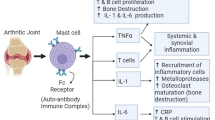
Abstract
Most species of fungi are recognized as etiological agents from a vast number of human and animal diseases. Contrarily, in the Natural Products research field, it is considered a promising source of bioactive metabolites. The chemical diversity of fungi metabolism combined with potent biological activities might grant fungi metabolites to unearth new anti-inflammatory pharmacological therapies. On this basis, the pathological inflammation process is associated with several diseases, such as gout arthritis, autoimmune diseases, neurodegenerative disorders and several types of cancer. In the last 20 years, it was isolated from different Aspergillus strains anti-inflammatory metabolites with unique structural chemical patterns and high potency effect. Therefore, this systematic review aimed to identify the metabolites from members of the genus Aspergillus evaluated by anti-inflammatory assays as the main targets of pharmacological activity. From 231 anti-inflammatory metabolites that were descripted in this review, four exhibited high potential for multi-targeting inhibition. The current evidence demonstrates that the Aspergillus species are a rich source of compounds with significant anti-inflammatory properties that includes the alkaloids, butenolides, terpenoids and polyketides. In addition, an anti-inflammatory Aspergillus chemical database was built to support the chemical aspects of the review. Nevertheless, still, there is a lack of in vivo evaluations in the literature, and that is crucial to increase the knowledge about the mechanisms of actions of these fungal metabolites.
Graphical Abstract






Similar content being viewed by others
References
Abdel-Azeem AM, Abdel-Azeem MA, Khalil WF (2019) Endophytic fungi as a new source of antirheumatoid metabolites. In: Watson RR, Preedy VR (eds) Bioactive food as dietary interventions for arthritis and related inflammatory diseases. Academic Press, Massachusetts, pp. 355–384. https://doi.org/10.1016/B978-0-12-813820-5.00021-0
Chagas-Paula DA, Oliveira TB, Zhang T, Edrada-Ebel R, Da Costa FB (2015a) Prediction of anti-inflammatory plants and discovery of their biomarkers by machine learning algorithms and metabolomic studies. Planta Med 81:450–458. https://doi.org/10.1055/s-0034-1396206
Chagas-Paula DA, Zhang T, Da Costa FB, Edrada-Ebel R (2015b) A metabolomic approach to target compounds from the Asteraceae family for dual COX and LOX inhibition. Metabolites 5:404–430. https://doi.org/10.3390/metabo5030404
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420:860–867. https://doi.org/10.1038/nature01322
Cragg GM, Pezzuto JM (2016) Natural products as a vital source for the discovery of cancer chemotherapeutic and chemopreventive agents. Med Princ Pract 25:41–59. https://doi.org/10.1159/000443404
Ding K, Zhou Z, Hou S, Yuan Y, Zhou S, Zheng X, Chen J, Loftin C, Zheng F, Zhan CG (2018) Structure-based discovery of mPGES-1 inhibitors suitable for preclinical testing in wild-type mice as a new generation of anti-inflammatory drugs. Sci Rep 8:5205. https://doi.org/10.1038/s41598-018-23482-4
Du C, Bhatia M, Tang SCW, Zhang M, Steiner T (2015) Mediators of inflammation: inflammation in cancer, chronic diseases, and wound healing. Mediators Inflamm 2015:2–4. https://doi.org/10.1155/2015/570653
Du X, Liu D, Huang J, Zhang C, Proksch P, Lin W (2018) Polyketide derivatives from the sponge associated fungus Aspergillus europaeus with antioxidant and NO inhibitory activities. Fitoterapia 130:190–197
El-Kashef DH, Youssef FS, Hartmann R, Knedel T-O, Janiak C, Lin W, Reimche I, Teusch N, Liu Z, Proksch P (2020) Azaphilones from the red sea fungus Aspergillus falconensis. Mar Drugs 18:1-10. https://doi.org/10.1016/j.intimp.2019.04.045
El-Hawary SS, Moawad AS, Bahr HS, Abdelmohsen UR, Mohammed R (2020) Natural product diversity from the endophytic fungi of the genus Aspergillus. RSC Adv 10:22058–22079. https://doi.org/10.1039/d0ra04290k
