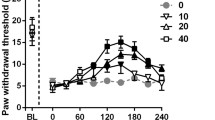
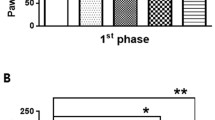
Abstract
Neuropathic pain is a disabling syndrome difficult to manage. Currently, pharmacological treatment of neuropathic patients provides an unsatisfactory relief of pain. Recent reports suggested that piperine has anti-inflammatory and antinociceptive effects. Therefore, the aim of this study was to investigate the antiallodynic effect of piperine, as well as the possible antinociceptive mechanism involved in its antiallodynic effect. Piper nigrum L., Piperaceae, was used to extract the piperine. Spinal nerve ligation L5/L6 model was used to induce allodynia in rats. Intraperitoneal administration of increasing doses of piperine (3.1–100 mg/kg) reduced allodynia in rats. The administration of capsazepine (selective TRPV1 antagonist, 5 and 30 μg/i.t.) and bicuculline (GABAA antagonist, 3 and 30 μg/i.t.), but not SKF-96365 (TRPC antagonist, 3 and 30 μg/i.t.), prevented the piperine-induced (56 mg/kg, i.p.) antiallodynic effect in a dose-dependent manner. HC-030031 (selective TRPA1 antagonist, 3 and 30 μg/i.t.) avoided only partially piperine-induced (56 mg/kg, i.p.) antiallodynic effect. Data suggest that piperine induces antiallodynic effect in neuropathic rats, and its antiallodynic effect involves the activation of TRPV1 and GABAA receptors. Piperine could be useful to treat neuropathic pain in human beings.

Graphical Abstract


Similar content being viewed by others
References
Abbott CA, Malik RA, Van Ross ER, Kulkarni J, Boulton AJ (2011) Prevalence and characteristics of painful diabetic neuropathy in a large community-based diabetic population in the U.K. Diabetes Care 34:2220–2224. https://doi.org/10.2337/dc11-1108
Ahmed M, Rahman MW, Rahman MT, Hossain CF (2002) Analgesic principle from the bark of Careya arborea. Pharmazie 57:698–701
Attal N, Cruccu G, Baron R, Haanpää M, Hansson P, Jensen TS, Nurmikko T, European Federation of Neurological Societies (2010) EFNS guidelines on the pharmacological treatment of neuropathic pain: 2010 revision. Eur J Neurol 17:1113–1e88. https://doi.org/10.1111/j.1468-1331.2010.02999.x
Bang JS, Oh DH, Choi HM, Bong-Jun S, Sung-Jig L, Jung YK, Hyung-In Y, Myung CY, Dae-Hyun H, Kyoung SK (2009) Anti-inflammatory and antiarthritic effects of piperine in human interleukin 1β-stimulated fibroblast-like synoviocytes and in rat arthritis models. Arthritis Res Ther 11:R49. https://doi.org/10.1186/ar2662
Barbosa AF, de Carvalho MG, Smith RE, Sabaa-Srur AUO (2016) Spilanthol: occurrence, extraction, chemistry and biological activities. Rev Bras Farmacogn 26:128–133. https://doi.org/10.1016/j.bjp.2015.07.024
Baron R, Binder A, Wasner G (2010) Neuropathic pain: diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol 9:807–819. https://doi.org/10.1016/S1474-4422(10)70143-5
Boonen J, Bronselaer A, Nielandt J, Veryser L, De Tré G, De Spiegeleer B (2012) Alkamid database: chemistry, occurrence and functionality of plant N-alkylamides. J Ethnopharmacol 142:563–590. https://doi.org/10.1016/j.jep.2012.05.038
Bukhari IA, Alhumayyd MS, Mahesar AL, Gilani AH (2013) The analgesic and anticonvulsant effects of piperine in mice. J Physiol Pharmacol 64:789–794
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL (1994) Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 53:55–63. https://doi.org/10.1016/0165-0270(94)90144-9
Correa EA, Högestätt ED, Sterner O, Echeverri F, Zygmunt PM (2010) In vitro TRPV1 activity of piperine derived amides. Bioorg Med Chem 18:3299–3306. https://doi.org/10.1016/j.bmc.2010.03.013
Da Cruz GMP, Felipe CFB, Scorza FA, da Costa MA, Tavares AF, Menezes ML, de Andrade GM, Leal LK, Brito GA, da Graça Naffah-Mazzacoratti M, Cavalheiro EA, de Barros Viana GS (2013) Piperine decreases pilocarpine-induced convulsions by GABAergic mechanisms. Pharmacol Biochem Behav 104:144–153. https://doi.org/10.1016/j.pbb.2013.01.002
Docherty RJ, Yeats JC, Bevan S, Boddeke HW (1996) Inhibition of calcineurin inhibits the desensitization of capsaicin- evoked currents in cultured dorsal root ganglion neurones from adult rats. Pflugers Arch 431:828–837. https://doi.org/10.1007/s004240050074
Dong Y, Yin Y, Vu S, Yang F, Yarov-Yarovoy V, Tian Y, Zheng J (2019) A distinct structural mechanism underlies TRPV1 activation by piperine. Biochem Biophys Res Commun 516:365–372. https://doi.org/10.1016/j.bbrc.2019.06.039
Gilhotra N, Dhingra D (2014) Possible involvement of GABAergic and nitriergic systems for antianxiety-like activity of piperine in unstressed and stressed mice. Pharmacol Rep 66:885–891. https://doi.org/10.1016/j.pharep.2014.05.008
Hung CY, Tan CH (2018) TRP channels in nociception and pathological pain. Adv Exp Med Biol 1099:13–27. https://doi.org/10.1007/978-981-13-1756-9_2
Jensen TS, Baron R, Haanpää M, Kalso E, Loeser JD, Rice AS, Treede RD (2011) A new definition of neuropathic pain. Pain 152:2204–2205. https://doi.org/10.1016/j.pain.2011.06.017
