Abstract
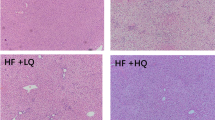
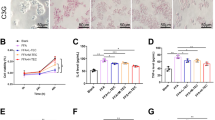
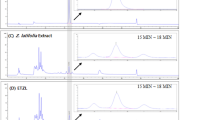
Non-alcoholic fatty liver disease is characterized by liver damage and steatosis, which is often accompanied by inflammation. Tartary buckwheat flavone has been shown to have a range of anti-inflammatory effects, but its mechanism of action on non-alcoholic fatty liver disease remains unclear. In this study, a non-alcoholic fatty liver disease model induced by high-fat diet in vivo and HepG2 cells stimulated by palmitic acid in vitro were constructed, and the effect of tartary buckwheat flavone on high-fat diet-induced non-alcoholic fatty liver disease and its potential mechanism was explored by H&E staining, PCR, Western blot, and 16S rRNA high-throughput sequencing. The results showed that compared with the high-fat diet group, tartary buckwheat flavone significantly reduced liver and body weight and alleviated pathological damage of liver tissue in mice, and the biochemical indexes of total cholesterol, triacylglyceride, alanine aminotransferase, and aspartate aminotransferase were significantly decreased. Further studies showed that tartary buckwheat flavone significantly reduced the protein levels of inflammatory mediators myeloperoxidase, iNOS, COX2, IL-6, IL-1β, and TNF-α. In vitro studies also showed that tartary buckwheat flavone significantly improved palmitic acid–induced HepG2 cell lipid accumulation and increased inflammatory factors. Mechanism studies showed that tartary buckwheat flavone alleviated the disorder of lipid metabolism by activating AMPK, decreasing sterol regulatory element-binding protein-1 (SREBP1) and acetyl-COA carboxylase protein levels. Tartary buckwheat flavone significantly inhibited high-fat diet-induced MAPK and NF-κB signaling pathways to reduce the release of pro-inflammatory mediators, and then alleviate the inflammatory response. In addition, tartary buckwheat flavone significantly alleviated the intestinal flora disorder induced by high-fat diet through increasing the relative abundance of beneficial bacteria Dubosiella and Bacteroidetes. Our study found that tartary buckwheat flavone alleviates non-alcoholic fatty liver disease by mitigating the disorder of lipid metabolism and inflammatory response and regulating the composition of intestinal flora.
Graphical Abstract








Similar content being viewed by others
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
AlAsmari AF, Alharbi M, Alqahtani F, Alasmari F, AlSwayyed M, Alzarea SI, Al-Alallah IA, Alghamdi A, Hakami HM, Alyousef MK, Sari Y, Ali N (2021) Diosmin alleviates doxorubicin-induced liver injury via modulation of oxidative stress-mediated hepatic inflammation and apoptosis via NfkB and MAPK pathway: a preclinical study. Antioxidants 10:1998. https://doi.org/10.3390/antiox10121998
Aron-Wisnewsky J, Vigliotti C, Witjes J, Le P, Holleboom AG, Verheij J, Nieuwdorp M, Clement K (2020) Gut microbiota and human NAFLD: disentangling microbial signatures from metabolic disorders. Nat Rev Gastroenterol Hepatol 17:279–297. https://doi.org/10.1038/s41575-020-0269-9
Baldus S, Heeschen C, Meinertz T, Zeiher AM, Eiserich JP, Munzel T, Simoons ML, Hamm CW, Investigators C (2003) Myeloperoxidase serum levels predict risk in patients with acute coronary syndromes. Circulation 108:1440–1445. https://doi.org/10.1161/01.CIR.0000090690.67322.51