Fang W, Lin X, Wang J, Liu Y, Tao H, Zhou X (2016) Asperpyrone-type bis-naphtho-a-pyrones with COX-2-inhibitory activities from marine-derived fungus Aspergillus niger. Molecules 21:4–11. https://doi.org/10.3390/molecules21070941
Fiorucci S, Meli R, Bucci M, Cirino G (2001) Dual inhibitors of cyclooxygenase and 5-lipoxygenase. A new avenue in anti-inflammatory therapy? Biochem Pharmacol 62:1433–1438. https://doi.org/10.1016/s0006-2952(01)00747-x
Flandroy L, Poutahidis T, Berg G, Clarke G, Dao MC, Decaestecker E, Furman E, Haahtela T, Massart S, Plovier H, Sanz Y, Rook G (2018) The impact of human activities and lifestyles on the interlinked microbiota and health of humans and of ecosystems. Sci Total Environ 627:1018–1038. https://doi.org/10.1016/j.scitotenv.2018.01.288
Gu BB, Jiao FR, Wu W, Jiao WH, Li L, Sun F, Wang SP, Yang F, Lin HW (2018) Preussins with Inhibition of IL-6 Expression from Aspergillus flocculosus 16D–1, a fungus isolated from the marine sponge Phakellia fusca. J Nat Prod 81:2275–3228. https://doi.org/10.1021/acs.jnatprod.8b00662
Gubiani JR, Zeraik ML, Oliveira CM, Ximenes VF, Nogueira CR, Fonseca LM, Silva DHS, Bolzani VS, Araujo AR (2014) Biologically active eremophilane-type sesquiterpenes from Camarops sp., an endophytic fungus isolated from Alibertia macrophylla. J Nat Prod 77:668–672. https://doi.org/10.1021/np400825s
Gupta S, Chaturvedi P, Kulkarni MG, Van Staden J (2020) A critical review on exploiting the pharmaceutical potential of plant endophytic fungi. Biotechnol Adv 39:107462. https://doi.org/10.1016/j.biotechadv.2019.107462
Hartung T, de Vries R, Hoffmann S, Hogberg HT, Smirnova L, Tsaioun K, Whaley P, Leist M (2019) Toward good in vitro reporting standards. ALTEX 36:3–17
Hawksworth DL (2011) Naming Aspergillus species: Progress towards one name for each species. Med Mycol J 49:70–76. https://doi.org/10.3109/13693786.2010.504753
Hu Y, Zhang W, Zhang P, Ruan W, Zhu X (2013) Nematicidal activity of chaetoglobosin a produced by Chaetomium globosum NK102 against Meloidogyne incognita. J Agric Food Chem 61:41–46. https://doi.org/10.1021/jf304314g
Huang X, Men P, Tang S, Lu X (2021) Aspergillus terreus as an industrial filamentous fungus for pharmaceutical biotechnology. Curr Opin Biotechnol 69:273–280. https://doi.org/10.1016/j.copbio.2021.02.004
Hwang D, Son BW, Shin PG, Choi JS, Seo YB, Kim GDo (2015) Toluhydroquinone from Aspergillus sp. suppress inflammatory mediators via nuclear factor-κB and mitogen-activated protein kinases pathways in lipopolysaccharide-induced RAW264.7 cells. J Pharm Pharmacol 67:1297–1305. https://doi.org/10.1111/jphp.12421
Kim DC, Cho KH, Ko W, Yoon CS, Sohn JH, Yim JH, Kim YC, Oh H (2016) Anti-inflammatory and cytoprotective effects of TMC-256C1 from marine-derived fungus Aspergillus sp. SF-6354 via up-regulation of heme oxygenase-1 in murine hippocampal and microglial cell lines. Int J Mol Sci 17:1–18. https://doi.org/10.3390/ijms17040529
Kim NH, Jung HI, Choi WS, Son BW, Seo YB, Choi JS, Kim GD (2015) Toluhydroquinone, the secondary metabolite of marine algae symbiotic microorganism, inhibits angiogenesis in HUVECs. Biomed Pharmacother 70:129–139. https://doi.org/10.1016/j.biopha.2015.01.004
Kim KW, Kim HJ, Sohn JH, Yim JH, Kim YC, Oh H (2018) Anti-neuroinflammatory effect of 6,8,1′-tri-O-methylaverantin, a metabolite from a marine-derived fungal strain Aspergillus sp., via upregulation of heme oxygenase-1 in lipopolysaccharide-activated microglia. Neurochem Int 113:8–22. https://doi.org/10.1016/j.neuint.2017.11.010
Kwon J, Lee H, Ko W, Kim DC, Kim KW, Kwon HC, Guo Y, Sohn JH, Yim JH, Kim YC, Oh H, Lee D (2017) Chemical constituents isolated from Antarctic marine-derived Aspergillus sp. SF-5976 and their anti-inflammatory effects in LPS-stimulated RAW 264.7 and BV2 cells. Tetrahedron 73:3905–3912. https://doi.org/10.1016/j.tet.2017.05.060
Landskron G, De-La-Fuente M, Thuwajit P, Thuwajit C, Hermoso MA (2014) Chronic inflammation and cytokines in the tumor microenvironment. J Immunol Res 2014:1–19. https://doi.org/10.1155/2014/149185
