie J (2021) Novel EGFR-bispecific recombinant immunotoxin based on cucurmosin shows potent anti-tumor efficiency in vitro. Oncol Rep 45:493–500. https://doi.org/10.3892/or.2020.7878
Acknowledgements
We thank the public technology service center of Fujian Medical University for helping with the experiment.
Funding
This research was funded by the National Science Foundation of China (No. 30772587), Science Foundation of State Key Laboratory of Structural Chemistry (No. 20210001), Fujian Provincial Central Leading Local Science and Technology Development Fund Project (No. 2021L3025), the Natural Science Foundation of Fujian Province (No. 2020J01436), the Fujian Province Health and Family Planning Scientific Research Talent Training Project (No. 2018-CX-40), Young and Middle-aged Scientific Research Project of Ningde Normal University (No. 2020Q103), the Startup Fund for Scientific Research (No. 2018QH1013), and the Innovation Team project of Ningde Normal University (No. 2022T03).
Author information
Authors and Affiliations
Contributions
Study concept and design: HX, JX, XY; acquisition of data: HX, YF; analysis and interpretation of data: HX, ZL, YF; drafting of the manuscript: HX; critical revision of the manuscript for important intellectual content: JX, XY; statistical analysis: HX, YF; obtained funding: JX, XY; administrative, technical, or material support: JX; study supervision: JX, XY.
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xie, H., Fu, Y., Lin, Z. et al. Cyclosporin A Enhance the Cytotoxicity of Immunotoxin to the HER-2-Overexpressing SK-OV-3 Cells: A Prospective Study. Rev. Bras. Farmacogn. (2024). https://doi.org/10.1007/s43450-024-00551-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43450-024-00551-6