Bohm T, Berger H, Nejabat M, Riegler T, Kellner F, Kuttke M, Sagmeister S, Bazanella M, Stolze K, Daryabeigi A, Bintner N, Murkovic M, Wagner KH, Schulte-Hermann R, Rohr-Udilova N, Huber W, Grasl-Kraupp B (2013) Food-derived peroxidized fatty acids may trigger hepatic inflammation: a novel hypothesis to explain steatohepatitis. J Hepatol 59:563–570. https://doi.org/10.1016/j.jhep.2013.04.025
Boursier J, Mueller O, Barret M, Machado M, Fizanne L, Araujo-Perez F, Guy CD, Seed PC, Rawls JF, David LA, Hunault G, Oberti F, Cales P, Diehl AM (2016) The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology 63:764–775. https://doi.org/10.1002/hep.28356
Bulutoglu B, Rey-Bedon C, Kang YBA, Mert S, Yarmush ML, Usta OB (2019) A microfluidic patterned model of non-alcoholic fatty liver disease: applications to disease progression and zonation. Lab Chip 19:3022–3031. https://doi.org/10.1039/c9lc00354a
Cani PD, Bibiloni R, Knauf C, Waget A, Neyrinck AM, Delzenne NM, Burcelin R (2008) Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 57:1470–1481. https://doi.org/10.2337/db07-1403
Carling D (2017) AMPK signalling in health and disease. Curr Opin Cell Biol 45:31–37. https://doi.org/10.1016/j.ceb.2017.01.005
Deng Y, Tang K, Chen R, Liu Y, Nie H, Wang H, Zhang Y, Yang Q (2018) Effects of Shugan-Jianpi recipe on the expression of the p38 MAPK/NF-kappaB signaling pathway in the hepatocytes of NAFLD rats. Medicines 5:106. https://doi.org/10.3390/medicines5030106
Eslam M, Valenti L, Romeo S (2018) Genetics and epigenetics of NAFLD and NASH: clinical impact. J Hepatol 68:268–279. https://doi.org/10.1016/j.jhep.2017.09.003
Fabjan N, Rode J, Kosir IJ, Wang Z, Zhang Z, Kreft I (2003) Tartary buckwheat (Fagopyrum tataricum Gaertn.) as a source of dietary rutin and quercitrin. J Agric Food Chem 51:6452–6455. https://doi.org/10.1021/jf034543e
Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ (2018) Mechanisms of NAFLD development and therapeutic strategies. Nat Med 24:908–922. https://doi.org/10.1038/s41591-018-0104-9
Ge X, Wang C, Chen H, Liu T, Chen L, Huang Y, Zeng F, Liu B (2020) Luteolin cooperated with metformin hydrochloride alleviates lipid metabolism disorders and optimizes intestinal flora compositions of high-fat diet mice. Food Funct 11:10033–10046. https://doi.org/10.1039/d0fo01840f
Heinonen I, Rinne P, Ruohonen ST, Ruohonen S, Ahotupa M, Savontaus E (2014) The effects of equal caloric high fat and western diet on metabolic syndrome, oxidative stress and vascular endothelial function in mice. Acta Physiol 211:515–527. https://doi.org/10.1111/apha.12253
Jasirwan COM, Muradi A, Hasan I, Simadibrata M, Rinaldi I (2021) Correlation of gut Firmicutes/Bacteroidetes ratio with fibrosis and steatosis stratified by body mass index in patients with non-alcoholic fatty liver disease. Biosci Microbiota Food Health 40:50–58. https://doi.org/10.12938/bmfh.2020-046
Kameyama K, Itoh K (2014) Intestinal colonization by a Lachnospiraceae bacterium contributes to the development of diabetes in obese mice. Microbes Environ 29:427–430. https://doi.org/10.1264/jsme2.ME14054
Kan X, Liu B, Guo W, Wei L, Lin Y, Guo Y, Gong Q, Li Y, Xu D, Cao Y, Huang B, Dong A, Ma H, Fu S, Liu J (2019) Myricetin relieves LPS-induced mastitis by inhibiting inflammatory response and repairing the blood-milk barrier. J Cell Physiol 234:16252–16262. https://doi.org/10.1002/jcp.28288