Latgé JP (1999) Aspergillus fumigatus and aspergillosis. Clin Microbiol Rev 12:310–350. https://doi.org/10.1128/cmr.12.2.310
Lee DS, Jeong GS, Li B, Lee SU, Oh H, Kim YC (2011) Asperlin from the marine-derived fungus Aspergillus sp. SF-5044 exerts anti-inflammatory effects through heme oxygenase-1 expression in murine macrophages. J Pharmacol Sci 116:283–295. https://doi.org/10.1254/jphs.10219FP
Leutou AS, Yun KJ, Choi HD, Kang JS, Son BW (2012) New production of 5-bromotoluhydroquinone and 4-O-methyltoluhydroquinone from the marine-derived fungus Dothideomycete sp. J Microbiol Biotechnol 22:80–83. https://doi.org/10.4014/jmb.1108.08069
Li SJ, Zhang X, Wang XH, Zhao CQ (2018) Novel natural compounds from endophytic fungi with anticancer activity. Eur J Med Chem 156:316–343. https://doi.org/10.1016/j.ejmech.2018.07.015
Liao G, Wu P, Xue J, Liu L, Li H, Wei X (2018) Asperimides A-D, anti-inflammatory aromatic butenolides from a tropical endophytic fungus Aspergillus terreus. Fitoterapia 131:50–54. https://doi.org/10.1016/j.fitote.2018.10.011
Liu M, Zhou Q, Wang J, Liu J, Qi C, Lai Y, Zhu H, Xue Y, Hu Z, Zhang Y (2018a) Anti-inflammatory butenolide derivatives from the coral-derived fungus: Aspergillus terreus and structure revisions of aspernolides D and G, butyrolactone VI and 4′,8′′-diacetoxy butyrolactone VI. RSC Adv 8:13040–13047. https://doi.org/10.1039/c8ra01840e
Liu Z, Dong Z, Qiu P, Wang Q, Yan J, Lu Y, Wasu PA, Hong K, She Z (2018b) Two new bioactive steroids from a mangrove-derived fungus Aspergillus sp. Steroids 140:32–38. https://doi.org/10.1016/j.steroids.2018.08.009
Lopes F, Regasini O, Pinheiro M, Soares R (2013) Encyclopedia of toxicology. Nat Prod 3:468–469. https://doi.org/10.1016/B978-0-12-386454-3.00972-6
Ma Y, Ling TJ, Su XQ, Jiang B, Nian B, Chen LJ, Liu ML, Zhang ZY, Wang DP, Mu YY, Jiao WW, Liu QT, Pan YH, Zhao M, (2021) Integrated proteomics and metabolomics analysis of tea leaves fermented by Aspergillus niger, Aspergillus tamarii and Aspergillus fumigatus. Food Chem 334:127560. https://doi.org/10.1016/j.foodchem.2020.127560
Marinho AMR, Rodrigues-filho E, Moitinho MLR, Santos LS (2005) Biologically active polyketides produced by Penicillium janthinellum isolated as an endophytic fungus from fruits of Melia azedarach. J Braz Chem Soc 16:280–283. https://doi.org/10.1590/s0103-50532005000200023
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, Shekelle P (2015) Preferred reporting items for systematic review and meta-analysis protocols (prisma-p) 2015 statement. Syst Rev 4:2–9. https://doi.org/10.1186/2046-4053-4-1
Patil RH, Patil MP, Maheshwari VL (2016) Bioactive secondary metabolites from endophytic fungi: a review of biotechnological production and their potential applications. Stud Nat Prod Chem 49:189–205. https://doi.org/10.1016/B978-0-444-63601-0.00005-3
Rai M, Rathod D, Agarkar G, Dar M, Brestic M, Pastore GM, Junior MRM (2014) Fungal growth promotor endophytes: a pragmatic approach towards sustainable food and agriculture. Symbiosis 62:63–79. https://doi.org/10.1007/s13199-014-0273-3
Rodrigues KF, Dias-Filho MB (1996) Fungal endophytes in the tropical grasses Brachiaria brizantha cv. marandu and B. humidicola. Pesqui Agropecu Bras 31:905–909. https://doi.org/10.1590/1678-992X-2018-0210
Segaran G, Sathiavelu M (2019) Fungal endophytes: a potent biocontrol agent and a bioactive metabolites reservoir. Biocatal Agric Biotechnol 21:101284. https://doi.org/10.1016/j.bcab.2019.101284
Stajich JE, Berbee ML, Blackwell M, Hibbett DS, James TY, Spatafora JW, Taylor JW (2009) The Fungi. Curr Biol 19:840–845. https://doi.org/10.1016/j.cub.2009.07.004
Tian Y, Qin X, Lin X, Kaliyaperumal K, Zhou X, Liu J, Ju Z, Tu Z, Liu Y (2015) Sydoxanthone C and acremolin B produced by deep-sea-derived fungus Aspergillus sp. SCSIO Ind09F01. J Antibiot Tokyo 68:703–706. https://doi.org/10.1038/ja.2015.55
Toghueo RMK (2019) Anti-leishmanial and anti-inflammatory agents from endophytes: a review. Nat Products Bioprospect 9:311–328. https://doi.org/10.1007/s13659-019-00220-5