Kim SH, Chung JM (1992) An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 50:355–363
McNamara FN, Randall A, Gunthorpe MJ (2005) Effects of piperine, the pungent component of black pepper, at the human vanilloid receptor (TRPV1). Br J Pharmacol 144:781–790. https://doi.org/10.1038/sj.bjp.0706040
Mestre C, Pelissier T, Fialip J, Wilcox G, Eschalier A (1994) A method to perform direct transcutaneous intrathecal injection in rats. J Pharmacol Toxicol Methods 32:197–200. https://doi.org/10.1016/1056-8719(94)90087-6
Okumura Y, Narukawa M, Iwasaki Y, Ishikawa A, Matsuda H, Yoshikawa M, Watanabe T (2010) Activation of TRPV1 and TRPA1 by black pepper components. Biosci Biotechnol Biochem 74:1068–1072. https://doi.org/10.1271/bbb.90964
Tallarida RJ (2000) Drug synergism and dose-effect data analysis, 1st edn. Chapman and Hall/CRC, New York
Tasleem F, Azhar I, Ali SN, Perveen S, Mahmood ZA (2014) Analgesic and anti-inflammatory activities of Piper nigrum L. Asian Pac J Trop Med 7S1(S):461–468. https://doi.org/10.1016/S1995-7645(14)60275-3
Touska F, Marsakova L, Teisinger J, Vlachova V (2011) A “cute” desensitization of TRPV1. Curr Pharm Biotechnol 12:122–129. https://doi.org/10.2174/138920111793937826
Tsunozaki M, Lennertz RC, Vilceanu D, Katta S, Stucky CL, Bautista DM (2013) A “toothache tree” alkylamide inhibits Aδ mechanonociceptors to alleviate mechanical pain. J Physiol 591:3325–3340. https://doi.org/10.1113/jphysiol.2013.252106
Van Hecke O, Austin SK, Khan RA, Smith BH, Torrance N (2014) Neuropathic pain in the general population: a systematic review of epidemiological studies. Pain 155:654–662. https://doi.org/10.1016/j.pain.2013.11.013
Venkatesh S, Durga KD, Padmavathi Y, Reddy BM, Mullangi R (2011) Influence of piperine on ibuprofen induced antinociception and its pharmacokinetics. Arzneimittelforschung. 61:506–509. https://doi.org/10.1055/s-0031-1296235
Yasir A, Ishtiaq S, Jahangir M, Ajaib M, Salar U, Khan KM (2018) Biology-oriented synthesis (BIOS) of piperine derivatives and their comparative analgesic and antiinflammatory activities. Med Chem 14:269–280. https://doi.org/10.2174/1573406413666170623083810
Zabela V, Hettich T, Schlotterbeck G, Wimmer L, Mihovilovic MD, Guillet F, Bouaita B, Shevchenko B, Hamburger M, Oufir M (2018) GABAA receptor activity modulating piperine analogs: In vitro metabolic stability, metabolite identification, CYP450 reaction phenotyping, and protein binding. J Chromatogr B Anal Technol Biomed Life Sci 1072:379–389. https://doi.org/10.1016/j.jchromb.2017.11.036
Zimmermann M (1983) Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16:109–110. https://doi.org/10.1016/0304-3959(83)90201-4
Acknowledgments
This work is part of the thesis of Luis Arturo Sánchez-Trujillo and Jorge Luis Mendoza-Monroy, who acknowledges fellowship from Consejo Nacional de Ciencia y Tecnología (604160).
Funding
This work was supported by the Grants Programa de Apoyo a la Investigación y al Posgrado 5000-9143, from Facultad de Química and Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica IN216516 from Dirección General de Asuntos del Personal Académico, Universidad Nacional Autónoma de México and grant A1-S-9698 from Consejo Nacional de Ciencia y Tecnología.
Author information
Authors and Affiliations
Contributions
LAST and JLMM carried the laboratorial work as a part of the academic thesis degree. HIRG and AN participated in the conception and design study. JLBL participated in the supervision of experiments. Data analysis and interpretation data were carried by LAST, JLMM, HIRG, GNQB, JLBL, and AN. GNQB, HIRG, and AN participated in the draft of the manuscript as well the intellectual content of this; final approval of the version was revised and submitted by GNQB, HIRG, and AN.
Corresponding authors
Ethics declarations
All experiments were conducted in accordance with the guidelines on ethical standards for investigation of experimental pain in animals (Zimmermann 1983), with the requirements published by the Secretariat of Agriculture and Rural Development (SAGARPA, Mexico) in the technical specifications for the production, care, and use of laboratory animals (NOM-062-ZOO-1999), and in a compliance with international rules as the guide for the care and use of laboratory animals (National Research Council). In addition, the study was approved by the Ethics Committee for the Use of Animals in Pharmacological and Toxicological Testing (Faculty of Chemistry, UNAM) with the code OFICIO/FQ/CICUAL/199/17.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Electronic Supplementary Material
ESM 1
(DOCX 72 kb)
Rights and permissions
About this article
Cite this article
Sánchez-Trujillo, L.A., Mendoza-Monroy, J.L., Rocha-González, H.I. et al. Antiallodynic Effect of Piperine in Neuropathic Rats. Rev. Bras. Farmacogn. 30, 482–487 (2020). https://doi.org/10.1007/s43450-020-00047-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-020-00047-z