Kang OH, Kim SB, Mun SH, Seo YS, Hwang HC, Lee YM, Lee HS, Kang DG, Kwon DY (2015) Puerarin ameliorates hepatic steatosis by activating the PPARalpha and AMPK signaling pathways in hepatocytes. Int J Mol Med 35:803–809. https://doi.org/10.3892/ijmm.2015.2074
Keirns BH, Hart SM, Sciarrillo CM, Poindexter KL, Clarke SL, Emerson SR (2022) Postprandial triglycerides, endothelial function, and inflammatory cytokines as potential candidates for early risk detection in normal-weight obesity. Obes Res Clin Pract 16:386–392. https://doi.org/10.1016/j.orcp.2022.08.008
Koh YC, Lin YC, Lee PS, Lu TJ, Lin KY, Pan MH (2020) A multi-targeting strategy to ameliorate high-fat-diet- and fructose-induced (western diet-induced) non-alcoholic fatty liver disease (NAFLD) with supplementation of a mixture of legume ethanol extracts. Food Funct 11:7545–7560. https://doi.org/10.1039/d0fo01405b
Kolarova H, Klinke A, Kremserova S, Adam M, Pekarova M, Baldus S, Eiserich JP, Kubala L (2013) Myeloperoxidase induces the priming of platelets. Free Radic Biol Med 61:357–369. https://doi.org/10.1016/j.freeradbiomed.2013.04.014
Krithika R, Jyothilakshmi V, Verma RJ (2016) Phyllanthin inhibits CCl4-mediated oxidative stress and hepatic fibrosis by down-regulating TNF-alpha/NF-kappaB, and pro-fibrotic factor TGF-beta1 mediating inflammatory signaling. Toxicol Ind Health 32:953–960. https://doi.org/10.1177/0748233714532996
Lau E, Carvalho D, Freitas P (2015) Gut microbiota: association with NAFLD and metabolic disturbances. Biomed Res Int 2015: 979515. https://doi.org/10.1155/2015/979515
Lee Y, Hu S, Park YK, Lee JY (2019) Health benefits of carotenoids: a role of carotenoids in the prevention of non-alcoholic fatty liver disease. Prev Nutr Food Sci 24:103–113. https://doi.org/10.3746/pnf.2019.24.2.103
Li J, Wang T, Liu P, Yang F, Wang X, Zheng W, Sun W (2021) Hesperetin ameliorates hepatic oxidative stress and inflammation via the PI3K/AKT-Nrf2-ARE pathway in oleic acid-induced HepG2 cells and a rat model of high-fat diet-induced NAFLD. Food Funct 12:3898–3918. https://doi.org/10.1039/d0fo02736g
Li Y, Li J, Su Q, Liu Y (2019) Sinapine reduces non-alcoholic fatty liver disease in mice by modulating the composition of the gut microbiota. Food Funct 10:3637–3649. https://doi.org/10.1039/c9fo00195f
Li Y, Xu S, Mihaylova MM, Zheng B, Hou X, Jiang B, Park O, Luo Z, Lefai E, Shyy JY, Gao B, Wierzbicki M, Verbeuren TJ, Shaw RJ, Cohen RA, Zang M (2011) AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metab 13:376–388. https://doi.org/10.1016/j.cmet.2011.03.009
Liu CL, Chen YS, Yang JH, Chiang BH (2008) Antioxidant activity of tartary (Fagopyrum tataricum (L.) Gaertn.) and common (Fagopyrum esculentum moench) buckwheat sprouts. J Agric Food Chem 56:173–178. https://doi.org/10.1021/jf072347s
Liu W, Baker RD, Bhatia T, Zhu L, Baker SS (2016) Pathogenesis of nonalcoholic steatohepatitis. Cell Mol Life Sci 73:1969–1987. https://doi.org/10.1007/s00018-016-2161-x
Long P, Hu J, Piesco N, Buckley M, Agarwal S (2001) Low magnitude of tensile strain inhibits IL-1beta-dependent induction of pro-inflammatory cytokines and induces synthesis of IL-10 in human periodontal ligament cells in vitro. J Dent Res 80:1416–1420. https://doi.org/10.1177/00220345010800050601
Luedde T, Schwabe RF (2011) NF-kappaB in the liver–linking injury, fibrosis and hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 8:108–118. https://doi.org/10.1038/nrgastro.2010.213