Uzma F, Mohan CD, Hashem A, Konappa NM, Rangappa S, Kamath PV, Singh BP, Mudili V, Gupta VK, Siddaiah CN, Chowdappa S, Alqarawi AA, Abdallah EF (2018) Endophytic fungi-alternative sources of cytotoxic compounds: a review. Front Pharmacol 9:309. https://doi.org/10.3389/fphar.2018.00309
Vasundhara M, Sudhakara RM, Kumar A (2019) Chapter 18 - Secondary metabolites from endophytic fungi and their biological activities, new and future developments in microbial biotechnology and bioengineering. In: Gupta V (ed) Microbial secondary metabolites biochemistry and applications. Elsevier, Amsterdam, pp. 237–258. https://doi.org/10.1016/B978-0-444-63504-4.00018-9
Wen H, Zang Y, Zhu Q, Ouyang S, Luo J, Luo N, Zhu H, Zhang Y (2020) Two new phenolic glucosides from marine-derived fungus Aspergillus sp. Nat Prod Res 0:1–7. https://doi.org/10.1080/14786419.2020.1851226
Wilson D (1995) Endophyte: The evolution of a term, and clarification of its use and definition. Oikos 73:274–276. https://doi.org/10.2307/3545919
Wortman JR, Gilsenan JM, Joardar V et al (2009) The 2008 update of the Aspergillus nidulans genome annotation: a community effort. Fungal Genet Biol 46 Suppl 1:S2–S13. https://doi.org/10.1016/j.fgb.2008.12.003
Xu J, Yi M, Ding L, He S (2019) A review of anti-inflammatory compounds from marine fungi, 2000–2018. Mar Drugs 17:2000–2018. https://doi.org/10.3390/md17110636
Yang Y, Dai JH, Shi CY, Tao L, He SL, Tian Y, Sheng J (2020) Identification of novel targets of aurantiamide acetate against osteoporosis through in silico and in vitro approaches. Mater Express 10:2056–2062. https://doi.org/10.1166/mex.2020.1851
Yang B, Tao H, Qin XC, Wang Z, Dong J, Lin X, Zhou X, Li JL, Tu ZC, Liu Y (2017) Aspergone, a new chromanone derivative from fungus Aspergillus sp. SCSIO41002 derived of mangrove soil sample. J Antibiot Tokyo 70:788–790. https://doi.org/10.1038/ja.2016.169
Yoon CS, Kim DC, Lee DS, Kim KS, Ko W, Sohn JH, Yim JH, Kim YC, Oh H (2014) Anti-neuroinflammatory effect of aurantiamide acetate from the marine fungus Aspergillus sp. SF-5921: inhibition of NF-κB and MAPK pathways in lipopolysaccharide-induced mouse BV2 microglial cells. Int Immunopharmacol 23:568-574. https://doi.org/10.1016/j.intimp.2014.10.006
Zhang C, Hu L, Liu D, Huang J, Lin W (2020) Circumdatin D exerts neuroprotective effects by attenuating LPS-induced pro-inflammatory responses and downregulating acetylcholinesterase activity in vitro and in vivo. Front Pharmacol 11:1–16. https://doi.org/10.3389/fphar.2020.00760
Zhou Y, Chen R, Liu D, Wu C, Guo P, Lin W (2017) Asperlin inhibits LPS-evoked foam cell formation and prevents atherosclerosis in ApoE−/− mice. Mar Drugs 15:358. https://doi.org/10.3390/md15110358
Funding
This work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Finance Code 001), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq: 427497/2018–3), and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG: APQ-02353–17, APQ-00207–18), Brazil.
Author information
Authors and Affiliations
Contributions
AKN, DACP and MSF conceived and conceptualized the idea. AKN, KJN, JCC and MSF performed the investigation and data curation. AKN and JCC performed PRISMA guidelines validation. AKN, KJN and performed the criteria selection. AKN, JCC and MSF built the in-house database. KJN and MSF performed data structure manipulation. AKN draft preparation. DACP, DFD, KJN and MGS contributed to the critical reading and organization of the main points of the review. DACP, DFD and MGS supervised and provided resources and funding acquisition. All of the authors have read the final manuscript and approved the submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
This article is part of a Special Issue to celebrate the 35th anniversary of the Brazilian Journal of Pharmacognosy
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ferreira, M.S., Katchborian-Neto , A., Cruz, J.C. et al. Systematic Review of Anti-inflammatory Agents from Aspergillus Species. Rev. Bras. Farmacogn. 31, 519–530 (2021). https://doi.org/10.1007/s43450-021-00166-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-021-00166-1