Lupp C, Robertson ML, Wickham ME, Sekirov I, Champion OL, Gaynor EC, Finlay BB (2007) Host-mediated inflammation disrupts the intestinal microbiota and promotes the overgrowth of Enterobacteriaceae. Cell Host Microbe 2:204. https://doi.org/10.1016/j.chom.2007.08.002
Mavrelis PG, Ammon HV, Gleysteen JJ, Komorowski RA, Charaf UK (1983) Hepatic free fatty acids in alcoholic liver disease and morbid obesity. Hepatology 3:226–231. https://doi.org/10.1002/hep.1840030215
Mazzoncini M, Antichi D, Silvestri N, Ciantelli G, Sgherri C (2015) Organically vs conventionally grown winter wheat: effects on grain yield, technological quality, and on phenolic composition and antioxidant properties of bran and refined flour. Food Chem 175:445–451. https://doi.org/10.1016/j.foodchem.2014.11.138
Musso G, Gambino R, Cassader M (2009) Recent insights into hepatic lipid metabolism in non-alcoholic fatty liver disease (NAFLD). Prog Lipid Res 48:1–26. https://doi.org/10.1016/j.plipres.2008.08.001
Owecki M, Nikisch E, Miczke A, Pupek-Musialik D, Sowinski J (2010) Serum resistin is related to plasma HDL cholesterol and inversely correlated with LDL cholesterol in diabetic and obese humans. Neuro Endocrinol Lett 31:673–678
Peng L, Zhang Q, Zhang Y, Yao Z, Song P, Wei L, Zhao G, Yan Z (2020) Effect of tartary buckwheat, rutin, and quercetin on lipid metabolism in rats during high dietary fat intake. Food Sci Nutr 8:199–213. https://doi.org/10.1002/fsn3.1291
Pierantonelli I, Svegliati-Baroni G (2019) Nonalcoholic fatty liver disease: basic pathogenetic mechanisms in the progression from NAFLD to NASH. Transplantation 103:e1–e13. https://doi.org/10.1097/TP.0000000000002480
Ran X, Liu J, Fu S, He F, Li K, Hu G, Guo W (2022) Phytic acid maintains the integrity of the blood-milk barrier by regulating inflammatory response and intestinal flora structure. J Agric Food Chem 70:381–391. https://doi.org/10.1021/acs.jafc.1c06270
Santamarina AB, Oliveira JL, Silva FP, Carnier J, Mennitti LV, Santana AA, de Souza GH, Ribeiro EB, Oller do Nascimento CM, Lira FS, Oyama LM (2015) Green tea extract rich in epigallocatechin-3-gallate prevents fatty liver by AMPK activation via LKB1 in mice fed a high-fat diet. PLoS ONE 10:e0141227. https://doi.org/10.1371/journal.pone.0141227
Sharma A, Anand SK, Singh N, Dwarkanath A, Dwivedi UN, Kakkar P (2021) Berbamine induced activation of the SIRT1/LKB1/AMPK signaling axis attenuates the development of hepatic steatosis in high-fat diet-induced NAFLD rats. Food Funct 12:892–909. https://doi.org/10.1039/d0fo02501a
Stojanov S, Berlec A, Strukelj B (2020) The influence of probiotics on the Firmicutes/Bacteroidetes ratio in the treatment of obesity and inflammatory bowel disease. Microorganisms 8:1715. https://doi.org/10.3390/microorganisms8111715
Sun W, Liu P, Wang T, Wang X, Zheng W, Li J (2020) Baicalein reduces hepatic fat accumulation by activating AMPK in oleic acid-induced HepG2 cells and high-fat diet-induced non-insulin-resistant mice. Food Funct 11:711–721. https://doi.org/10.1039/c9fo02237f
Valenzuela R, Illesca P, Echeverria F, Espinosa A, Rincon-Cervera MA, Ortiz M, Hernandez-Rodas MC, Valenzuela A, Videla LA (2017) Molecular adaptations underlying the beneficial effects of hydroxytyrosol in the pathogenic alterations induced by a high-fat diet in mouse liver: PPAR-alpha and Nrf2 activation, and NF-kappaB down-regulation. Food Funct 8:1526–1537. https://doi.org/10.1039/c7fo00090a
Wang L, Pan MH, Lo CY, Zhao H, Li S, Ho CT, Yang G (2018) Anti-fibrotic activity of polyphenol-enriched sugarcane extract in rats via inhibition of p38 and JNK phosphorylation. Food Funct 9:951–958. https://doi.org/10.1039/c7fo01617d
Weiss J, Rau M, Geier A (2014) Non-alcoholic fatty liver disease: epidemiology, clinical course, investigation, and treatment. Dtsch Arztebl Int 111:447–452. https://doi.org/10.3238/arztebl.2014.0447
Wu PS, Ding HY, Yen JH, Chen SF, Lee KH, Wu MJ (2018) Anti-inflammatory activity of 8-hydroxydaidzein in LPS-stimulated BV2 microglial cells via activation of Nrf2-antioxidant and attenuation of Akt/NF-kappaB-inflammatory signaling pathways, as well as inhibition of COX-2 activity. J Agric Food Chem 66:5790–5801. https://doi.org/10.1021/acs.jafc.8b00437
Wu Z, Zhang Y, Gong X, Cheng G, Pu S, Cai S (2020) The preventive effect of phenolic-rich extracts from Chinese sumac fruits against nonalcoholic fatty liver disease in rats induced by a high-fat diet. Food Funct 11:799–812. https://doi.org/10.1039/c9fo02262g
Xie SJ, Diao LT, Cai N, Zhang LT, Xiang S, Jia CC, Qiu DB, Liu C, Sun YJ, Lei H, Hou YR, Tao S, Hu YX, Xiao ZD, Zhang Q (2021) mascRNA and its parent lncRNA MALAT1 promote proliferation and metastasis of hepatocellular carcinoma cells by activating ERK/MAPK signaling pathway. Cell Death Discov 7:110. https://doi.org/10.1038/s41420-021-00497-x
Xu W, Chen J, Lin J, Liu D, Mo L, Pan W, Feng J, Wu W, Zheng D (2015) Exogenous H2S protects H9c2 cardiac cells against high glucose-induced injury and inflammation by inhibiting the activation of the NF-kappaB and IL-1beta pathways. Int J Mol Med 35:177–186. https://doi.org/10.3892/ijmm.2014.2007
Yang CJ, Lin JL, Lin-Tan DT, Weng CH, Hsu CW, Lee SY, Lee SH, Chang CM, Lin WR, Yen TH (2012) Spectrum of toxic hepatitis following intentional paraquat ingestion: analysis of 187 cases. Liver Int 32:1400–1406. https://doi.org/10.1111/j.1478-3231.2012.02829.x
Acknowledgements
The authors would like to thank all friends and colleagues for their valuable insights and recommendation and also for their contribution in conducting some of the experiments for this research.
Funding
This work was supported by the Jilin Young Scientific and Technological Talents Promotion Project, QT202127.
Author information
Authors and Affiliations
Contributions
PX and JL performed the experiments, prepared the figures, and wrote the manuscript. SF designed and supervised the study. ZL and WG participated in the feeding of mice, sample collection, and preparation in the experimental process. XK and YC performed the statistical analyses. GH contributed to the editing of the manuscript. All authors have read and approved the manuscript prior to submission.
Corresponding author
Ethics declarations
Ethical Approval
The experiment was carried out in the Experimental Animal Center of Jilin University (SYXK (JI) 2016-0001) under the supervision of the Animal Ethics and Welfare Committee of Jilin University (IACUC). It complies with the requirements of Jilin University and the state for the ethical welfare of experimental animals.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, P., Liu, J., Li, Z. et al. Tartary Buckwheat Flavonoids Relieve Non-alcoholic Fatty Liver Disease by Inhibiting Lipid Accumulation, Inflammation, and Regulating Intestinal Flora. Rev. Bras. Farmacogn. 33, 965–979 (2023). https://doi.org/10.1007/s43450-023-00406-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-023-00406-6